Localized Colon Cancer
| Site: | EHC | Egyptian Health Council |
| Course: | Oncology and Hematological Oncology Guidelines |
| Book: | Localized Colon Cancer |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:47 AM |
Description
"last update: 23 December 2025" Download Guideline
- Executive Summary
|
Strength of the recommendation |
|
|
Diagnostic work-up for localized colon cancer
|
|
|
In the absence of a bowel obstruction or massive haemorrhage, which may constitute indications of an urgent tumour resection, a total colonoscopy is recommended for diagnostic confirmation of colon cancer as there are many advantages of endoscopy including determination and marking of the exact tumour location, biopsy of the lesion, and detection and removal of (further) synchronous precancerous or cancerous lesions.
|
Strong |
|
In cases where complete colonic exploration cannot be carried out before surgery, a complete colonoscopy should be carried out within 3 to 6 months. |
Strong |
|
A complete work-up should be carried out to achieve an accurate histological diagnosis of the primary tumor, assess the baseline characteristics of the patient and determine the extent of the disease. |
Strong |
|
Besides a comprehensive physical examination, blood tests including complete blood count and chemistry profile should be performed. |
Strong |
|
In addition, serum levels of CEA (although not sufficient for colon cancer diagnosis themselves in the absence of a confirmatory tumor biopsy) should be evaluated before surgery and monitored during the follow-up period to help the early detection of metastatic disease. |
Strong |
|
CT of the thoracic, abdominal and pelvic cavities with i.v. contrast administration is the preferred radiological method for the evaluation of the extent of CRC. |
Strong |
|
Contrast-enhanced MRI constitutes the reference test for evaluation of the relationship of locally advanced tumors with surrounding structures or in defining ambiguous hepatic lesions. |
Strong |
|
FDG/PET,with or without integrated CT (PET/CT), does not add significant information to the CT scans on preoperative staging of CRC and is not recommended for routine use in staging of localized CRC except if assisting in interpretation of ambiguous findings. |
Strong |
|
MANAGEMENT OF LOCAL/LOCOREGIONAL DISEASE |
|
|
General recommendations
|
|
|
En bloc endoscopic resection of the polyp is recommended and sufficient for non-invasive (pTis, i.e. intraepithelial or intramucosal) adenocarcinomas. |
Strong |
|
The presence of invasive carcinoma (pT1) in a polyp should require a thorough review with the pathologist and surgeon. High-risk features mandating surgical resection with lymphadenectomy include lymphatic or venous invasion, grade 2 or 3 differentiation, or tumour budding. |
Strong |
|
Laparoscopic colectomy can be safely carried out for colon cancer when technical expertise is available in the absence of contraindications, in view of reduced morbidity, improved tolerance and similar oncological outcomes. |
Conditional |
|
Obstructive CRCs can be treated in one- or two-stage procedures, as indicated. |
Strong |
|
A standard surgical/pathological report should include specimen description and surgical procedure, tumour site and size, macroscopic tumour perforation, histological type and grade, extension into the bowel wall and adjacent organs, distance of cancer from resected margins (proximal, distal and radial), presence or absence of tumour deposits, lymphovascular and/or perineural invasion, tumour budding, site and number of removed and involved regional lymph nodes, and involvement of other organs. |
Strong |
|
Adjuvant therapy options should be fully discussed with the patient, taking into consideration tumour risk of recurrence, expected benefit from chemotherapy and risk of complications. |
Good Practice Statement
|
|
The risk of relapse after a colon cancer resection should be assessed by integrating the TNM staging, biologic profile and number of lymph nodes sampled. |
Strong |
|
Other additional clinicopathological features such as the histological subtype and grading, lymphatic or venous or perineural invasion, lymphoid inflammatory response ,involvement of resection margins and bowel obstruction should be taken into consideration for refining the risk assessment on stage II tumours. |
Strong |
|
Conditional |
|
|
Management of stage II and III disease |
|
|
Stage III disease |
|
|
Combinations of fluoropyrimidines, either 5-FU or capecitabine, and oxaliplatin constitute the basis for stage III colon cancer adjuvant treatment. |
Strong |
|
Strong |
|
|
The length of oxaliplatin-based adjuvant treatment of stage III colon cancer based on the IDEA data should be tailored to 3 or 6 months for CAPOX or 6 months for FOLFOX, and also taking into consideration pathological risk characteristics, patient comorbidity and risk assessment (see Annex 4). |
Strong |
|
For patients not fit for or not tolerating oxaliplatin, either capecitabine or LV5FU2 (de Gramont) infusion are acceptable alternative adjuvant regimens for a 6-month duration. |
Strong |
|
Stage II disease |
|
|
Strong |
|
|
For patients with intermediate risk (non-MMR/MSI + any risk factor except pT4 or multiple intermediate risk factors, regardless of MSI) consider the addition of oxaliplatin (see Annex 3). |
Conditional |
|
Patients with high-risk stage II colon cancer (see Annex 3) are to be considered for 3 months of CAPOX, as the IDEA-pooled analysis showed non-inferiority of 3 months of CAPOX and inferiority of 3 months of FOLFOX when compared with 6 months of FOLFOX, with all the limitations of post-hoc analyses (see Annex 4). |
Strong |
|
Follow up |
|
|
Intensive follow-up allows earlier detection of relapses in patients at risk |
Strong |
|
History and physical examination and CEA level determination are advised every 3-6 months for 2 years and every 6 months for the following 3 years. |
Strong |
|
Colonoscopy must be carried out at year 1 and if advanced adenoma, repeat in 1 year, but if no advanced adenoma, repeat after 3 years, then every 5 years |
Strong |
|
Perform CT scan of chest and abdomen every 6-12 months from date of surgery for a total of 5 years. |
Strong |
|
Other laboratory and radiological examinations are of unproven benefit and must be restricted to patients with suspicious symptoms. |
Conditional |
|
Long-term follow-up, rehabilitation and survivorship care programmes should be implemented, aiming at detection of recurrent or new cancers, assessment and management of late and psychosocial effects and implementation of health promotion measures. |
Strong |
- Recommendations
1. Diagnostic work-up for localized colon cancer
➡️ In the absence of a bowel obstruction or massive haemorrhage, which may constitute indications of an urgent tumour resection, a total colonoscopy is recommended for diagnostic confirmation of colon cancer as there are many advantages of endoscopy including determination and marking of the exact tumour location, biopsy of the lesion, and detection and removal of (further) synchronous precancerous or cancerous lesions.
Strong recommendation, High grade evidence (5).
➡️ In cases where complete colonic exploration cannot be carried out before
surgery, a complete colonoscopy should be carried out within 3 to 6 months.
Strong recommendation, Low grade evidence (5).
➡️ A complete work-up should be carried out to achieve an accurate histological diagnosis of the primary tumor, assess the baseline characteristics of the patient and determine the extent of the disease
Strong recommendation, High grade evidence (5)
➡️ Besides a comprehensive physical examination, blood tests including complete blood count and chemistry profile should be performed.
Strong recommendation, High grade evidence (5).
➡️ In addition, serum levels of CEA (although not sufficient for colon cancer diagnosis themselves in the absence of a confirmatory tumor biopsy) should be evaluated before surgery and monitored during the follow-up period to help the early detection of metastatic disease.
Strong recommendation, Low grade evidence (6-8)
➡️ CT of the thoracic, abdominal and pelvic cavities with i.v. contrast administration is the preferred radiological method for the evaluation of the extent of CRC.
Strong recommendation, High grade evidence (9,10)
➡️ Contrast-enhanced MRI constitutes the reference test for evaluation of the relationship of locally advanced tumors with surrounding structures or in defining ambiguous hepatic lesions.
Strong recommendation, High grade evidence (11).
➡️ FDG/PET,with or without integrated CT (PET/CT), does not add significant information to the CT scans on preoperative staging of CRC and is not recommended for routine use in staging of localized CRC except if assisting in interpretation of ambiguous findings.
Strong recommendation, High grade evidence (8,12).
2. MANAGEMENT OF LOCAL/LOCOREGIONAL DISEASE
General recommendations
➡️ En bloc endoscopic resection of the polyp is recommended and sufficient for non-invasive (pTis, i.e. intraepithelial or intramucosal) adenocarcinomas
Strong recommendation, Low grade evidence (13-15)
➡️ The presence of invasive carcinoma (pT1) in a polyp should require a thorough review with the pathologist and surgeon. High-risk features mandating surgical resection with lymphadenectomy include lymphatic or venous invasion, grade 2 or 3 differentiation, or tumour budding.
Strong recommendation, Low grade evidence (16-18)
➡️ Laparoscopic colectomy can be safely carried out for colon cancer when technical expertise is available in the absence of contraindications, in view of reduced morbidity, improved tolerance and similar oncological outcomes.
Conditional recommendation, Hogh grade evidence (19,20)
➡️ Obstructive CRCs can be treated in one- or two-stage procedures, as indicated.
Strong recommendation, Low grade evidence (21,22)
➡️ A standard surgical/pathological report should include specimen description and surgical procedure, tumour site and size, macroscopic tumour perforation, histological type and grade, extension into the bowel wall and adjacent organs, distance of cancer from resected margins (proximal, distal and radial), presence or absence of tumour deposits, lymphovascular and/or perineural invasion, tumour budding, site and number of removed and involved regional lymph nodes, and involvement of other organs.
Strong recommendation, Low grade evidence (16,23)
➡️ Adjuvant therapy options should be fully discussed with the patient, taking into consideration tumour risk of recurrence, expected benefit from chemotherapy and risk of complications.
Good Practice Statement
➡️ The risk of relapse after a colon cancer resection should be assessed by integrating the TNM staging, biologic profile and number of lymph nodes sampled.
Strong recommendation, low grade evidence (24).
➡️ Other additional clinicopathological features such as the histological subtype and grading, lymphatic or venous or perineural invasion, lymphoid inflammatory response ,involvement of resection margins and bowel obstruction should be taken into consideration for refining the risk assessment on stage II tumours.
Strong recommendation, low grade evidence (25-27).
➡️ It can be generalised that benefits of treatment with fluoropyrimidines alone or with oxaliplatin, seems to be more limited with a higher likelihood for toxicity in older patients.
Conditional recommendation, very low grade evidence (28).
Management of stage II and III disease
Stage III disease
· Combinations of fluoropyrimidines, either 5-FU or capecitabine, and oxaliplatin constitute the bases for stage III colon cancer adjuvant treatment
Strong recommendation, high grade evidence (29-31).
· The length of oxaliplatin-based adjuvant treatment of stage III colon cancer based on the IDEA data should be tailored to 3 or 6 months for CAPOX or 6 months for FOLFOX, and also taking into consideration pathological risk characteristics, patient comorbidity and risk assessment (see Annex 4).
Strong recommendation, high grade evidence (32).
· Further adaptation of the treatment according to risk subgroups: 3 months for CAPOX or 3-6 months for folfox (T 1-3 N1 disease), 3-6 months for CAPOX or 6 months for FOLFOx (T4 or N2 disease)based on IDEA collaboration should be made with caution, since this was based on a post-hoc analysis, non-significant for interaction (see Annex 4).
Strong recommendation,high grade evidence (32)
· For patients not fit for or not tolerating oxaliplatin, either capecitabine or LV5FU2 (de Gramont) infusion are acceptable alternative adjuvant regimens for a 6-month duration.
Strong recommendation, high grade evidence (32).
Stage II disease
· For patients with low-risk stage II colon cancer (see Annex 3), follow-up is recommended or consider capecitabine (6 mo) or 5-FU/leucovorin (6 mo)
Strong recommendation, high grade evidence (33-37).
Conditional recommendation, high grade evidence (33-37).
· Patients with high-risk stage II colon cancer (see Annex 3) are to be considered for 3 months of CAPOX, as the IDEA-pooled analysis showed non-inferiority of 3 months of CAPOX and inferiority of 3 months of FOLFOX when compared with 6 months of FOLFOX, with all the limitations of post-hoc analyses (see Annex 4).
Strong recommendation, high grade evidence (32).
· It is important to start adjuvant chemotherapy as soon as possible after surgery and ideally not later than 8 weeks.
Strong recommendation, high grade evidence (38-40).
➡️ Follow up (41-47)
· Intensive follow-up allows earlier detection of relapses in patients at risk
Strong recommendation, high grade evidence.
· History and physical examination and CEA level determination are advised every 3-6 months for 2 years and every 6 months for the following 3 years
Strong recommendation, high grade evidence.
· Colonoscopy must be carried out at year 1 and every 3-5 years thereafter, looking for metachronous adenomas and cancers.
Strong recommendation, moderate grade evidence.
l Perform CT scan of chest and abdomen every 6-12 months from date of surgery for a total of 5 years.
Strong recommendation, high grade evidence.
· Other laboratory and radiological examinations are of unproven benefit and must be restricted to patients with suspicious symptoms.
Conditional recommendation, very low grade evidence.
· Long-term follow-up, rehabilitation and survivorship care programmes should be implemented, aiming at detection of recurrent or new cancers, assessment and management of late and psychosocial effects and implementation of health promotion measures.
Strong recommendation, low grade evidence.
- Acknowledgments
- We would like to acknowledge the Oncology Committee of the Egyptian Health Council (EHC) Guidelines, for adapting these Guidelines.
- Chair of the Oncology Committee of Egyptian Health Council Guidelines: Prof Hussein Khaled.
- The Oncology Committee Members: Emad Hamada, Samir Shehata, Hesham Elghazaly, Hesham Tawfik, Fouad Abuotaleb, Ebtesam Saad Eldin, Ihab Khalil, Khaled Abdelkarim, Lobna EZZ Elarab, Mary Gamal, Mohamed Abdel Mooti, Mohamed Gamil, Nervana Hussein, Ola Khorshid, Omar Sherif Omar, Rasha Fahmi, Rasha Shaltout, Yousri Wasef & Yousri Rostom.
- Chair of the GIT Cancer Scientific Committee: Prof Yousri Rostom
- The gastric cancer Scientific Group Members: (Alphabitical order): Ahmed EL Kassed, Fouad Aboutaleb, Khaled Abdelkarim, Manal EL Mahdy, Omar S. Omar, and Yousri Wasef.
- Abbreviations
5FU 5 fluorouracil
CEA carcinoembryonic antigen
CT computed tomography
CRC colo-rectal cancer
EMVI extramural venous invasion
FDG-PET scan (Fluorodeoxyglucose-Positron Emission Tomography
MDT multidisciplinary team
MRI magnetic resonance imaging
- Introduction
Colo-rectal cancer is the 7th most common cancer in the Egyptian population with more than 5900 newly diagnosed cases and more than 3000 deaths in 2022 (1).
- Purpose and scope
These guidelines will help to improve the quality of care for localized colon cancer patients via providing a uniform standard of care across the country to help in early diagnosis and treatment for localized colon cancer, with less aggressive treatment options and improved clinical outcomes. These guidelines cover primary diagnosis, staging, treatment and follow-up of localized colon cancer patients.
- Target audience
Clinicians who are involved in the care and treatment of patients with localized colon cancer, including medical oncologists, radiation oncologists, clinical oncologists, onco- and gastrointestinal surgeons, radiologists and pathologists.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
- Selecting only evidence-based guidelines (guidelines must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence). - Selecting only national and/or international guidelines.
- Specific range of dates for publication (using Guidelines published or updated 2015 and later).
- Selecting peer reviewed publications only.
- Selecting guidelines written in English language.
- Excluding guidelines written by a single author not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
- Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided a cutoff point or rank the guidelines (any guideline scoring above 50% on the rigor dimension was retained)
The NCCN, ESMO, NICE guidelines are the main sources used while formulating the national guidelines for colon cancer (2-4).
- Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available through the on the following sites:
. GRADE working group:http://www.gradeworkinggroup.org
. GRADE online training modules: http://cebgrade.mcmaster.ca/
Table 1: Quality of evidence in GRADE
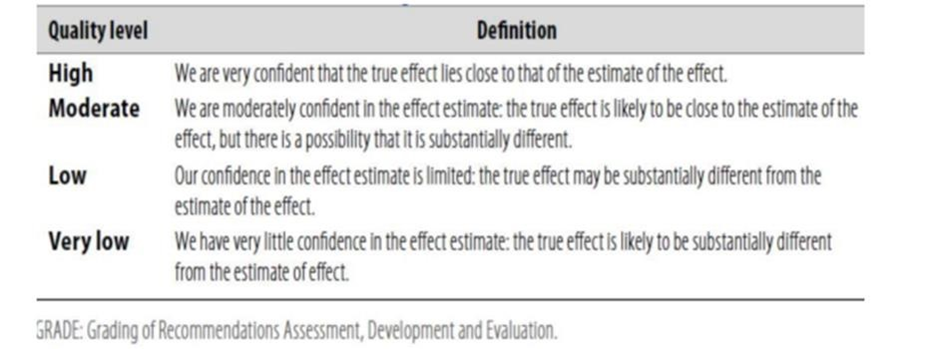
Table 2: Significance of the four levels of evidence

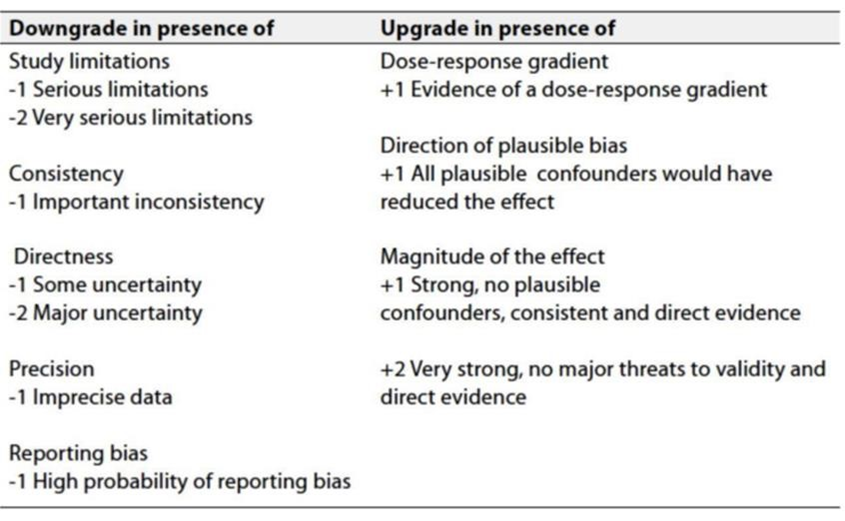
Table 3: Factors that determine how to upgrade or downgrade the quality of evidence.
➡️ The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation:
Strong recommendations: With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations: These are made when there is greater uncertainty about the four factors above (Table 2) or if local adaptation must account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations; when there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Research Gaps
• Evaluation of real world data on the use on new targeted and immune-therapeutic agents in colon cancer in Egypt.
• Cost effective analysis of new therapeutic agents in Egypt.
• Define the molecular biologic profiles of our patients.
- Update of the guideline
• This guideline will be updated whenever there is new evidence.
- Annexes
Annex 1.
Annex 2.
DOSES OF ADJUVANT CHEMOTHERAPY REGIMENS
- mFOLFOX 6
Oxaliplatin 85 mg/m2 IV, day 1a Leucovorin 400 mg/m2 IV, day 1b
5-FU 400 mg/m2 IV bolus on day 1, followed by 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) continuous infusion.
Repeat every 2 weeks.1,2,3
- Capecitabine
Capecitabine 1000–1250c mg/m2 PO twice daily for 14 days every 3 weeks
- CAPEOX
Oxaliplatin 130 mg/m2 IVa day 1
Capecitabine 1000c mg/m2 PO twice daily for 14 days every 3 weeks
- 5-FU/leucovorin
- Leucovorin 500 mg/m2 given as a 2-hour infusion and repeated weekly x 6. 5-FU 500 mg/m2 given bolus 1 hour after the start of leucovorin and repeated 6 x weekly. Every 8 weeks for 4 cycles.
- Simplified biweekly infusional 5-FU/leucovorin (sLV5FU2)
Leucovorin 400b mg/m2 IV day 1, followed by 5-FU bolus 400 mg/m2, followed by 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) continuous infusion. Repeat every 2 weeks.
Annex 3.
l When determining if adjuvant therapy should be administered for stage II, the following should be taken into consideration:
- Number of lymph nodes analyzed after surgery (<12)
- Poor prognostic features (eg, poorly differentiated histology [exclusive of those that are MSI-H]; lymphatic/vascular invasion; bowel obstruction; PNI; localized perforation; close, indeterminate, or positive margins; or high tumor budding)

Annex 4. Recommendations from the IDEA study for adjuvant treatment of colon cancer (32).
➡️Diagnostic work-up for localized colon cancer
|
Other additional clinicopathological features such as the histological subtype and grading, lymphatic or venous or perineural invasion, lymphoid inflammatory response, involvement of resection margins and < 12 lymph nodes examined should be taken into consideration for refining the risk assessment on stage II tumours. |
- References
1. https://gco.iarc.who.int/media/globocan/factsheets/populations/818-egypt-fact-sheet.pdf
2. https://www.nccn.org/guidelines/guidelines-detail?category=1&id=1453.
3. https://www.esmo.org/guidelines/guidelines-by-topic/esmo-clinical-practice-guideline
4. https://www.nice.org.uk/about/what-we-do/our-programmes/nice-guidance/nice-guidelines
5. Hamilton W, Round A, Sharp D, Peters TJ. Clinical features of colorectal cancer before diagnosis: a population-based case-control study. Br J Cancer. 2005;93:399-405.
6. Konishi T, Shimada Y, Hsu M, et al. Association of preoperative and postoperative serum carcinoembryonic antigen and colon cancer outcome. JAMA Oncol. 2018;4(3):309-315.
7. Duffy MJ, van Dalen A, Haglund C, et al. Clinical utility of biochemical markers in colorectal cancer: European Group on Tumour Markers (EGTM) guidelines. Eur J Cancer. 2003;39:718-727.
8. Locker GY, Hamilton S, Harris J, et al. ASCO 2006 update of recommendations for the use of tumor markers in gastrointestinal cancer. J Clin Oncol. 2006;24:531305327.
9. Horton KM, Abrams RA, Fishman EK. Spiral CTof colon cancer: imaging features and role in management. Radiographics. 2000;20:419-430.
10. Nerad E, Lahaye MJ, Maas M, et al. Diagnostic accuracy of CT for local staging of colon cancer: a systematic review and meta-analysis. AJR Am J Roentgenol. 2016;207:984-995.
11. Sahani DV, Bajwa MA, Andrabi Y, et al. Current status of imaging and merging techniques to evaluate liver metastases from colorectal carcinoma. Ann Surg. 2014;259:861-872.
12. Niekel MC, Bipat S, Stoker J. Diagnostic imaging of colorectal liver metastases with CT, MR imaging, FDG PET and/or FDG PET/CT: a meta-analysis of prospective studies including patients who have not previously undergone treatment. Radiology. 2010;257:674-684.
13. Ferlitsch M, Moss A, Hassan C, et al. Colorectal polypectomyand endoscopic mucosal resection (EMR): European Society of Gastrointestinal Endoscopy (ESGE) clinical guideline. Endoscopy. 2017;49:270-297.
14. Aarons CB, Shanmugan S, Bleier JI. Management of malignant colon polyps: current status and controversies. World J Gastroenterol. 2014;20:16178-16183.
15. Haggitt RC, Glotzbach RE, Soffer EE, Wruble LD. Prognostic factors in colorectal carcinomas arising in adenomas: implications for lesions removed by endoscopic polypectomy. Gastroenterology. 1985;89:328-336.
16. Lugli A, Kirsch R, Ajioka Y, et al. Recommendations for reporting tumor budding in colorectal cancer based on the International Tumor Budding Consensus Conference (ITBCC) 2016. Mod Pathol. 2017;30: 1299-1311.
17. Backes Y, Elias SG, Groen JN, et al. Histologic factors associated with need for surgery in patients with pedunculated T1 colorectal carcinomas Gastroenterology. 2018;154:1647-1659.
18. Bujanda L, Cosme A, Gil I, Arenas-Mirave JI. Malignant colorectal polyps. World J Gastroenterol. 2010;16:3103-3111.
19. Nelson H, Sargent DJ, Wieand HS, et al. A comparison of laparoscopically assisted and open colectomy for colon cancer. N Engl J Med. 2004;350:2050-2059.
20. Hewett PJ, Allardyce RA, Bagshaw PF, et al. Short-term outcomes of the Australasian randomized clinical study comparing laparoscopic and conventional open surgical treatments for colon cancer: the ALCCaS trial. Ann Surg. 2008;248:728-738.
21. Ribeiro IB, Bernardo WM, Martins BDC, et al. Colonic stent versus emergency surgery as treatment of malignant colonic obstruction in the palliative setting: a systematic review and meta-analysis [published correction appears in Endosc Int Open 2018;6:C1] Endosc Int Open. 2018;6:E558-E567.
22. van Hooft JE, Veld JV, Arnold D, et al. Self-expandable metal stents for obstructing colonic and extracolonic cancer: European Society of Gastrointestinal Endoscopy (ESGE) Guideline e Update 2020. Endoscopy. 2020;52:389-407.
23. Washington MK, Berlin J, Branton P, et al. Protocol for the examination of specimens from patients with primary carcinoma of the olon and rectum. Arch Pathol Lab Med. 2009;133:1539-1551.
24 Atkin WS, Valori R, Kuipers EJ, et al. European guidelines for quality assurance in colorectal cancer screening and diagnosis. First edition colonoscopic surveillance following adenoma removal. Endoscopy.2012;44:E151-E163.
25 Benson III AB, Schrag D, Somerfield MR, et al. American Society of Clinical Oncology recommendations on adjuvant chemotherapy for stage II colon cancer. J Clin Oncol 2004;16:3408-3419.
26 Figueredo A, Charette ML, Maroun J, et al. Adjuvant therapy for stage II colon cancer: a systematic review from the Cancer Care Ontario Program in evidence-based care’s gastrointestinal cancer disease site group. J Clin Oncol 2004;16:3395-3407.
27 Gill S, Loprinzi CL, Sargent DJ, et al. Pooled analysis of fluorouracil-based adjuvant therapy for stage II and III colon cancer: who benefits and by how much? J Clin Oncol 2004;22:1797-1806.
28 Haller DG, O’Connell MJ, Cartwright TH, et al. Impact of age and medical comorbidity on adjuvant treatment outcomes for stage III colon cancer: a pooled analysis of individual patient data from four randomized, controlled trials. Ann Oncol. 2015;26:715-724.
29 Andre T, Boni C, Navarro M, et al. Improved overall survival with oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment in stage II or III colon cancer in the MOSAIC trial. J Clin Oncol. 2009;27: 3109-3116.
30 Kuebler JP, Wieand HS, O’Connell MJ, et al. Oxaliplatin combined with weekly bolus fluorouracil and leucovorin as surgical adjuvant chemotherapy for stage II and III colon cancer: results from NSABP C-07. J Clin Oncol. 2007;25:2198-2204.
31 Haller DG, Tabernero J, Maroun J, et al. Capecitabine plus oxaliplatin compared with fluorouracil and folinic acid as adjuvant therapy for stage III colon cancer. J Clin Oncol. 2011;29:1465-1471.
32. Axel Grothey, MD, and Alan P. Venook, Optimizing Adjuvant Therapy for Localized Colon Cancer and Treatment Selection in Advanced Colorectal Cancer, MD J Natl Compr Canc Netw 2018;16(5.5):611–615.
33. Ribic CM, Sargent DJ, Moore MJ, et al. Tumor microsatellite-instability status as a predictor of benefit from fluorouracil-based adjuvant chemotherapy for colon cancer. N Engl J Med. 2003;349:247-257.
34. Sargent DJ, Marsoni S, Monges G, et al. Defective mismatch repair as a predictive marker for lack of efficacy of fluorouracil-based adjuvant therapy in colon cancer. J Clin Oncol. 2010;28:3219-3226.
35. Sinicrope FA, Foster NR, Thibodeau SN, et al. DNA mismatch repair status and colon cancer recurrence and survival in clinical trials of 5-fluorouracil-based adjuvant therapy. J Natl Cancer Inst. 2011;103: 863-875.
36. Tejpar S, Saridaki Z, Delorenzi M, et al. Microsatellite instability,prognosis and drug sensitivity of stage II and III colorectal cancer: more complexity to the puzzle. J Natl Cancer Inst. 2011;103:841-844.
37. Kim JE, Hong YS, Kim HJ, et al. Defective mismatch repair status was not associated with DFS and OS in stage II colon cancer treated with adjuvant chemotherapy. Ann Surg Oncol. 2015;22(Suppl 3):S630-S637.
38. Des Guetz G, Nicolas P, Perret GY, et al. Does delaying adjuvant chemotherapy after curative surgery for colorectal cancer impair survival? A meta-analysis. Eur J Cancer. 2010;46:1049-1055.
39. Hershman D, Hall MJ, Wang X, et al. Timing of adjuvant chemotherapy initiation after surgery for stage III colon cancer. Cancer. 2006;107:2581-2588.
40 . Gao P, Huang XZ, Song YX, et al. Impact of timing of adjuvant chemotherapy on survival in stage III colon cancer: a populationbased study. BMC Cancer. 2018;18:234.
41. Wille-Jorgensen P, Syk I, Smedh K, et al. Effect of more vs less frequent follow-up testing on overall and colorectal cancerspecific mortality in patients with stage II or III colorectal cancer: the COLOFOL randomized clinical trial. JAMA. 2018;319: 2095-2103.
42. Alhayek-Aí M, López-Calviño B, Pértega-Díaz S, et al. Intensive followup strategies improve outcomes in nonmetastatic colorectal cancer patients after curative surgery: a systematic review and meta-analysis. Ann Oncol. 2014;26:644-656.
43. Chau I, Allen MJ, Cunningham D, et al. The value of routine serum carcino-embryonic antigen measurement and computed tomography in the surveillance of patients after adjuvant chemotherapy for colorectal cancer. J Clin Oncol. 2004;22:1420-1429.
44. Tsikitis VL, Malireddy K, Green EA, et al. Postoperative surveillance recommendations for early stage colon cancer based on results from the clinical outcomes of surgical therapy trial. J Clin Oncol. 2009;27: 3671-3676.
45. Rex DK, Kahi CJ, Levin B, et al. Guidelines for colonoscopy surveillance after cancer resection: a consensus update by the American Cancer Society and the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2006;130:1865-1871.
46. Howell D, Hack TF, Oliver TK, et al. Models of care for post-treatment follow-up of adult cancer survivors: a systematic review and quality appraisal of the evidence. J Cancer Surviv. 2012;6:359-371.
47. Grunfeld E, Earle CC. The interface between primary and oncology specialty care: treatment through survivorship. J Natl Cancer Inst Monogr. 2010;2010:25-30.