diabetic retinopathy and maculopathy management
| Site: | EHC | Egyptian Health Council |
| Course: | Ophthalmology Guidelines |
| Book: | diabetic retinopathy and maculopathy management |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 25 December 2025" Download Guideline
- EXECUTIVE SUMMARY
In PDR and severe NPDR
Severe non proliferative DR
· compliant patient without diabetic maculopathy: regular follow up
Recommendation: conditional
· non-compliant patient without diabetic maculopathy: laser PRP
Recommendation: Strong
PDR and high risk PDR without diabetic maculopathy and no abundant fibrosis
· Non-compliant patient: laser PRP
Recommendation: strong
· Compliant patient: Laser PRP or antiVEGF injection within 4 weeks;
Recommendation: strong
PDR and high risk PDR with diabetic maculopathy
· Compliant patient: antiVEGF before PRP
Recommendation: moderate
Non compliant patient: laser PRP with focal/grid laser for the macula
Recommendation: moderate
NCI-DME:
· Vision 6/12 or better in compliant patients: observe with control of risk factors.
Recommendation: strong
· Vision 6/12 or better in non-compliant patients: laser treatment based on angiography and OCT
Recommendation: strong
· Vision less than 6/12: no treatment and investigate to exclude ischemic maculopathy, if present, IVI of anti VEGF or steroids
Recommendation: consensous
CI-DME
· Vision better than 6/12 compliant patient: observe with control of risk factors
Recommendation: Conditional
· Vision better than 6/12 non compliant patient: laser treatment
Recommendation: strong
· Vision less than 6/12 in compliant patient: AntiVEGF
Recommendation: strong
· Vision less than 6/12 in non compliant patient: laser
Recommendation: strong
· Vision less than 6/12 with signs of vitreo-retinal traction: Vitrectomy +/-AntiVEGF
Recommendation: strong
· Refractory DME: received a minimum of three monthly injections of AntiVEGF with poor anatomical and functional response in compliant patient: intra vitreal steroids/vitrectomy
only in places with subspecialty retinal service.
Recommendation: strong
Introduction:
Diabetes mellitus (DM) is an alarming public health issue globally and its prevalence is increasing heavily in developing countries [1-4]. The number of diabetics between 20 and 79 years of age is estimated to be 415 million people in the world and is expected to rise to 642 million in 2040.[5] In Egypt, DM is a dilemma and a fast-expanding concern. According to the International Diabetes Federation (IDF), the prevalence of DM among Egyptian adults is 15.2%, which may be an underestimation [6]. Diabetic retinopathy (DR) is one of the major complications of diabetes and leading cause of visual impairment or blindness. One of the most devastating complications of diabetes is diabetic retinopathy, which can be potentially blinding [7-14]. Evidence through the years reveals that almost all type I and around 60% of type II diabetic patients develop some degree of retinopathy within 20 years after diagnosis [15]. Given the increasing number of diabetics worldwide, developing countries are expected to receive the greatest toll [14].
- Recommendations
Severe non proliferative DR
· compliant patient without diabetic maculopathy: regular follow up
Recommendation: conditional
Evidence level: high [19-21]
· non-compliant patient without diabetic maculopathy: laser PRP
Recommendation: Strong
Evidence level: high [19-21]
PDR and high risk PDR without diabetic maculopathy and no abundant fibrosis
· Non-compliant patient: laser PRP
Recommendation: strong
Evidence level: high [22-27]
· Compliant patient: Laser PRP or antiVEGF injection within 4 weeks;
Recommendation: strong
Evidence level: high [28-121]
PDR and high risk PDR with diabetic maculopathy
· Compliant patient: antiVEGF before PRP
Recommendation: moderate
Evidence level: moderate
Non compliant patient: laser PRP with focal/grid laser for the macula
Recommendation: moderate
Evidence level: high [22-121]
NCI-DME:
· Vision 6/12 or better in compliant patients: observe with control of risk factors.
Recommendation: conditional
Evidence level: high [22-121]
· Vision 6/12 or better in non-compliant patients: laser treatment based on angiography and OCT
Recommendation: strong
Evidence level: high [22-121]
· Vision less than 6/12: no treatment and investigate to exclude ischemic maculopathy, if present, IVI of anti VEGF or steroids
Recommendation: consensous
Evidence level: low [22-121]
CI-DME
· Vision better than 6/12 compliant patient: observe with control of risk factors
Recommendation: Conditional
Evidence level: low [28-121]
· Vision better than 6/12 non compliant patient: laser treatment
Recommendation: strong
Evidence level: high [28-121]
· Vision less than 6/12 in compliant patient: AntiVEGF
Recommendation: strong
Evidence: high [28-121]
· Vision less than 6/12 in non compliant patient: laser
Recommendation: strong
Evidence: low [28-121]
· Vision less than 6/12 with signs of vitreo-retinal traction: Vitrectomy +/-AntiVEGF
Recommendation: strong
Evidence: very low [22-118]
Refractory DME: received a minimum of three monthly injections of AntiVEGF with poor anatomical and functional response in compliant patient: intra vitreal steroids/vitrectomy only in places with subspecialty retinal service.
Recommendation: strong
Evidence level: low [22-118]
Remarks on treatment
1. Laser PRP :
• Laser for mild PDR (neovessels less than 1/3 DD) should be between 1200-1800 shot 500um size, moderate PDR (NVD: greater than third of disc diameter, and forward NVD extending beyond the disc margin or NVE: complexes in all quadrants, forward NVE in any quadrant) 2000-2500 shot, severe PDR (Large, NVE complexes in any quadrant, NVE with tractional retinal detachment, large, forward NVD covering whole optic disc surface, NVD with tractional retinal detachment.These cases are high risk of continued traction and haemorrhagic complications following PRP) full PRP 3000 sots
Evidence level: high [22-118]
• If possible, PRP should be delivered on the same day of the diagnosis of high‑risk PDR and if not, within 2 weeks from the time of diagnosis.
Evidence level moderate [22-118]
• PRP should be postponed until clinically significant macular edema (CSME) is treated.
Evidence level moderate [22-118]
• Coexisting high-risk PDR and CSME should be treated with combined PRP plus intravitreal antivascular endothelial growth factor (VEGF) injection or macular laser photocoagulation (MPC).
Evidence level: very low [22-118]
• In cases with fresh vitreous hemorrhage, PRP is usually applied after intravitreal injection of anti‑VEGF when the ocular media is clear.
Evidence level: very low [22-118]
• In patients with florid type DR, PRP may be applied with shorter intervals (3‑5 days between PRP sessions instead of 1‑4 weeks).
Evidence level: very low [22-118]
Recommendations for follow up laser therapy:
• First follow up should be at three to four months – ideally at two months
Evidence level: high [22-118]
• PDR: Retreatment is required if vessels have not regressed, or further new vessels develop [22-121]
2. AntiVEGF monotherapy for PDR
• Frequent, regular, follow up i.e., every two to three months for at least a year is an essential prerequisite for the treatment of PDR with AntiVEGF agents.
• Repeat injections, if required. [119-120]
3. Vitrectomy
Recommendations for treatment
• Early vitrectomy (within three months) is indicated for people with Type 1 DM who develop severe vitreous haemorrhage in whom severe PDR is suspected
Evidence level: high [123-127]
• Consider early vitrectomy in eyes where PDR does not respond to extensive and aggressive laser PRP
Evidence level: moderate [123-127]
• Consider vitreoretinal surgery to relieve vitreoretinal traction if the macular is detached or threatening to detach, to salvage some vision
Evidence level: very low [123-127]
• Consider combined cataract surgery / vitrectomy in eyes with DR and/or DME with lens opacities, to enable subsequent management of PDR and/or DME.
Evidence: moderate [123-127]
Recommendations for practice
• Counselling patients before treatment, regarding improving the control of their diabetes and comorbidities is essential.
The procedure should be explained, including the likely outcome as well as the need for and timing and frequency of follow up, the likelihood of repeat treatment and the need for lifelong care. [123-127]
Intravitreal injection before vitrectomy
• Patients with PDR and active neovascularization who have been planned for vitrectomy may receive intravitreal anti‑VEGF injection within one week before surgery to minimize intraoperative and early
postoperative bleeding.
Evidence level: high [123-127]
• Patients with advanced DR and active fibrovascular tissue who are vitrectomy candidates may receive intravitreal anti‑VEGF injection within one week before the surgery to minimize the risk of bleeding during and after the surgical procedure.
Evidence level: high[123-127]
• Extensive fibrovascular tissue increases the risk of traction retinal detachment following intravitreal injection of anti‑VEGF drugs; the time interval between the injection and vitrectomy should be not more than 2‑3 days in such cases.
Evidence level: moderate [123-127]
➡️Updates:
To keep these recommendations up to date and ensure its validity it will be periodically updated. This will be done whenever new strong evidence is available and necessitates updating.
- ACKNOWLEDGEMENTS
The Ophthalmology Committee of National Egyptian Guidelines, Ministry of Health and
Ophthalmology
Chair of the committee: Prof. Hany S. Nasr.
Committee members professors: A. Farag, A. Alghoneimy, E. El-Toukhy, K. Rizk, M. Maher, M. Zaatar, M. Mourad, R. Zaki, W. Ahmad
- ABBREVIATIONS
Anti-VEGF: See Anti-vascular endothelial growth factor.
BCVA: best corrected visual acuity
CI-DME: center-involving macular edema
CSME: clinically significant macular edema
DME: diabetic macular edema
DR: diabetic retinopathy
ERM: epiretinal membrane
ETDRS: Early Treatment for Diabetic Retinopathy Study
FFA: fundus fluorescein angiography
IRMA: Intraretinal microvascular abnormalities.
IVI: intravitreal injections
NCI-DME: non center-involving macular edema
NPDR: non proliferative diabetic retinopathy
OCT: optical coherence tomography
PDR: proliferative diabetic retinopathy
PRP: pan retinal photocoagulation
VEGF: vascular endothelial growth factor
- GLOSSARY
Anti-vascular endothelial growth factor (anti-VEGF): Substances that inhibit the action of vascular endothelial growth factor protein.
Clinically significant macular edema (CSME): Retinal thickening at or within 500 µm of the center of the macula; and/or hard exudates at or within 500 µm of the center of the macula, if associated with thickening of the adjacent retina; and/or a zone or zones of retinal thickening 1 disc area in size, any part of which is within 1 disc diameter of the center of the macula.
Diabetes mellitus: According to the American Diabetes Association Expert Committee on the Diagnosis and Classification of Diabetes Mellitus, the criteria for the diagnosis of diabetes mellitus are as follows.
· Fasting plasma glucose equal to or exceeding 126 mg/dL (7.0 mmol/L). Fasting is defined as no caloric intake for at least 8 hours.
or
· Symptoms of hyperglycemia and a casual plasma glucose concentration equal to or exceeding 200 mg/dL (11.1 mmol/L). “Casual” is defined as any time of day without regard to time since last meal. The classic symptoms of hyperglycemia include polyuria, polydipsia, and unexplained weight loss. or
· A plasma glucose measurement at 2 hours postload equal to or exceeding 200 mg/dL (11.1 mmol/L) during an oral glucose tolerance test. The test should be performed as described by the World Health Organization, using a glucose load containing the equivalent of 75 g anhydrous glucose dissolved in water. However, the expert committee has recommended against oral glucose tolerance testing for routine clinical use. (Source: Report of the Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 2008;31 (suppl):55-60.)
Diabetic macular edema: The accumulation of fluid in the macula due to leaky blood vessels.
Early proliferative diabetic retinopathy (i.e., proliferative retinopathy without DRS high-risk characteristics): New vessels that do not meet the criteria of high-risk proliferative retinopathy.
Focal photocoagulation: A laser technique directed to abnormal blood vessels with specific areas of focal leakage (i.e., microaneurysms) to reduce chronic fluid leakage in patients with macular edema.
Grid photocoagulation: A laser technique in which a grid pattern of scatter burns is applied in areas of diffuse macular edema and nonperfusion. Typically, fluorescein angiograms of these areas show a diffuse pattern rather than focal leakage.
High-risk proliferative diabetic retinopathy: New vessels on or within 1 disc diameter of the optic disc about one-quarter to one-third disc area, with or without vitreous or preretinal hemorrhage; or vitreous and/or preretinal hemorrhage accompanied by new vessels either on the optic disc less than ¼ disc area, or new vessels elsewhere equaling or exceeding one-quarter disc area.
Intraretinal microvascular abnormalities: Tortuous intraretinal vascular segments, varying in caliber from barely visible to 31 µm in diameter (one-quarter the width of a major vein at the disc margin); they occasionally can be larger. Intraretinal microvascular abnormalities may be difficult to distinguish from neovascularization.
Macular edema: Thickening of the retina within 1 or 2 disc diameters of the center of the macula. (See Clinically significant macular edema.) Any other thickening of the macula not within this area is non-CSME.
Mild nonproliferative diabetic retinopathy: At least 1 microaneurysm and less than moderate nonproliferative diabetic retinopathy.
Moderate nonproliferative diabetic retinopathy: Hemorrhages and/or microaneurysms greater than standard photograph 2A, and/or soft exudates, venous beading, or IRMA present but less than severe nonproliferative retinopathy.
Moderate visual loss: The loss of 15 or more letters on the ETDRS visual acuity chart, or doubling of the visual angle (e.g., 6/6 to 6/12 etc.).
New vessels at the optic disc: New vessels at the optic disc; neovascularization on or within 1 disc diameter of the optic disc.
New vessels elsewhere in the retina: New vessels elsewhere in the retina; neovascularization elsewhere in the retina and greater than 1 disc diameter from the optic disc margin.
New vessels on the iris: New vessels on the iris; neovascularization of the iris.
Nonproliferative diabetic retinopathy: The phases of diabetic retinopathy with no evidence of retinal neovascularization.
Optical coherence tomography: A diagnostic test using low energy lasers that takes a cross-section image of the retina, Used mostly to determine if there are membranes on the surface of the macula or fluid within or beneath it.
Panretinal photocoagulation: A type of laser surgery used for patients with PDR. The surgery is delivered in a scatter pattern throughout the peripheral fundus and is intended to lead to a regression of neovascularization.
Proliferative diabetic retinopathy: Advanced disease characterized by NVD and/or new vessels elsewhere in the retina.
Retinal hard exudate: Protein and lipid accumulation within the retina.
Refractory DME: in which the patient received 3 monthly doses of antiVEGF with poor anatomical and functional response.
Scatter photocoagulation: See Panretinal photocoagulation.
Severe nonproliferative diabetic retinopathy: Using the 4-2-1 rule, the presence of at least one of the following features: (1) severe intraretinal hemorrhages and microaneurysms, in 4 quadrants; (2) venous beading in 2 or more quadrants; or (3) moderate IRMA in 1 or more quadrants.
Severe visual loss: Occurrence of visual acuity worse than 5/200 at any two consecutive visits scheduled at 4-month intervals.
- Purpose
Identifying the stages of diabetic retinopathy and maculopathy
Initiate an appropriate management plan for all stages of diabetic retinopathy and maculopathy
- Scope
Methodology for treating diabetic retinopathy and maculopathy
- Target audience
Ophthalmologists - Pediatricians – Family doctors - High nurses.
- Methods
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
The inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author not on behalf of an organization in order to be valid and comprehensive, a guideline ideally requires multidisciplinary input
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in a table:
• Developing organization/authors
• Date of publication, posting, and release
• Country/language of publication
• Date of posting and/or release
• Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided a cut-off point or rank the guidelines (any guideline scoring above 50% on the rigour dimension was retained). This guideline was adapted from the Indian, Iranian guidelines and preferred practice in the American ophthalmological society as well as the royal college of ophthalmology [15-18].- Evidence assessment
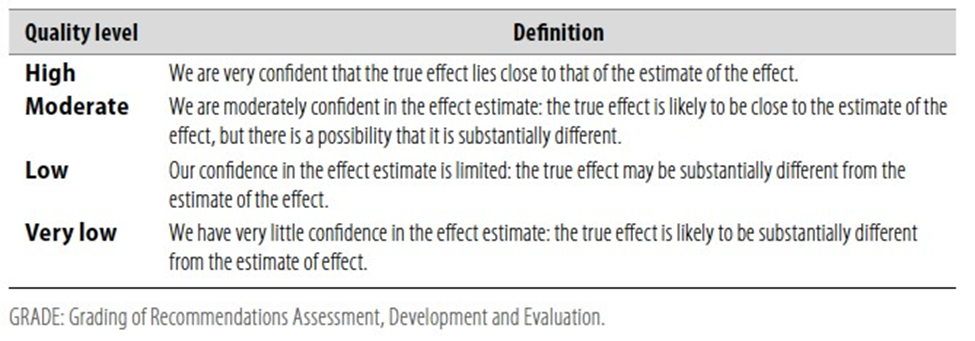
According to WHO handbook for Guidelines (5) we used the GRADE (Grading of
Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO
because they represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available on the following sites:
■ GRADE working group: http://www.gradeworkinggroup.org
■ GRADE online training modules:http://cebgrade.mcmaster.ca/
Table 1 Quality of evidence in GRADE
WHO guideline reporting tool - V1.5, December 2021
Table 2 Significance of the four levels of evidence
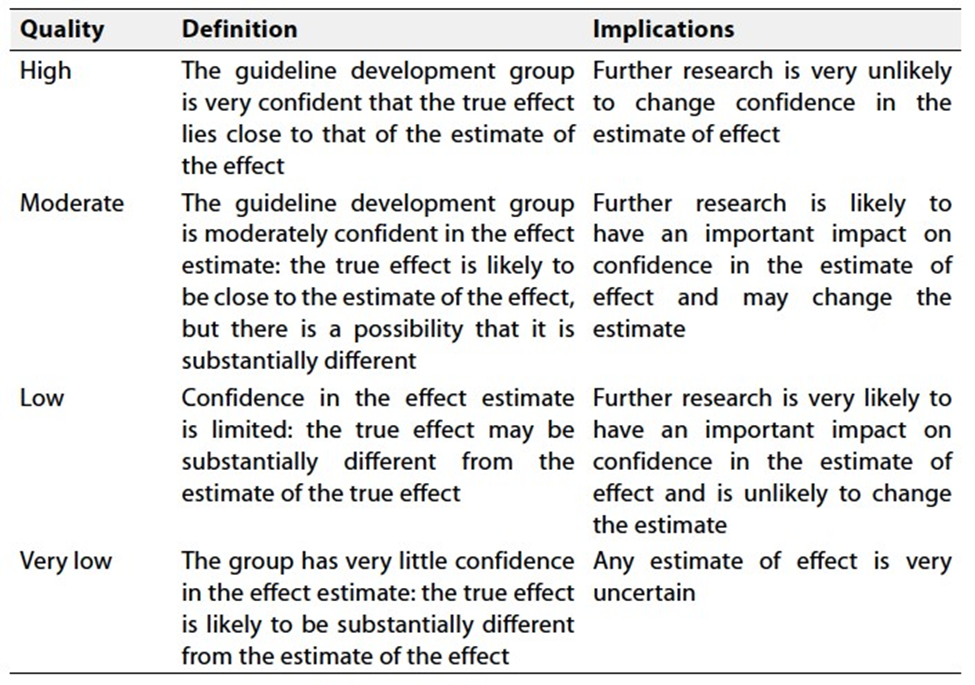
Table 3 Factors that determine how to upgrade or downgrade the quality of evidence

WHO guideline reporting tool - V1.5, December 2021
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to this recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
These are made when there is greater uncertainty about the four factors mentioned in table 3 or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Research Gaps
It is recommended to have high quality randomised controlled studies from combining different types of antiVEGF and concomitant use of laser.
- Clinical indications for monitoring
1. History of vascular occlusions, and associated conditions.
2. Blood sugar level.
3. Corrected visual acuity in all visits.
4. Detailed fundus biomicroscopy.
5. Prior to treatment, fundus fluorescein angiography or OCTA and OCT macula.
6. OCT macula after one month at the end of initial loading doses.
7. According to retreatment protocol OCT in each visit has to be presented with measurement of visual acuity.
- Appendix 1
Major study results
The Diabetic Retinopathy Study (DRS) was designed to investigate the value of laser photocoagulation surgery for patients with severe nonproliferative diabetic retinopathy (NPDR) and proliferative diabetic retinopathy (PDR). The results are shown in Table A4-1.
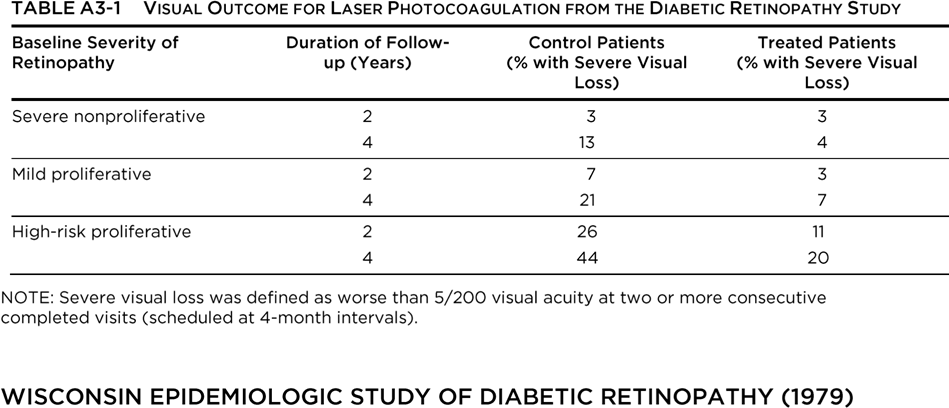
The Wisconsin Epidemiologic Study of Diabetic Retinopathy (WESDR) began in 1979. It was initially funded by the National Eye Institute, which is part of the National Institutes of Health. The purpose of the WESDR is to describe the frequency and incidence of complications associated with diabetes (eye complications such as diabetic retinopathy and visual loss, kidney complications such as diabetic nephropathy, and amputations), and to identify risk factors (such as poor glycemic control, smoking, and high blood pressure) that may contribute to the development of these complications.
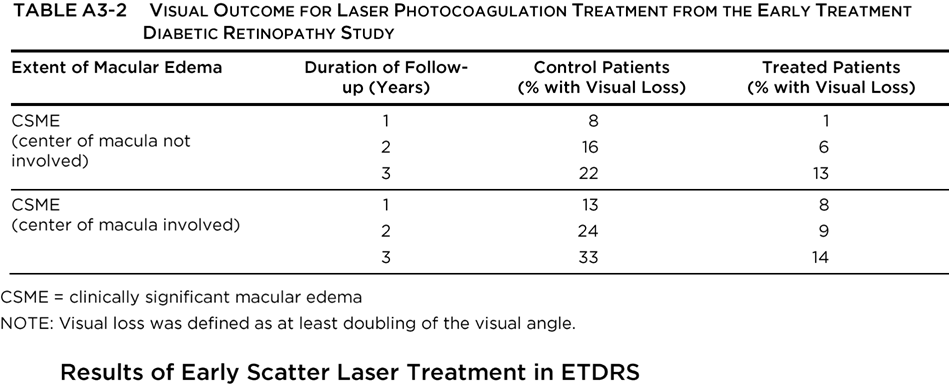
The Early Treatment Diabetic Retinopathy Study (ETDRS) investigated the value of photocoagulation surgery for patients with NPDR or PDR without high-risk characteristics. The results for eyes with macular edema are shown in Table A4-2. Visual loss was defined as at least doubling of the visual angle (e.g., 20/20 to 20/40, or 20/50 to 20/100). In eyes with NPDR or non-high-risk PDR, early panretinal photocoagulation (PRP) was compared with deferral of photocoagulation, and although there was a beneficial treatment effect, the outlook for maintaining vision was good in both groups. The 5-year rates of severe visual loss or vitrectomy ranged from 2% to 6% in eyes assigned to early photocoagulation and from 4% to 10% in eyes assigned to deferral. Early PRP was associated with side effects (small decreases in visual acuity and visual field) in some eyes, and the ETDRS concluded that deferral of photocoagulation was preferable at least until retinopathy was approaching the highrisk stage. Eyes approaching that stage had a 50% risk of reaching it within 12 to 18 months. Eyes in this category had very severe NPDR or non-high-risk PDR characterized by NVD less than one-quarter to one-third disc area and/or NVE, without vitreous or preretinal hemorrhage.
Recent additional analyses of visual outcome in ETDRS patients with severe NPDR to nonhigh-risk PDR suggest that the recommendation to consider PRP before the development of high-risk PDR is particularly appropriate for patients with type 2 diabetes. The risk of severe vision loss or vitrectomy was reduced by 50% in patients who were treated early compared with those who deferred treatment until high-risk PDR developed.
For patients with type 1 diabetes, the timing of the PRP will depend on the compliance with follow-up, status and response to treatment of the fellow eye, impending cataract surgery, and/or pregnancy status.
The Diabetic Retinopathy Vitrectomy Study (DRVS) investigated the role of vitrectomy in managing eyes with very severe PDR. The benefit of early vitrectomy for severe vitreous hemorrhage (defined as hemorrhage obscuring the macula or major retinal vessels for 3 disc diameters from the macular center) was seen in type 1 patients, but no such advantage was found in type 2 patients, who did not benefit from earlier surgery. Early vitrectomy was beneficial among patients with visual acuity of 5/200 or worse and severe vitreous hemorrhage with reduced vision for at least 1 month and without previous treatment or complications such as retinal detachment or neovascularization of the iris. Overall, at 2 years after surgery, 25% of the early vitrectomy group and 15% of the deferral group had visual acuity of 20/40 or better. The advantage was most pronounced in patients with type 1 diabetes (36% vs. 12% for early vitrectomy versus deferral of vitrectomy, respectively) and was not statistically significant for patients with type 2 diabetes.
The DRVS showed that early vitrectomy was beneficial for patients with visual acuity of 20/400 or better plus 1 of the following: (1) severe neovascularization and fibrous proliferation; (2) fibrous proliferation and moderate vitreous hemorrhage; or (3) moderate neovascularization, severe fibrous proliferation, and moderate vitreous hemorrhage. Among such patients, 44% with early vitrectomy and 28% in the observation group had visual acuity of 20/40 or better at 4 years of follow-up.
The results of the DRVS should be interpreted in light of subsequent advances in vitreoretinal surgery, such as the introduction of small-gauge vitrectomy technology, endoscopic and indirect ophthalmoscopic laser photocoagulation surgery, and advanced instrumentation. The use of longacting intraocular gases such as sulfur hexafluoride (SF6) and perfluoropropane (C3F8), the use of viscodissection, and the use of heavier-than-water liquids such as perfluoro-octane are advances in vitreoretinal surgery that developed after the DRVS. Thus, the results may actually be better than those reported in the DRVS. Early vitrectomy should be considered for selected patients with type 2 diabetes, particularly those in whom severe vitreous hemorrhage prohibits laser therapy photocoagulation of active neovascularization.
The FIELD study was a randomized controlled trial that evaluated long-term fenofibrate therapy for the reduction of cardiovascular events in 9795 patients with type 2 diabetes mellitus. Fenofibrate did not significantly reduce the risk of the primary outcome of coronary events. It did reduce total cardiovascular events, mainly due to fewer nonfatal myocardial infarctions and revascularizations. The higher rate of starting statin therapy in patients allocated to receive placebo might have masked a moderately larger treatment benefit.
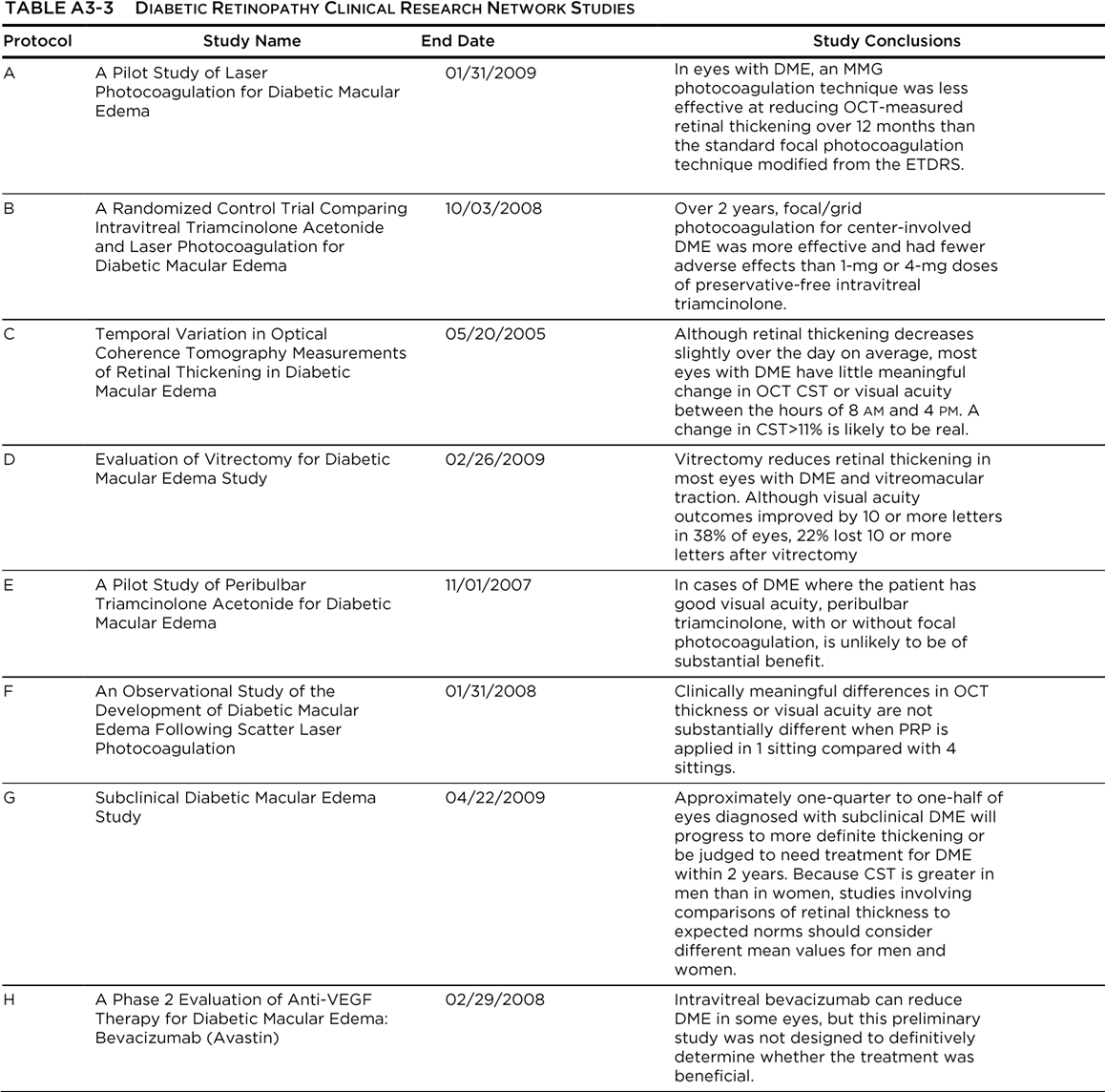
The Diabetic Retinopathy Clinical Research Network (DRCR.net) is a collaborative network dedicated to facilitating multicenter clinical research of diabetic retinopathy, diabetic macular edema
(DME), and associated conditions. The DRCR.net supports the identification, design, and implementation of multicenter clinical research initiatives focused on diabetes-induced retinal disorders. Principal emphasis is placed on clinical trials, but epidemiologic outcomes and other research may be supported as well. The DRCR.net was formed in 2002 and currently includes over 115 participating sites (offices) with over 400 physicians throughout the United States. The DRCR.net is funded by the National Eye Institute (NEI), which is a part of the National Institutes of Health, the branch of government that funds medical research. The DRCR.net has completed multiple clinical trials evaluating the role of anti-vascular endothelial growth factor (anti-VEGF), laser treatment, and corticosteroids in DME, anti-VEGF efficacy in PDR and vitreous hemorrhage, and even diabetes education effectiveness on DME (See Table A3-3). Most importantly, DRCR.net Protocol T (Comparative Effectiveness Study of Intravitreal Aflibercept, Bevacizumab, and Ranibizumab for Diabetic Macular Edema) compared the effectiveness of ranibizumab, aflibercept, and bevacizumab in the treatment of DME.96 This study found that all three drugs resulted in improvement in visual acuity at 1 year with similar safety profiles. However, the mean visual acuity using aflibercept was better for eyes with visual acuity of 20/50 or worse at 1 year. At 2 years, the mean visual acuity in the aflibercept was no longer superior to ranibizumab, although it remained superior to bevacizumab. Another important treatment comparison was done in Protocol I: Intravitreal Ranibizumab for Diabetic Macular Edema with Prompt vs. Deferred Laser Treatment. Three-year results were reported in 2012. The study utilized ranibizumab monthly until improvement no longer occurred (with resumption if the condition worsened) and random assignment to focal/grid laser treatment promptly or deferred (≥24 weeks). The 3-year results suggest that focal/grid laser treatment at the initiation of intravitreal ranibizumab is no better, and worse for vision outcomes, than deferring laser treatment for ≥24 weeks in eyes with DME involving the fovea and with vision impairment.94
A previous publication from Protocol I results confirmed the 1-year results that intravitreal ranibizumab with prompt or deferred laser was more effective through 2 years compared with prompt laser alone for the treatment of DME involving the central macula. Laser was not associated with endophthalmitis, the rare but potentially devastating complication of injecting ranibizumab. In pseudophakic eyes, results with intravitreal triamcinolone plus prompt laser appeared similar to results in the ranibizumab arms and were more effective than laser alone, but the triamcinolone plus prompt laser arm had an increased risk of IOP elevation.189
Most
recently, the DRCR.net Protocol S evaluated the effects of anti-VEGF versus
PRP.291 In a randomized, multicenter, noninferiority trial, 394 eyes
of 305 adults with PDR were randomized to receive either PRP or anti-VEGF therapy.
Ranibizumab 0.5 mg was given at baseline and as frequently as every 4 weeks
based on a structured retreatment design. Eyes in both groups were allowed
ranibizumab if DME was present. In eyes with PDR, ranibizumab was not inferior
to PRP in terms of visual acuity outcomes at 2 years. Mean visual acuity
improvement was +2.8 letters for ranibizumab and +0.2 letters for PRP-treated
eyes (P<0.001). When the totality
of the visual acuity data was included (area under the curve analysis), eyes
given ranibizumab had overall better visual acuity outcomes than eyes treated
with PRP. There was less mean reduction in peripheral visual field (-23 dB vs.
-422 dB; P<0.001) with ranibizumab
than with PRP treatment. The rates for vitrectomy were more frequent (15% vs. 4%;
P<0.001), and DME development was
more frequent (28% vs. 9%; P<0.001)
in the PRP group than in the ranibizumab group. Moreover, rates of active
neovascularization or rates of regression of neovascularization were similar
between the two groups.


The RISE and RIDE trials were parallel phase III multicenter double-masked sham injection– controlled randomized studies conducted at private and university-based retina specialty clinics in the
United States and South America. (See Glossary.)
The phase III results for both studies were published in 2012. The studies utilized monthly intravitreal ranibizumab (0.5 or 0.3 mg) or sham injections, with macular laser available if needed. The study concluded that ranibizumab rapidly and sustainably improved vision, reduced the risk of further vision loss, and improved macular edema in patients with DME, with low rates of ocular and nonocular side effects.
READ-2 was a phase II multicenter randomized controlled trial that compared 0.5 mg injections of ranibizumab versus focal laser treatment over 2 years in patients with type 1 or type 2 diabetes mellitus and DME. Patients randomized to one arm of the trial received ranibizumab at baseline, and at 1, 3 and 5 months after baseline; a second arm received laser treatment at baseline and at 3 months (if needed); the third arm received both ranibizumab and laser treatment at baseline and 3 months. From month 5, all subjects received ranibizumab every 2 months and/or maintenance laser treatment every 3 months.
At 24 months, differences between the groups were not statistically significant, and all groups experienced improved visual acuity. Patients receiving combined ranibizumab and laser treatment required fewer injections than patients receiving ranibizumab alone.
BOLT was a phase II 2-year randomized controlled trial that compared intravitreal 1.25 mg bevacizumab injections and focal laser treatment in patients with persistent DME and visual impairment. Bevacizumab patients received an injection every 6 weeks, whereas laser patients were treated every 4 weeks.
At 2 years, visual acuity results were substantially better in the bevacizumab group compared with the laser group, with significant differences in the proportions of patients gaining 10 letters and 15 letters. No patients lost 10 or more letters in the bevacizumab group, compared with 14% of patients treated
with laser.
These studies compared the efficacy and safety of intravitreal aflibercept injection (IAI) with macular laser photocoagulation surgery for DME. Visual improvement were observed in the IAI treatment regimens over laser control at 52, 100 and 148 weeks. Incidence of adverse events was consistent with the known safety profile of IAI.
The DRCR.net compared the efficacy and safety of bevacizumab, ranibizumab, and aflibercept in a multicentered, randomized clinical trial. At the primary endpoint at 1 year, the mean change in vision was greater for aflibercept than for either of the other two drugs. However, the mean visual acuity changes were dependent on the baseline visual acuity. For eyes with milder visual acuity loss, the drugs resulted in similar visual outcomes (8.0 with aflibercept, 7.5 with bevacizumab, and 8.3 with ranibizumab; P>0.50 for each pairwise comparison). However, for eyes with 20/50 or worse vision, the mean visual acuity in eyes treated with aflibercept had greater improvements in vision (18.9 with aflibercept, 11.8 with bevacizumab, and 14.2 with ranibizumab; P<0.001 for aflibercept vs. bevacizumab, P=0.003 for aflibercept vs. ranibizumab, and P=0.21 for ranibizumab vs.
bevacizumab). There were no significant differences in rates of adverse events. However, at 2 years, the mean visual acuity results were similar for ranibizumab and aflibercept, although aflibercept results remained significantly better than bevacizumab results. There was a slightly higher rate of Antiplatelet Trialists' Collaboration (APTC) events with ranibizumab compared with the other two drugs at the 2-year endpoint. All three drugs improved visual acuity at 2 years, and the number of injections decreased in year 2 compared to year 1.
- REFERENCES
1. Nanditha A, Ma RC, Ramachandran A, Snehalatha C, Chan JC, Chia KS, et al. Diabetes in Asia and the Pacific: Implications for the global epidemic. Diabetes Care 2016;39:472‑485.
2. Shaw JE, Sicree RA, Zimmet PZ. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract 2010;87:4‑14.
3. Prevention of blindness from diabetes mellitus: report of a WHO consultation in Geneva,
Switzerland, 9-11 November 2005. Available from: http://www.who.int/blindness/Prevention%20 of%20Blindness%20from%20Diabetes%20Mellitus‑with‑coversmall.pdf. [Last accessed on 2015 Dec 27].
4. Guariguata L, Whiting DR, Hambleton I, Beagley J, Linnenkamp U, Shaw JE. Global estimates of diabetes prevalence for 2013 and projections for 2035. Diabetes Res Clin Pract 2014;103:137‑49.
5. International Diabetes Federation. IDF Diabetes Atlas. 6th ed., 2015. Available from: http://www.diabetesatlas.org/component/ attachments/?task=download & id=116. [Last accessed on 2016 Jan 25].
6. Katibeh M, Pakravan M, Yaseri M, Pakbin M, Soleimanizad R. Prevalence and causes of visual impairment and blindness in central Iran; The Yazd Eye Study. J Ophthalmic Vis Res 2015;10:279‑285.
7. Dehghan MH, Katibeh M, Ahmadieh H, Nourinia R, Yaseri M. Prevalence and risk factors for diabetic retinopathy in the 40 to 80 year-old population in Yazd, Iran: The Yazd Eye Study. J Diabetes 2015;7:139‑141.
8. Judah G, Vlaev I, Gunn L, King D, King D, Valabhji J, et al. Incentives in Diabetic Eye Assessment by Screening (IDEAS): Study protocol of a three-arm randomized controlled trial using financial incentives to increase screening uptake in London. BMC Ophthalmol 2016;16:28.
9. Klein BE. Overview of epidemiologic studies of diabetic retinopathy. Ophthalmic Epidemiol 2007;14:179‑183.
10. Resnikoff S, Pascolini D, Etya’ale D, Kocur I, Pararajasegaram R, Pokharel GP, et al. Global data on visual impairment in the year 2002. Bull World Health Organ 2004;82:844‑851.
11. Moss SE, Klein R, Klein BE. The 14‑year incidence of visual loss in a diabetic population.
Ophthalmology 1998;105:998‑1003.
12. Cheung N, Mitchell P, Wong TY. Diabetic retinopathy. Lancet 2010;376:124‑136.
13. Klein R, Klein BE, Moss SE, Davis MD, DeMets DL. The Wisconsin epidemiologic study of diabetic retinopathy. III. Prevalence and risk of diabetic retinopathy when age at diagnosis is 30 or more years. Arch Ophthalmol 1984;102:527‑532.
14. Hatef E, Fotouhi A, Hashemi H, Mohammad K, Jalali KH. Prevalence of retinal diseases and their pattern in Tehran: The Tehran eye study. Retina 2008;28:755‑762.
15. Gilbert C, Gordon I, Mukherjee CR, Govindhari V. Guidelines for the prevention and management of diabetic retinopathy and diabetic eye disease in India: A synopsis. Indian J Ophthalmol 2020;68:S63-6.
16. Indian Institute of Public Health, Hyderabad, 2019. Guidelines for the Prevention and Management of Diabetic Retinopathy and Diabetic Eye Disease in India. Hyderabad, India: Indian Institute of Public Health.
17. Rajavi Z, Safi S, Javadi MA, Azarmina M, Moradian S, Entezari M, et al. Diabetic retinopathy clinical practice guidelines: Customized for Iranian population. J Ophthalmic Vis Res 2016;11:394-414.
18. Flaxel CJ, Adelman RA, Bailey ST, Fawzi A, Lim JI, Vemulakonda A,Ying G, Diabetic retinopathy preferred practice pattern, DOI:https://doi.org/10.1016/j.ophtha.2019.09.025
Royal college of ophthalmologists, Diabetic retinopathy guidelines.
19. Ferris F. Early photocoagulation in patients with either type I or type II diabetes. Trans Am Ophthalmol Soc. 1996;94: 505-37.
20. Early Treatment Diabetic Retinopathy Study Research Group. Early photocoagulation for diabetic retinopathy. ETDRS report number 9. . Ophthalmology. 1991;98(5 Suppl): 766-85.
21. Early Treatment Diabetic Retinopathy Study research Group. Photocoagulation for diabetic macular edema. Study report number 1. Arch Ophthalmol. 1985;103(12): 1796-806.
22. Lövestam‑Adrian M, Agardh CD, Torffvit O, Agardh E. Type 1 diabetes patients with severe non‑proliferative retinopathy may benefit from panretinal photocoagulation. Acta Ophthalmol Scand 2003;81:221‑225.
23. Treatment techniques and clinical guidelines for photocoagulation of diabetic macular edema. Early Treatment Diabetic Retinopathy Study Report Number 2. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology 1987;94:761‑774.
24. Diabetic Retinopathy Study Research Group. Indications for photocoagulation treatment of diabetic retinopathy: Diabetic Retinopathy Study Report number 14. Int Ophthalmol Clin 1987;27:239‑253.
25. Zaluski S, Marcil G, Lamer L, Lambert J. Study of the visual field using automated static perimetry following panretinal photocoagulation in the diabetic. J Fr Ophtalmol 1986;9:395‑401.
26. Buckley SA, Jenkins L, Benjamin L. Fields, DVLC and panretinal photocoagulation. Eye (Lond) 1992;6(Pt 6):623‑625.
27. Pahor D. Visual field loss after argon laser panretinal photocoagulation in diabetic retinopathy: Full‑versus mild‑scatter coagulation. Int Ophthalmol 1998;22:313‑319.
28. Diabetic Retinopathy Clinical Research Network (DRCR.net), Beck RW, Edwards AR, Aiello LP, Bressler NM, Ferris F, et al. Three-year follow-up of a randomized trial comparing focal/ grid photocoagulation and intravitreal triamcinolone for diabetic macular edema. Arch Ophthalmol 2009;127:245‑251.
29. Gillies MC, Simpson JM, Gaston C, Hunt G, Ali H, Zhu M, et al. Five-year results of a randomized trial with open-label extension of triamcinolone acetonide for refractory diabetic macular edema. Ophthalmology 2009;116:2182‑2187.
30. Lam DS, Chan CK, Mohamed S, Lai TY, Lee VY, Liu DT, et al. Intravitreal triamcinolone plus sequential grid laser versus triamcinolone or laser alone for treating diabetic macular edema: Six‑month outcomes. Ophthalmology 2007;114:2162‑2167.
31. Gillies MC, McAllister IL, Zhu M, Wong W, Louis D, Arnold JJ, et al. Pretreatment with intravitreal triamcinolone before laser for diabetic macular edema: 6-month results of a randomized, placebo‑controlled trial. Invest Ophthalmol Vis Sci 2010;51:2322‑2328.
32. Diabetic Retinopathy Clinical Research Network, Chew E, Strauber S, Beck R, Aiello LP, Antoszyk A, et al. Randomized trial of peribulbar triamcinolone acetonide with and without focal photocoagulation for mild diabetic macular edema: A pilot study. Ophthalmology 2007;114:1190‑1196.
33. Gillies MC, Simpson JM, Billson FA, Luo W, Penfold P, Chua W, et al. Safety of an intravitreal injection of triamcinolone: Results from a randomized clinical trial. Arch Ophthalmol 2004;122:336‑340.
34. Quiram PA, Gonzales CR, Schwartz SD. Severe steroid-induced glaucoma following intravitreal injection of triamcinolone acetonide. Am J Ophthalmol 2006;141:580‑582.
35. Haller JA, Kuppermann BD, Blumenkranz MS, Williams GA, Weinberg DV, Chou C, et al. Randomized controlled trial of an intravitreous dexamethasone drug delivery system in patients with diabetic macular edema. Arch Ophthalmol 2010;128:289‑296.
36. Campochiaro PA, Hafiz G, Shah SM, Bloom S, Brown DM, Busquets M, et al. Sustained ocular delivery of fluocinolone acetonide by an intravitreal insert. Ophthalmology
2010;117:1393‑1399.e3.
37. Campochiaro PA, Brown DM, Pearson A, Chen S, Boyer D, Ruiz-Moreno J, et al. Sustained delivery fluocinolone acetonide vitreous inserts provide benefit for at least 3 years in patients with diabetic macular edema. Ophthalmology 2012;119:2125‑2132.
38. Adamis AP, Miller JW, Bernal MT, D’Amico DJ, Folkman J, Yeo TK, et al. Increased vascular endothelial growth factor levels in the vitreous of eyes with proliferative diabetic retinopathy. Am J Ophthalmol 1994 15;118:445‑450.
39. Cunningham ET Jr., Adamis AP, Altaweel M, Aiello LP, Bressler NM, D’Amico DJ, et al. A phase II randomized double‑masked trial of pegaptanib, an anti‑vascular endothelial growth factor aptamer, for diabetic macular edema. Ophthalmology 2005;112:1747‑1757.
40. Nguyen QD, Shah SM, Heier JS, Do DV, Lim J, Boyer D, et al. Primary end point (Six Months) results of the ranibizumab for edema of the mAcula in diabetes (READ‑2) study. Ophthalmology 2009;116:2175‑2181.e1.
41. Nguyen QD, Shah SM, Khwaja AA, Channa R, Hatef E, Do DV, et al. Two-year outcomes of the ranibizumab for edema of the mAcula in diabetes (READ‑2) study. Ophthalmology 2010;117:2146‑2151.
42. Massin P, Bandello F, Garweg JG, Hansen LL, Harding SP, Larsen M, et al. Safety and efficacy of ranibizumab in diabetic macular edema (RESOLVE Study): A 12-month, randomized, controlled, double‑masked, multicenter phase II study. Diabetes Care 2010;33:2399‑2405.
43. Mitchell P, Bandello F, Schmidt-Erfurth U, Lang GE, Massin P, Schlingemann RO, et al. The RESTORE study: Ranibizumab monotherapy or combined with laser versus laser monotherapy for diabetic macular edema. Ophthalmology 2011;118:615‑625.
44. Diabetic Retinopathy Clinical Research Network, Elman MJ, Aiello LP, Beck RW, Bressler NM, Bressler SB, et al. Randomized trial evaluating ranibizumab plus prompt or deferred laser or triamcinolone plus prompt laser for diabetic macular edema. Ophthalmology 2010;117:1064‑1077.e35.
45. Goyal S, Lavalley M, Subramanian ML. Meta‑analysis and review on the effect of bevacizumab in diabetic macular edema. Graefes Arch Clin Exp Ophthalmol 2011;249:15‑27.
46. Yilmaz T, Cordero-Coma M, Gallagher MJ, Teasley LA. Systematic review of intravitreal bevacizumab injection for treatment of primary diabetic macular oedema. Acta Ophthalmol 2011;89:709‑717.
47. Michaelides M, Kaines A, Hamilton RD, Fraser‑Bell S, Rajendram R, Quhill F, et al. A prospective randomized trial of intravitreal bevacizumab or laser therapy in the management of diabetic macular edema (BOLT study) 12‑month data: Report 2. Ophthalmology 2010;117:1078‑1086.e2.
48. Arevalo JF, Sanchez JG, Wu L, Maia M, Alezzandrini AA, Brito M, et al. Primary intravitreal bevacizumab for diffuse diabetic macular edema: The Pan‑American Collaborative Retina Study Group at 24 months. Ophthalmology 2009;116:1488‑1497, 1497.e1.
49. Lam DS, Lai TY, Lee VY, Chan CK, Liu DT, Mohamed S, et al. Efficacy of 1.25 MG versus 2.5 MG intravitreal bevacizumab for diabetic macular edema: Six-month results of a randomized controlled trial. Retina 2009;29:292‑299.
50. Ahmadieh H, Ramezani A, Shoeibi N, Bijanzadeh B, Tabatabaei A, Azarmina M, et al. Intravitreal bevacizumab with or without triamcinolone for refractory diabetic macular edema; a placebo-controlled, randomized clinical trial. Graefes Arch Clin Exp Ophthalmol 2008;246:483‑489.
51. Soheilian M, Ramezani A, Obudi A, Bijanzadeh B, Salehipour M, Yaseri M, et al. Randomized trial of intravitreal bevacizumab alone or combined with triamcinolone versus macular photocoagulation in diabetic macular edema. Ophthalmology 2009;116:1142‑1150.
52. Solaiman KA, Diab MM, Abo-Elenin M. Intravitreal bevacizumab and/or macular photocoagulation as a primary treatment for diffuse diabetic macular edema. Retina 2010;30:1638‑1645.
53. Stewart MW, Rosenfeld PJ. Predicted biological activity of intravitreal VEGF Trap. Br J Ophthalmol 2008;92:667‑668.
54. Do DV, Nguyen QD, Shah SM, Browning DJ, Haller JA, Chu K, et al. An exploratory study of the safety, tolerability and bioactivity of a single intravitreal injection of vascular endothelial growth factor Trap‑Eye in patients with diabetic macular oedema. Br J Ophthalmol 2009;93:144‑149.
55. Do DV, Nguyen QD, Boyer D, Schmidt-Erfurth U, Brown DM, Vitti R, et al. One‑year outcomes of the da Vinci Study of VEGF Trap‑Eye in eyes with diabetic macular edema. Ophthalmology 2012;119:1658‑1665.
56. Lanzagorta-Aresti A, Palacios-Pozo E, Menezo Rozalen JL, Navea‑Tejerina A. Prevention of vision loss after cataract surgery in diabetic macular edema with intravitreal bevacizumab: A pilot study. Retina 2009;29:530‑555.
57. Akinci A, Muftuoglu O, Altinsoy A, Ozkilic E. Phacoemulsification with intravitreal bevacizumab and triamcinolone acetonide injection in diabetic patients with clinically significant macular edema and cataract. Retina 2011;31:755‑758.
58. Fard MA, Yazdanei Abyane A, Malihi M. Prophylactic intravitreal bevacizumab for diabetic macular edema (thickening) after cataract surgery: Prospective randomized study. Eur J Ophthalmol 2011;21:276‑281.
59. Ishibashi T, Miki K, Sorgente N, Patterson R, Ryan SJ. Effects of intravitreal administration of steroids on experimental subretinal neovascularization in the subhuman primate. Arch Ophthalmol 1985;103:708‑711.
60. Danis RP, Bingaman DP, Yang Y, Ladd B. Inhibition of preretinal and optic nerve head neovascularization in pigs by intravitreal triamcinolone acetonide. Ophthalmology 1996;103:2099‑2104.
61. Ciulla TA, Criswell MH, Danis RP, Hill TE. Intravitreal triamcinolone acetonide inhibits choroidal neovascularization in a laser‑treated rat model. Arch Ophthalmol 2001;119:399‑404.
62. Wang YS, Friedrichs U, Eichler W, Hoffmann S, Wiedemann P. Inhibitory effects of triamcinolone acetonide on bFGF‑induced migration and tube formation in choroidal microvascular endothelial cells. Graefes Arch Clin Exp Ophthalmol 2002;240:42‑48.
63. Jonas JB, Hayler JK, Söfker A, Panda‑Jonas S. Intravitreal injection of crystalline cortisone as adjunctive treatment of proliferative diabetic retinopathy. Am J Ophthalmol 2001;131:468‑471.
64. Nauck M, Karakiulakis G, Perruchoud AP, Papakonstantinou E, Roth M. Corticosteroids inhibit the expression of the vascular endothelial growth factor gene in human vascular smooth muscle cells. Eur J Pharmacol 1998;341:309‑315.
65. Brooks HL Jr., Caballero S Jr., Newell CK, Steinmetz RL, Watson D, Segal MS, et al. Vitreous levels of vascular endothelial growth factor and stromal‑derived factor 1 in patients with diabetic retinopathy and cystoid macular edema before and after intraocular injection of triamcinolone. Arch Ophthalmol 2004;122:1801‑1817.
66. Jonas JB, Kreissig I, Söfker A, Degenring RF. Intravitreal injection of triamcinolone for diffuse diabetic macular edema. Arch Ophthalmol 2003;121:57‑61.
67. Jonas JB, Söfker A. Intraocular injection of crystalline cortisone as adjunctive treatment of diabetic macular edema. Am J Ophthalmol 2001;132:425‑427.
68. Jonas JB. Intravitreal triamcinolone acetonide for treatment of intraocular oedematous and neovascular diseases. Acta Ophthalmol Scand 2005;83:645‑663.
69. Jonas JB, Degenring RF, Kamppeter BA. Outcome of eyes undergoing trabeculectomy after intravitreal injections of triamcinolone acetonide. J Glaucoma 2004;13:261.
70. Kuhn F, Barker D. Intravitreal injection of triamcinolone acetonide for diabetic macular edema. Arch Ophthalmol 2004;122:1082‑1083.
71. Savage H, Roh M. Safety and efficacy of intravitreal triamcinolone. Arch Ophthalmol 2004;122:1083.
72. Patelli F, Fasolino G, Radice P, Russo S, Zumbo G, DI Tizio FM, et al. Time course of changes in retinal thickness and visual acuity after intravitreal triamcinolone acetonide for diffuse diabetic macular edema with and without previous macular laser treatment. Retina 2005;25:840‑845.
73. Larsson J, Zhu M, Sutter F, Gillies MC. Relation between reduction of foveal thickness and visual acuity in diabetic macular edema treated with intravitreal triamcinolone. Am J Ophthalmol 2005;139:802‑806.
74. Massin P, Audren F, Haouchine B, Erginay A, Bergmann JF, Benosman R, et al. Intravitreal triamcinolone acetonide for diabetic diffuse macular edema: Preliminary results of a prospective controlled trial. Ophthalmology 2004;111:218‑224.
75. Avitabile T, Longo A, Reibaldi A. Intravitreal triamcinolone compared with macular laser grid photocoagulation for the treatment of cystoid macular edema. Am J Ophthalmol 2005;140:695‑702.
76. Sutter FK, Simpson JM, Gillies MC. Intravitreal triamcinolone for diabetic macular edema that persists after laser treatment: Three‑month efficacy and safety results of a prospective, randomized, double-masked, placebo-controlled clinical trial. Ophthalmology 2004;111:2044‑2049.
77. Audren F, Lecleire‑Collet A, Erginay A, Haouchine B, Benosman R, Bergmann JF, et al. Intravitreal triamcinolone acetonide for diffuse diabetic macular edema: Phase 2 trial comparing 4 mg vs 2 mg. Am J Ophthalmol 2006;142:794‑799.
78. Jonas JB, Kamppeter BA, Harder B, Vossmerbaeumer U, Sauder G, Spandau UH. Intravitreal triamcinolone acetonide for diabetic macular edema: A prospective, randomized study. J Ocul Pharmacol Ther 2006;22:200‑207.
79. Audren F, Erginay A, Haouchine B, Benosman R, Conrath J, Bergmann JF, et al. Intravitreal triamcinolone acetonide for diffuse diabetic macular oedema: 6‑month results of a prospective controlled trial. Acta Ophthalmol Scand 2006;84:624‑630.
80. Lam DS, Chan CK, Mohamed S, Lai TY, Li KK, Li PS, et al. A prospective randomised trial of different doses of intravitreal triamcinolone for diabetic macular oedema. Br J Ophthalmol 2007;91:199‑203.
81. Kang SW, Sa HS, Cho HY, Kim JI. Macular grid photocoagulation after intravitreal triamcinolone acetonide for diffuse diabetic macular edema. Arch Ophthalmol 2006;124:653‑658.
82. Parke DW. Intravitreal triamcinolone and endophthalmitis. Am J Ophthalmol 2003;136:918‑919.
83. Roth DB, Chieh J, Spirn MJ, Green SN, Yarian DL, Chaudhry NA. Noninfectious endophthalmitis associated with intravitreal triamcinolone injection. Arch Ophthalmol 2003;121:1279‑1282.
84. Jonas JB, Kreissig I, Spandau UH, Harder B. Infectious and noninfectious endophthalmitis after intravitreal high‑dosage triamcinolone acetonide. Am J Ophthalmol 2006;141:579‑580.
85. Jonas JB, Degenring RF, Kreissig I, Akkoyun I, Kamppeter BA. Intraocular pressure elevation after intravitreal triamcinolone acetonide injection. Ophthalmology 2005;112:593‑598.
86. Konstantopoulos A, Williams CP, Newsom RS, Luff AJ. Ocular morbidity associated with intravitreal triamcinolone acetonide. Eye (Lond) 2007;21:317‑320.
87. Lang Y, Leibu R, Shoham N, Miller B, Perlman I. Evaluation of intravitreal kenalog toxicity in humans. Ophthalmology 2007;114:724‑731.
88. Bandello F, Polito A, Pognuz DR, Monaco P, Dimastrogiovanni A, Paissios J. Triamcinolone as adjunctive treatment to laser panretinal photocoagulation for proliferative diabetic retinopathy. Arch Ophthalmol 2006;124:643‑650.
89. Spandau UH, Derse M, Schmitz-Valckenberg P, Papoulis C, Jonas JB. Dosage dependency of intravitreal triamcinolone acetonide as treatment for diabetic macular oedema. Br J Ophthalmol 2005;89:999‑1003.
90. Jonas JB, Degenring RF, Kamppeter BA, Kreissig I, Akkoyun I. Duration of the effect of intravitreal triamcinolone acetonide as treatment for diffuse diabetic macular edema. Am J Ophthalmol 2004;138:158‑160.
91. Rodriguez-Coleman H, Yuan P, Kim H, Gravlin L, Srivastava S, Csaky KG, et al. Intravitreal injection of triamcinolone for diffuse macular edema. Arch Ophthalmol 2004;122:1085‑1116.
92. Negi AK, Vernon SA, Lim CS, Owen‑Armstrong K. Intravitreal triamcinolone improves vision in eyes with chronic diabetic macular oedema refractory to laser photocoagulation. Eye (Lond) 2005;19:747‑751.
93. Gibran SK, Cullinane A, Jungkim S, Cleary PE. Intravitreal triamcinolone for diffuse diabetic macular oedema. Eye 2006;20:720‑724.
94. Jonas JB, Spandau UH, Kamppeter BA, Vossmerbaeumer U, Harder B, Sauder G. Repeated intravitreal high‑dosage injections of triamcinolone acetonide for diffuse diabetic macular edema. Ophthalmology 2006;113:800‑804.
95. Kim H, Csaky KG, Gravlin L, Yuan P, Lutz RJ, Bungay PM, et al. Safety and pharmacokinetics of a preservative‑free triamcinolone acetonide formulation for intravitreal administration. Retina 2006;26:523‑530.
96. Pearson P, Levy B, Comstock T, Fluocinolone Acetonide Implant Study Group. Fluocinolone acetonide intravitreal implant to treat diabetic macular edema: 3‑year results of a multi‑centre clinical trial. Invest Ophthalmol Vis Sci 2006;47 5442 [E‑abstract 5442].
97. Kuppermann BD, Blumenkranz MS, Haller JA, Williams GA, Weinberg DV, Chou C, et al. Randomized controlled study of an intravitreous dexamethasone drug delivery system in patients with persistent macular edema. Arch Ophthalmol 2007;125:309‑317.
98. Verma LK, Vivek MB, Kumar A, Tewari HK, Venkatesh P. A prospective controlled trial to evaluate the adjunctive role of posterior subtenon triamcinolone in the treatment of diffuse diabetic macular edema. J Ocul Pharmacol Ther 2004;20:277‑284.
99. Bakri SJ, Kaiser PK. Posterior subtenon triamcinolone acetonide for refractory diabetic macular edema. Am J Ophthalmol 2005;139:290‑294.
100. Cardillo JA, Melo LA Jr., Costa RA, Skaf M, Belfort R Jr., Souza-Filho AA, et al. Comparison of intravitreal versus posterior sub‑Tenon’s capsule injection of triamcinolone acetonide for diffuse diabetic macular edema. Ophthalmology 2005;112:1557‑1563.
101. Bonini‑Filho MA, Jorge R, Barbosa JC, Calucci D, Cardillo JA, Costa RA. Intravitreal injection versus sub‑Tenon’s infusion of triamcinolone acetonide for refractory diabetic macular edema: A randomized clinical trial. Invest Ophthalmol Vis Sci 2005;46:3845‑3849.
102. Caldwell RB, Bartoli M, Behzadian MA, El-Remessy AE, Al‑Shabrawey M, Platt DH, et al. Vascular endothelial growth factor and diabetic retinopathy: Role of oxidative stress. Curr Drug Targets 2005;6:511‑524.
103. Zhang SX, Ma JX. Ocular neovascularization: Implication of endogenous angiogenic inhibitors and potential therapy. Prog Retin Eye Res 2007;26:1‑37.
104. Ng EW, Shima DT, Calias P, Cunningham ET Jr., Guyer DR, Adamis AP. Pegaptanib, a targeted anti‑VEGF aptamer for ocular vascular disease. Nat Rev Drug Discov 2006;5:123‑132.
105. Gragoudas ES, Adamis AP, Cunningham ET Jr, Feinsod M, Guyer DR; VEGF Inhibition Study in Ocular Neovascularization Clinical Trial Group. Pegaptanib for neovascular age‑related macular degeneration. N Engl J Med 2004;351:2805‑2816.
106. Adamis AP, Altaweel M, Bressler NM, Cunningham ET Jr., Davis MD, Goldbaum M, et al. Changes in retinal neovascularization after pegaptanib (Macugen) therapy in diabetic individuals. Ophthalmology 2006;113:23‑28.
107. Churchill AJ, Carter JG, Lovell HC, Ramsden C, Turner SJ, Yeung A, et al. VEGF polymorphisms are associated with neovascular age‑related macular degeneration. Hum Mol Genet 2006 1;15:2955‑2961.
108. Brown DM, Kaiser PK, Michels M, Soubrane G, Heier JS, Kim RY, et al. Ranibizumab versus verteporfin for neovascular age-related macular degeneration. N Engl J Med 2006;355:1432‑1444.
109. Chun DW, Heier JS, Topping TM, Duker JS, Bankert JM. A pilot study of multiple intravitreal injections of ranibizumab in patients with center-involving clinically significant diabetic macular edema. Ophthalmology 2006;113:1706‑1712.
110. Spaide RF, Fisher YL. Intravitreal bevacizumab (Avastin) treatment of proliferative diabetic retinopathy complicated by vitreous hemorrhage. Retina 2006;26:275‑278.
111. Avery RL, Pearlman J, Pieramici DJ, Rabena MD, Castellarin AA, Nasir MA, et al. Intravitreal bevacizumab (Avastin) in the treatment of proliferative diabetic retinopathy. Ophthalmology 2006;113:1695.e1‑15.
112. Mason JO 3rd, Nixon PA, White MF. Intravitreal injection of bevacizumab (Avastin) as adjunctive treatment of proliferative diabetic retinopathy. Am J Ophthalmol 2006;142:685‑688.
113. Oshima Y, Sakaguchi H, Gomi F, Tano Y. Regression of iris neovascularization after intravitreal injection of bevacizumab in patients with proliferative diabetic retinopathy. Am J Ophthalmol 2006;142:155‑158.
114. Grisanti S, Biester S, Peters S, Tatar O, Ziemssen F, Bartz-Schmidt KU; Tuebingen Bevacizumab Study Group. Intracameral bevacizumab for iris rubeosis. Am J Ophthalmol 2006;142:158‑160.
115. Avery RL. Regression of retinal and iris neovascularization after intravitreal bevacizumab (Avastin) treatment. Retina 2006;26:352‑354.
116. Haritoglou C, Kook D, Neubauer A, Wolf A, Priglinger S, Strauss R, et al. Intravitreal bevacizumab (Avastin) therapy for persistent diffuse diabetic macular edema. Retina 2006;26:999‑1005.
117. Arevalo JF, Fromow-Guerra J, Quiroz-Mercado H, Sanchez JG, Wu L, Maia M, et al. Primary intravitreal bevacizumab (Avastin) for diabetic macular edema: Results from the Pan‑American Collaborative Retina Study Group at 6‑month follow‑up. Ophthalmology 2007;114:743‑750.
118. Yanyali A, Aytug B, Horozoglu F, Nohutcu AF. Bevacizumab (Avastin) for diabetic macular edema in previously vitrectomized eyes. Am J Ophthalmol 2007;144:124‑126.
119. Isaacs TW, Barry C. Rapid resolution of severe disc new vessels in proliferative diabetic retinopathy following a single intravitreal injection of bevacizumab (Avastin). Clin Experiment Ophthalmol 2006;34:802‑803.
120. Bakri SJ, Donaldson MJ, Link TP. Rapid regression of disc neovascularization in a patient with proliferative diabetic retinopathy following adjunctive intravitreal bevacizumab. Eye (Lond) 2006;20:1474‑1475.
121. Diabetic Retinopathy Clinical Research Network, Scott IU, Edwards AR, Beck RW, Bressler NM, Chan CK, et al. A phase II randomized clinical trial of intravitreal bevacizumab for diabetic macular edema. Ophthalmology 2007;114:1860‑1867.
122. Zhao LQ, Zhu H, Zhao PQ, Hu YQ. A systematic review and meta‑analysis of clinical outcomes of vitrectomy with or without intravitreal bevacizumab pretreatment for severe diabetic retinopathy. Br J Ophthalmol 2011;95:1216‑1222.
123. Ahmadieh H, Shoeibi N, Entezari M, Monshizadeh R. Intravitreal bevacizumab for prevention of early postvitrectomy hemorrhage in diabetic patients: A randomized clinical trial. Ophthalmology 2009;116:1943‑1948.
124. Rizzo S, Genovesi-Ebert F, Di Bartolo E, Vento A, Miniaci S, Williams G. Injection of intravitreal bevacizumab (Avastin) as a preoperative adjunct before vitrectomy surgery in the treatment of severe proliferative diabetic retinopathy (PDR). Graefes Arch Clin Exp Ophthalmol 2008;246:837‑842.
125. Yeoh J, Williams C, Allen P, Buttery R, Chiu D, Clark B, et al. Avastin as an adjunct to vitrectomy in the management of severe proliferative diabetic retinopathy: A prospective case series. Clin Experiment Ophthalmol 2008;36:449‑454.
126. Oshima Y, Shima C, Wakabayashi T, Kusaka S, Shiraga F, Ohji M, et al. Microincision vitrectomy surgery and intravitreal bevacizumab as a surgical adjunct to treat diabetic traction retinal detachment. Ophthalmology 2009;116:927‑938.
127. Zhang ZH, Liu HY, Hernandez-Da Mota SE, Romano MR, Falavarjani KG, Ahmadieh H, et al. Vitrectomy with or without preoperative intravitreal bevacizumab for proliferative diabetic retinopathy: A meta-analysis of randomized controlled trials. Am J Ophthalmol 2013;156:106‑115.e2.