Most common viral diseases affecting shrimp in aquaculture systems
| Site: | EHC | Egyptian Health Council |
| Course: | Aquatic animal medicine Guidelines |
| Book: | Most common viral diseases affecting shrimp in aquaculture systems |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 8 Jun 2026" Download Guideline
- Acknowledgment
We would like to acknowledge the committee of National Egyptian Guidelines for Veterinary Medical Interventions, Egyptian Health Council for adapting this guideline.
Executive Chief of the Egyptian Health Council: Prof. Mohamed Mustafa Lotief.
Head of the Committee: Prof. Ahmed M Byomi
The rapporteur of the Committee: Prof. Mohamed Mohamedy Ghanem.
Scientific Group Members: Prof. Gamal A. Sosa., Prof. Nabil Yassien, Prof. Ashraf Aldesoky Shamaa, Prof. Amany Abbass, Prof. Dalia Mansour, Dr. Essam Elmarakby, Dr. Mohamed Elsharkawy, Dr. Naglaa Radwan, Dr. Hend El Sheikh
Authors: Mohamed Faisal1,2; Adel A. Shaheen1, Amany A. Abbass1; Amel M. El Asely1; Eman A. Abd El-Gawad1; Hiam S. Elabd1; Aya F. Matter1; Hadeer A. Youssef1, and Amira M. El-Daim1.
1Department of Aquatic Animal Medicine, Faculty of Veterinary Medicine, Benha University, Egypt.
2College of Veterinary Medicine, Michigan State University, USA.
- Aim
The principal objective is to provide concise data regarding main viral infections of shrimp in aquaculture systems. It aims to underscore the diversity of viruses, their epidemiology, pathophysiology, and diagnostic methodologies, while accentuating their economic, ecological, and social ramifications. The evaluation seeks to assess current prevention and control techniques, pinpoint knowledge deficiencies, and suggest future research and management paths to improve aquaculture sustainability.
- Scope
• Shrimp biology and farming requirements.
• Epidemiology and transmission mechanisms of viral infections in aquaculture systems.
• Clinical signs and pathological findings related to viral infections in cultured species.
• Diagnostic methodologies, including molecular tools, immunological assays, and innovative technology.
• Prevention and control strategies, including biosecurity protocols, selective breeding for disease resistance and management techniques.
- Introduction
Giving a steady supply of protein while also making substantial contributions to economic growth and food security, aquaculture has quickly become one of the world's most dynamic food production industries (Obirikorang et al., 2024). However, infectious diseases, especially those with a viral origin, have been on the rise alongside the fast expansion of aquaculture (Valero and Cuesta, 2023) and considered a significant threat to global shrimp production, causing substantial economic losses through reducing production, increasing costs and trade restriction. Acute outbreaks, large mortality rates, and substantial economic losses are sometimes caused by viral infections, which pose a significant threat to farmed shrimp and fish (Rahaman et al., 2025). These diseases limit the potential for growth, decrease the likelihood of survival, and impede the global expansion of aquaculture businesses (Maezono et al., 2025). Viruses including White Spot Syndrome Virus (WSSV), Yellow Head Virus (YHV), and Taura Syndrome Virus (TSV) have also had a devastating effect on shrimp farming (Arulmoorthy et al., 2020).
Virus diseases have far-reaching effects that go beyond financial burdens. They threaten biodiversity and environmental stability by allowing it easier for pathogens to spread to wild populations (Sudhagar et al., 2024). Moreover, disease outbreaks have significant societal consequences because they threaten farmers' incomes and local and global market stability (Hounmanou et al., 2018).
Prevention and control procedures are of the greatest significance considering the lack of effective therapeutic measures against viral diseases (Munang’andu et al., 2016). When it comes to disease control, biosecurity protocols, early diagnostic tools, and pathogen-free broodstock are pivotal. Additionally, innovative techniques to improve aquaculture systems' resilience and sustainability necessitate continued research and international cooperation (Aly and Fathi 2024).
- Shrimp biology and farming requirements
Shrimp production, a significant sector in global aquaculture, plays vital role in food security, employment and economic growth, especially in coastal regions. With increasing demand for seafood, shrimp farming has expanded rapidly making it one of the most traded aquatic products in the world. Shrimps are playing crucial roles in marine and freshwater ecosystems.
Biology: Shrimps belong to the sub-order Pleocyemata which includes most decapod crustaceans like crabs, lobsters, and shrimp. Shrimp body curved as their thorax overlaps their head and abdomen, allowing them to bend their bodies more than prawns, have two pairs of claw-like legs and plate-like gills; Shrimps will carry their fertilized eggs on the undersides of female's abdomen and females brooding eggs on their pleopods known as (berried) females. While, Prawns belong to the sub-order Dendrobranchiata; each segment overlaps the one below (head overlaps the thorax, and the thorax overlaps the abdomen) so their bodies are straight and unable to bend as shrimps do. Prawns also have three pairs of claw-like legs, and branching gills. The sexes are separate, and females tend to be larger than males. Prawns release their eggs into the water then leave them to grow on their own.
Reproduction biology: Sex is separate, reproduction is hormonally regulated, synthesis and release of hormones occur during season, Reproduction is controlled by hormones released from sinus gland and associated centers in eye stalk. The common technique used to induce reproduction is eyestalk ablation, usually unilateral by cutting off eye stalk to induce spermatogenesis and acceleration vitellogenesis in male and female respectively. In shrimp gonad maturation in female occur very rapidly within 3-4 days after ablation. At mating male insert spermatophores (sperm) into thelycum (a specialized external structure formed from modified sternal plates on the underside of a female shrimp’s thorax that receive and store sperm from male during mating) on the ventral surface of female shrimp. Fertilization occurs externally upon ovulation and passage of the oocyte through the gonadophore, the fertilized eggs are retained on female abdominal appendages (pleopods) until the larvae hatch, the newly hatched larvae each must undergo up to 12 molts to attain final form as a juvenile shrimp.
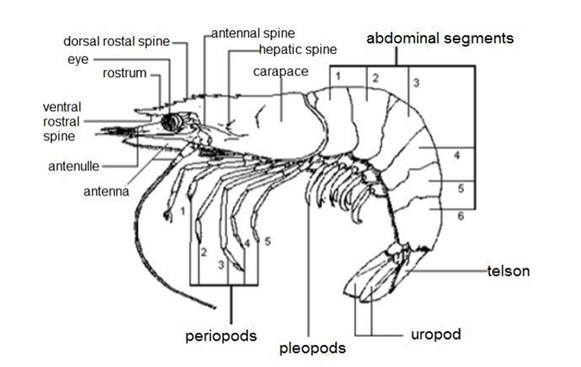
External anatomy of shrimp: Freshwater and saltwater have two body parts. The cephalothorax is part of the body that contains the head and thorax. It is protected by carapace plating. The cephalothorax has the rostrum, stalked eyes, carapace, first and second antennae, antennules, five sets of pereopods, maxillipeds, and mandibles. The abdomen has both upper and lower parts. Shrimp have swimmerets, which are also called pleopods. Shrimp’s tail is made up of three parts. Two are uropods, and the telson is the most pointed part in the middle. When swimming, pleopod tails work like airplane wings to steer. A thin membrane links six parts of the abdomen. The first through fifth segments are connected to five pairs of swimming legs. The sixth segment becomes a tail fan.
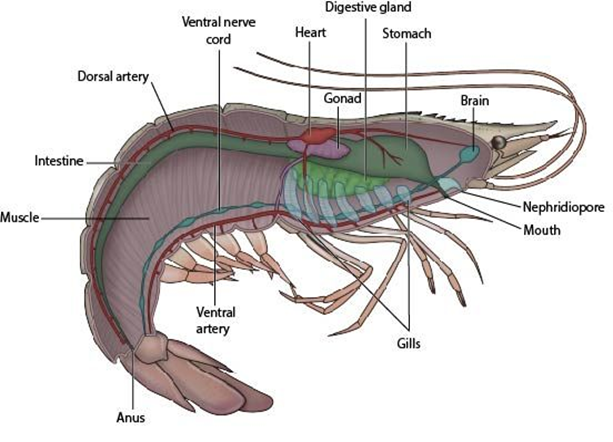
Internal anatomy of shrimp:
Most shrimps are omnivorous. The intestine appeared as dark line running down the back and along dorsal length of the abdomen; it is sometimes called the "mud vein"
https://www.dfo-mpo.gc.ca/science/aah-saa/publications/figures/shrimp-crevette-eng.html
Difference between shrimp and prawn:
|
Difference between shrimps and prawns | Martak.com
|
||
|
Items
|
Shrimp |
Prawn |
|
Body |
Curved body as thorax overlaps their head and abdomen and the second abdominal segment overlaps the first and third segment. |
Straight bodies with segments overlapping front-to-back. |
|
Legs
|
Have two pairs of claw-like legs |
Have three pairs of claw-like legs |
|
Gills
|
Plate-like gills |
Branching gills
|
|
Reproduction
|
Females brooding eggs on their pleopods known as (berried ) females. |
Fertilized eggs release into the water |
|
Habitat
|
Most are marine |
Most are freshwater |
|
Most cultured shrimp worldwide |
|||
|
Species (Scientific Name) |
Common Name |
Key Features |
Culture Regions |
|
Penaeus monodon |
Black Tiger Shrimp |
Large size, high market value, hardy |
Widely farmed in Asia (India, Thailand, Philippines) |
|
Penaeus vannamei ذات الارجل البيضاء (الفانمي) |
Whiteleg Shrimp |
Fast growth, high survival, dominates global production |
Latin America, Asia (China, Vietnam, Indonesia) "Egypt" |
|
Penaeus indicus الهندي الأبيض |
Indian White Shrimp |
Adaptable to varied salinity, good for semi-intensive farming |
India, Middle East "Egypt" |
|
Penaeus japonicus |
Kuruma Shrimp |
High-value species, prefers sandy bottoms, sensitive to salinity |
Japan, Taiwan |
|
Penaeus merguiensis |
Banana Shrimp |
Short culture period, moderate size |
Southeast Asia |
|
Penaeus semisulcatus الجمبري السويسي |
Green Tiger Shrimp |
Hardy, moderate growth |
Middle East, South Asia "Egypt" |
|
Metapenaeus ensis |
Greasyback Shrimp |
Smaller size, local importance |
India, Southeast Asia |
|
Metapenaeus monoceros |
Speckled Shrimp |
Short culture cycle, moderate demand |
South Asia |
|
Penaeus pulchricaudatus الجمبري القزاز https://marinebiodiversity.org.bd/species/penaeus-pulchricaudatus/ |
Kuruma Shrimp Tiger prawn |
Short culture cycle Market size 30 gm (4 months) |
Indo-Pacific, Red Sea, and Mediterranean Wild-caught prawn in Egypt around Bardawil lagoon and the Mediterranean coast. |
|
Freshwater prawn |
|||
|
Macrobrachium rosenbergii https://pescaflora.com/prawn جمبري المياة العذبة |
Giant river prawn |
Rearing period 6-8 months Market size 25-60 gm
|
Farming of Freshwater prawn: Integrated system: it cultured alongside freshwater fish using tilapia wastewater as nutrient source as well as efficient use of freshwater |
|
Central Institute of Freshwater Aquaculture. TNAU 2009-15 |
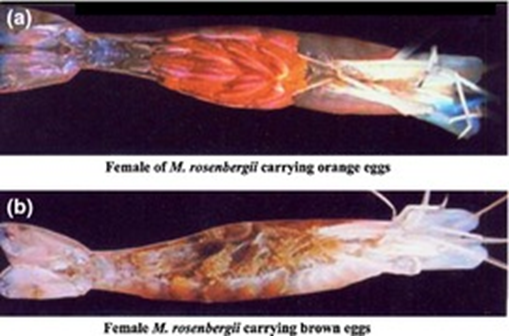
(a) Female of Macrobrachium rosenbergii carrying orange eggs. (b) Female M. rosenbergii carrying brown eggs. Madlen (2013) |
||
|
The Giant freshwater prawn, Macrobrachium rosenbergii is a valuable aquaculture species large market size , fast growth, tolerate wide range of environmental conditions, easily breeding under hatchery condition and higher survival from stocking to harvest (New, 2005), it found in freshwater estuarine areas and can culture in rice fields in brackish water with high water depth (0.5– 0.6m ) (Lan et al., 2023). Mature males are larger (about 25 cm) than the females (about 15 cm); second chelipeds are much larger and thicker, head is larger, and the abdomen is narrower. The head of the mature female and its second walking legs are much smaller than the adult male. A ripe or ‘ovigerous' female can easily be detected because the ovaries can be seen as large, orange-colored masses occupying a large portion of the dorsal and lateral parts of the cephalothorax. Mature prawn easily mates and spawns in captivity throughout the year with berried females (gravid females carrying ripe brown colored eggs) and successful mating can only take place between ripe females, which have just completed their pre-mating moult (usually at night) and are therefore soft-shelled, and hard-shelled males. |
|||
|
Life stages and corresponding feeding |
||
|
Stage |
Feeding Type |
Notes |
|
Egg |
None |
Embryo relies on yolk reserves until hatching |
|
Nauplius |
None (yolk-dependent) |
Non-feeding stage; lasts ~18–24 hours |
|
Protozoea |
Microalgae (Chaetoceros, Skeletonema) |
First feeding stage; requires clean water and stable salinity |
|
Mysis |
Microalgae + rotifers + Artemia nauplii |
Active swimmers need live feed for survival and growth |
|
Postlarva (PL) |
Artemia + formulated microdiet |
Transition to dry feed; monitor gut fill and hepatopancreas |
|
Juvenile |
Formulated pellet feed (40–45% protein) |
Feed 3–5 times/day; ensure uniform size and reduce competition |
|
Adult |
Pellet feed + supplements |
Optimize feed conversion ratio (FCR); avoid overfeeding |
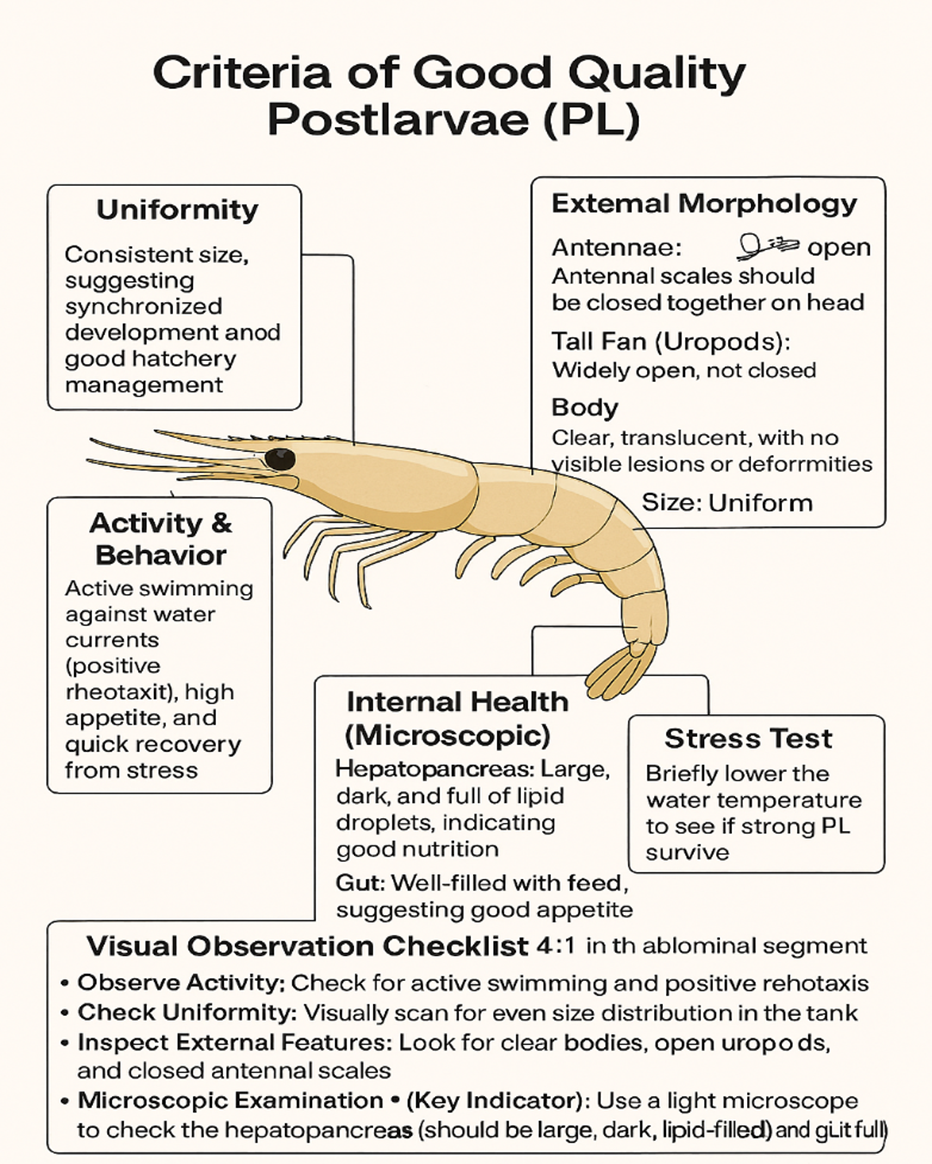
Culturing systems
· Traditional/extensive pond culture: low yield in earthen pond with minimal input.
· Semi-intensive and recirculating system: controlled environment in tanks or pool to reduce water exchange
· Intensive pond culture: use aeration, higher stocking densities and formulated feeds for increased production.
· Closed system: land-based, recirculating system that treats and reuse water, needs high aeration and technology.
Management
· Water quality: maintaining optimal salinity, temperature, Dissolved oxygen and ammonia level.
· Stocking: acclimating post-larvae to pond conditions and adjusting stocking rate (30-50/ m2 in pond). Ideal stage for stocking is post-larvae 10-12 days old as they are potent enough for pond conditions.
· Feeding: using formulated diet to meet requirements
· Biosecurity: Practices should be applied to prevent introduction and spread of diseases.
· Harvesting: during spring tides to collect hardened-shell shrimp, using nets at pond outlets.
- Most common shrimp viral diseases
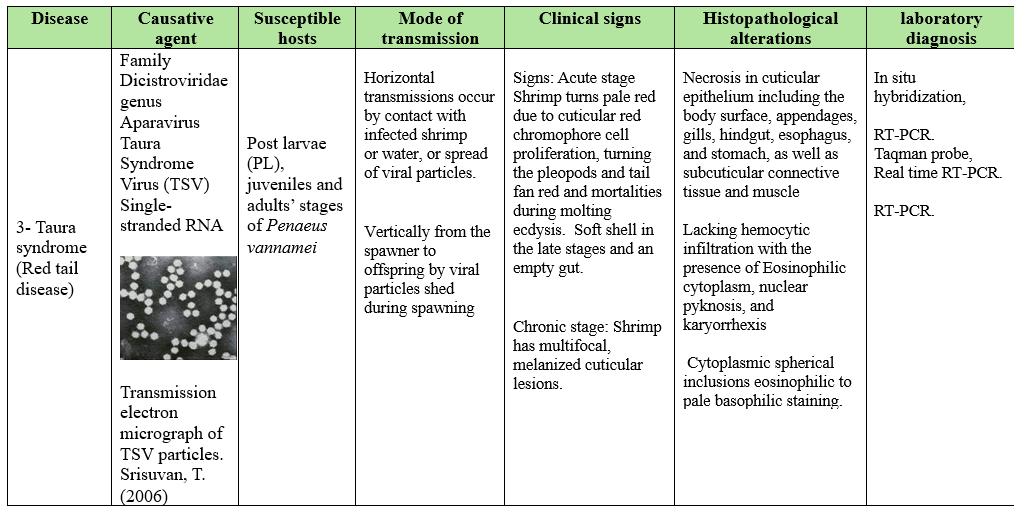
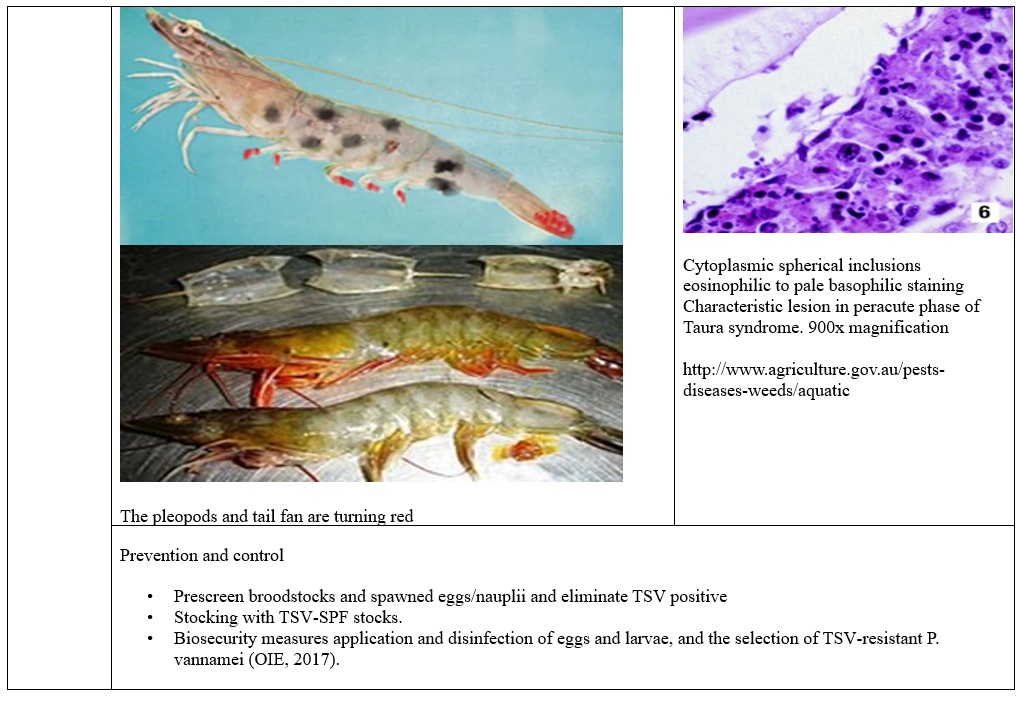
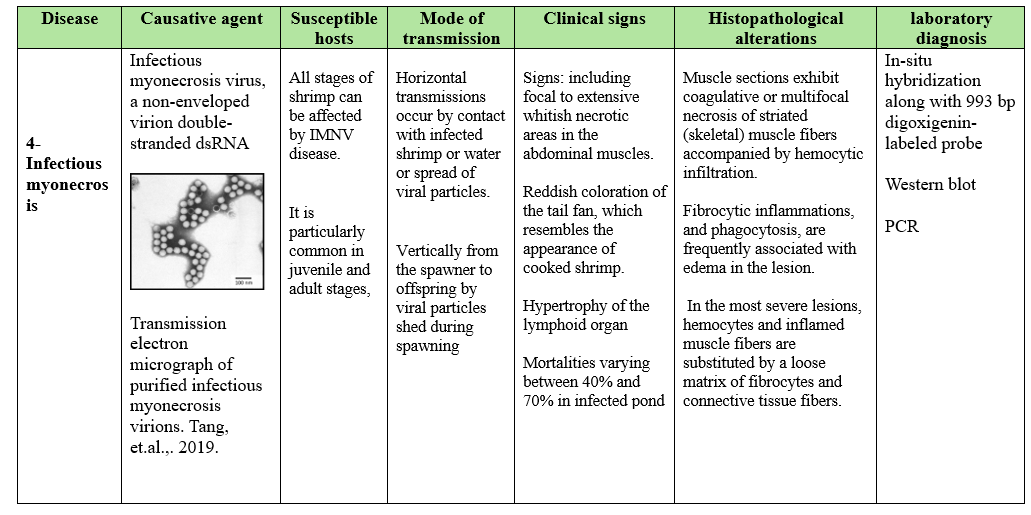
Viral diseases are very important in global shrimp aquaculture. The most serious viral diseases affecting shrimp include white spot syndrome virus (WSSV) highly lethal causing up to 100% mortality within 2-7 days, Yellow Head Virus (YHV) causing up to 100% mortality within 3-5 days and Taura syndrome Virus (TSV) causes significant economic impact on shrimp production.
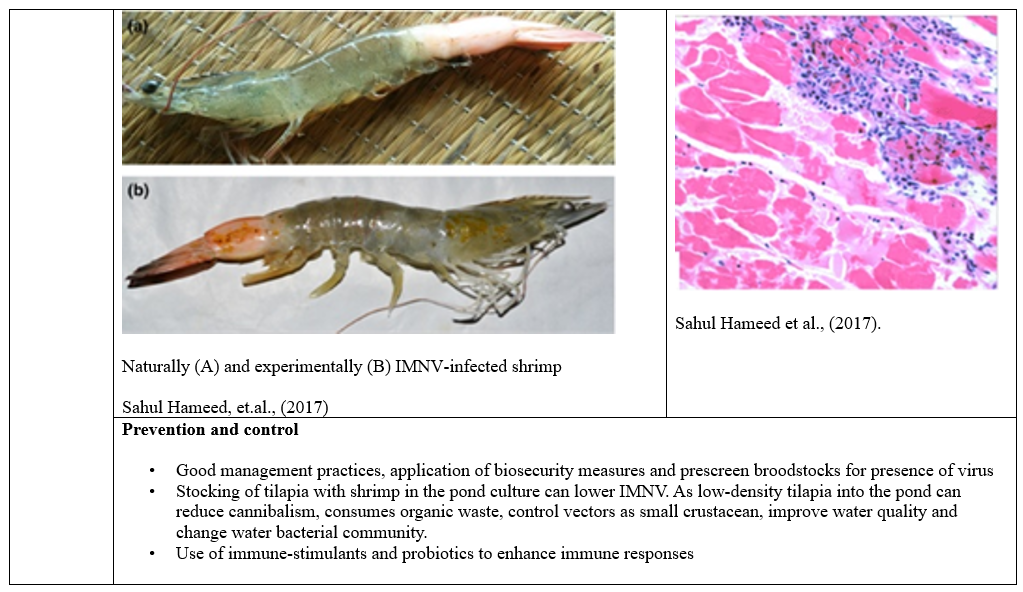
The diseases spread through contaminated water, direct contact and movement of infected shrimp so effective biosecurity measures and management practices are crucial to mitigate viral diseases impact on shrimp production. Disinfection of the surface of the eggs by iodophor treatment, a hatchery practice used to reduce the transmission of viral pathogens associated with egg, coelomic fluid or milt (A 10-30 min bath in 50-100 mg/L iodine solution as standard procedure performed both on recently fertilizes eggs during the water hardening step (Wood, 1979). Moreover, stocking of tilapia with shrimp in the pond culture can lower Infectious myonecrosis virus. As low-density tilapia into the pond can reduce cannibalism, consumes organic waste, control vectors as small crustacean, improve water quality and change water bacterial community.
Disease management strategies include:
· Specific pathogen free (SPF) shrimp post-larvae stocks: use SPF broodstock and post-larvae to reduce disease
· Vaccinations: explore vaccination to enhance shrimp immune response.
· Using RNA interference technology, it works by introducing double strand RNA (dsRNA) that trigger the suppression of virus replication and has potential to boost the shrimp innate immune response. Despite its promise, challenges like cost-effective dsRNA production and efficient delivery methods for large-scale farms still need to be overcome.
· Immunostimulation: Use immunostimulants to enhance innate immunity and provide viral resistance.
· Early viral detection: use PCR diagnostics for early detection and rapid response.
· Integrated disease management: combine biosecurity, management practices and treatment strategies.
· Regulating water quality parameters: monitoring water quality parameters such as oxygen, salinity, ammonia, and temperature.
· Selection of the resistant strains of shrimp.
Biosecurity measures include:
· Quarantine: quarantine for new shrimp before introducing to production system
· Sanitation: disinfected equipment, vehicles and personnel.
· Pathogen screening: regularly screen for pathogens using PCR or other diagnostic tools.
· Access control: restrict access to production areas.
Prevention and control
1. Developing domesticated SPF stocks, free of prevalent pathogens, as critical for predictable production stability. Significant efforts in breeding programs have already yielded stocks of *Penaeus vannamei* that demonstrate tolerance to TSV and IHHNV, despite lacking clarity on the underlying tolerance mechanisms, highlighting the need for persistent research in shrimp immunology.
2. The efficacy of shrimp vaccinations, traditional vaccination methods based on adaptive immunity are noted as inapplicable to invertebrates, leading to proposals for “immune priming” or “trained immunity.” Despite initial successes in protecting shrimp using killed pathogens and other immune response activators, no commercial products are presently available.
3. Additional research avenues include harnessing AMPs produced by shrimp in response to infections, which could provide targeted responses against specific pathogens.
4. Emerging techniques involve using heterologous proteins—either viral or host-derived—to impede virus attachment to shrimp cells. However, practical delivery methods for these proteins remain a barrier to commercialization.
5. Using dsRNA has shown promise in combating viral infections and could pave the way for large-scale implementation, pending reductions in production costs and advancements in delivery methods.
- Most common shrimp viral diseases




- References
· Aly, S. M., & Fathi, M. (2024). Advancing aquaculture biosecurity: a scientometric analysis and future outlook for disease prevention and environmental sustainability. Aquaculture International, 32(7), 8763-8789.
· Arulmoorthy, M. P., Anandajothi, E., Vasudevan, S., & Suresh, E. (2020). Major viral diseases in culturable penaeid shrimps: a review. Aquaculture International, 28(5), 1939-1967.
· Hounmanou, Y. M. G., Mdegela, R. H., Dougnon, T. V., Achoh, M. E., Mhongole, O. J., Agadjihouèdé, H., ... & Dalsgaard, A. (2018). Tilapia lake virus threatens tilapiines farming and food security: Socio-economic challenges and preventive measures in Sub-Saharan Africa. Aquaculture, 493, 123-129.
· Lan Lam ,Tam Hoang Liem Duc Vo , Nhut Long Duong , Thi Ngoc Anh Nguyen , Kurihara Akira , Ngoc Hai Tran (2023): The Effect of Water Depth on Efficiency of Freshwater Prawn (Macrobrachium rosenbergii, de Man) Culture in the Brackish Area. Egyptian Journal of Aquatic Biology & Fisheries Zoology Department, Faculty of Science, Ain Shams University, Cairo, Egypt. ISSN 1110 – 6131 Vol. 27(5): 289 – 302(2023).
· Lee, D.; Yu, Y.-B.; Choi, J.-H.; Jo, A.-H.; Hong, S.-M.; Kang, J.-C.; Kim, J.-H. Viral Shrimp Diseases Listed by the OIE: A Review.
· Madlen M Habashy (2013): On the breeding behaviour and reproduction of the freshwater prawn, Macrobrachium rosenbergii (de Man 1879) (Decapoda-Crustacea) under laboratory conditions. Aquaculture Research, 2013, 44, 395–403.
· Maezono, M., Nielsen, R., Buchmann, K., & Nielsen, M. (2025). The Current State of Knowledge of the Economic Impact of Diseases in Global Aquaculture. Reviews in Aquaculture, 17(3), e70039.
· Munang’andu, H. M., Mutoloki, S., & Evensen, Ø. (2016). Prevention and control of viral diseases in aquaculture. In Aquaculture virology (pp. 77-93). Academic Press.
· New M.B. (2005). Freshwater prawn farming: Global status, recent research and a glance at the future. Aquaculture Research, 35: 310-230.https://doi.org/10.1111/j.1365- 2109.2005.01237.x
· Obirikorang KA, Quagrainie K, Kassah JE and Von Ahnen M (2024) Editorial: Sustainable aquaculture production for improved food security. Front. Sustain. Food Syst. 8:1485956. doi: 10.3389/fsufs.2024.1485956.
· Rahaman, M. M., Sharma, B., Talukder, S., Uddin, M. J., Siddik, M. A., & Sarker, S. (2025). Viral Threats to Australian Fish and Prawns: Economic Impacts and Biosecurity Solutions—A Systematic Review. Viruses, 17(5), 692.
· Sahul Hameed, A S, S Abdul Majeed, S Vimal, N Madan, T Rajkumar, S Santhoshkumar, S Sivakumar, (2017). Studies on the occurrence of infectious myonecrosis virus in pond-reared Litopenaeus vannamei (Boone, 1931) in India. Journal of fish diseases, 40(12), 1823-1830.
· Sudhagar, A., Swaminathan, T. R., & Sood, N. (2024). Implications of Transboundary Diseases in Wild Finfish Populations: Insights for Conservation. In Sustainable Management of Fish Genetic Resources (pp. 223-236). Singapore: Springer Nature Singapore.
· Wood JW. (1979): Diseases of pacific salmon; their prevention and treatment. State of Washington, department of fisheries, hatchery division).
· Valero, Y., & Cuesta, A. (2023). Reassortant viruses threatening fish aquaculture. Reviews in Aquaculture, 15(4), 1720-1731.