Tilapia Lake Virus disease (TiLVD)
| Site: | EHC | Egyptian Health Council |
| Course: | Aquatic animal medicine Guidelines |
| Book: | Tilapia Lake Virus disease (TiLVD) |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:52 AM |
Description
"last update: 8 Jun 2026" Download Guideline
- Acknowledgment
We would like to acknowledge the committee of National Egyptian Guidelines for Veterinary Medical Interventions, Egyptian Health Council for adapting this guideline.
Executive Chief of the Egyptian Health Council: Prof. Mohamed Mustafa Lotief.
Head of the Committee: Prof. Ahmed M Byomi
The rapporteur of the Committee: Prof. Mohamed Mohamedy Ghanem.
Scientific Group Members: Prof. Gamal A. Sosa., Prof. Nabil Yassien, Prof. Ashraf Aldesoky Shamaa, Prof. Amany Abbass, Prof. Dalia Mansour, Dr. Essam Elmarakby, Dr. Mohamed Elsharkawy, Dr. Naglaa Radwan, Dr. Hend El Sheikh
Authors: Mohamed Faisal1,2; Adel A. Shaheen1, Amany A. Abbass1; Amel M. El Asely1; Eman A. Abd El-Gawad1; Hiam S. Elabd1; Aya F. Matter1; Hadeer A. Youssef1, and Amira M. El-Daim1.
1Department of Aquatic Animal Medicine, Faculty of Veterinary Medicine, Benha University, Egypt.
2College of Veterinary Medicine, Michigan State University, USA.
- Scope
Tilapia Lake Virus disease (TiLVD) is a globally emerging and notifiable viral disease that infects all tilapia species, regardless of whether they are farmed or wild. TiLVD outbreaks can cause massive losses as high as 95% that impact the community economies, where 65% of fish production in Egypt comes from tilapia.
- Summary
The causative agent of the disease is recently identified as Tilapia tilapine virus, which sometimes referred to as tilapia lake virus, is the only species belonging to the Tilapine virus genus associated with summer mortality syndrome of tilapia. There have been instances of co-infection involving multiple infections caused by bacteria, particularly with Aeromonas spp. The virus can infect any species of tilapia, whether wild or cultured. Any life stage of tilapia is vulnerable to the virus, and its mortality can range from 10 to 90%. Infected fish may display non pathgnomonic signs and lesions. The effective diagnostic approaches comprise the culture on specific cell lines, examination of pathological changes, and observation of virus particles using electron microscopy. A highly accurate PCR technique utilizes the semi-nested RT-PCR for detection. A trial for the development of treatment and vaccination still in progress.
- Introduction
Tilapia Lake virus disease (TiLVD ) is an emerging disease that was first identified at Galili Lake in 2014 (Eyngor et al., 2014). Ferguson et al. (2014) were the first to identify a novel disease in Ecuador called syncytial hepatitis of tilapia. Since then, outbreaks of TiLV have been reported in multiple countries, including Egypt (Fathi et al., 2017). In Egypt, there is speculation that the disease may be one of the factors contributing to the mortalities that have occurred over the summer since 2015, however, this has not been confirmed yet.
- Etiological agent
o Tilapia Lake Virus disease (TiLVD) is caused by Tilapia tilapine virus. Currently, TiLV has been categorized as a distinct family called Amnoonviridae, belonging to the genus Tilapinevirus (Adams et al., 2017), which is an enveloped, negative-sense single strand RNA virus with a genome composed of ten segments.
o TiLV infection can be transmitted horizontally through physical contact, mucus, feed and\ or water. It is believed that intragastric route is the primary route of infection (Pierezan et al., 2019). The virus can be transferred vertically from an asymptomatic adult carrier to their offsprings (Dong et al., 2020)
- Diagnosis
o Presumptive diagnostic assays
Common clinical signs
The diseased fish show general clinical signs as anorexia, anemia, emaciation, skin abrasion and discoloration, haemorrhagic patches on the skin, raised scales, exophthalmia, and abdominal distention (Figure 1).

Fig.1: Genral clinical signs of tilapia lake virus infected tilapia showing detached sclaes, skin discoloration and ulceration (red arrow head) [a], skin hemorrahges [b], abdominal distension (black star) [c], and bilateral exophthalmia (blue arrow) [d], corneal opacity [E]. Cited from Jansen, M. D., Dong, H. T., & Mohan, C. V. (2019).
Although TiLV doesn't cause specific signs on fish but the high mortality accompanied with corneal opacity (Eyngor et al., 2014).
Behavioral sign
behavioral changes like gathering in the bottom, sluggish movement, abnormal swimming behavior, and avoidance of schooling before death were also observed during the outbreak (Dong et al., 2017a).
Internal clinical signs
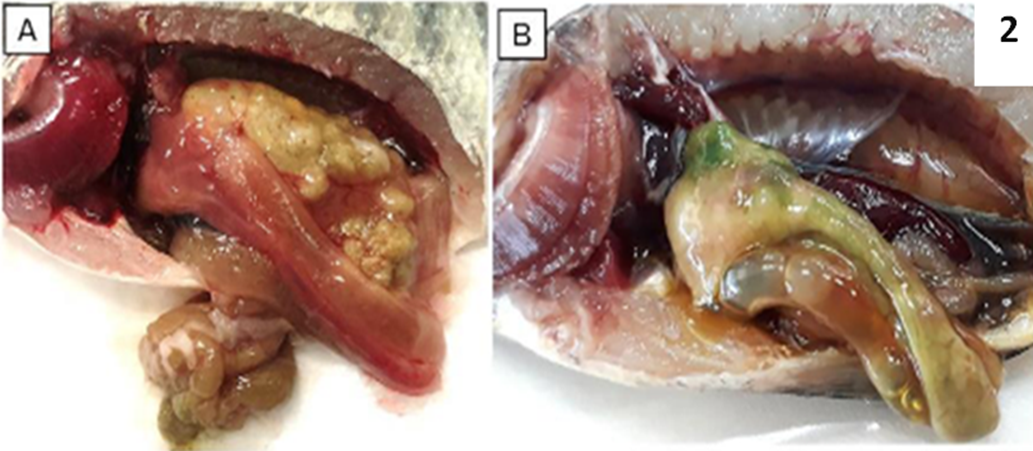
Liver appeared pale and in some cases, the liver tissue turned green or dark color. Also, infected fish show enlarged spleen and gall bladder, accumulation of fluid in the intestine and abdomen cavity (Figure, 2).
Fig 2: PM examination of normal tilapia (A), and TiLV infected tilapia showing pale gills and liver, congested spleen and kidney, enlarged gall bladder and green colored liver (B). cited from Tang, K.F.J., Bondad-Reantaso, M.G., Surachetpong, W., Dong, H.T., Fejzic, N., Wang, Q., Wajsbrot, N. & Hao, B. 2021.Tilapia lake virus disease strategy manual. FAO Fisheries and Aquaculture Circular No. 1220. Rome, FAO.https://doi.org/10.4060/cb7293en
· Lab Diagnosis
Collection of samples
Clinical samples include liver, spleen, kidney, heart, brain, gills, and whole fry. These samples used for histopathology, molecular assays and virus isolation on cell line.
The samples that will be used for:
- Histopathological examination: must be fixed in 10 % buffered formalin.
- Molecular techniques: the sample should be frozen (at -20 °C or -80 °C) and the others are preserved in 95% ethanol (or RNAlater, or equivalent reagent). Indeed, the combination of RNAlater® with deep-freezing at -20 ºC proved to be the most optimal storage conditions for preserving TiLV genomic RNA (Tang et al., 2021).
- Viral isolation: should be aseptically collected and immersed in Hanks’ balanced salt solution (HBSS). Alternatively, the sample can be promptly frozen at -80 °C or preserved on dry ice..
- Non-lethal samples, such as mucus, skin scraping, and blood, can be used as alternatives to killing fish for research purposes. These samples can be used for pond site diagnosis when using portable diagnostic instrument. After the fish have been sampled, they can be safely returned to the ponds.
➡️ Histopathological diagnosis
The liver of tilapia exhibits the presence of syncytial cells, which are characterised by the presence of many nuclei within each cell. These nuclei can range from 3-4 up to 15-20 per cell (Del-Pozo et al., 2017).Other lesions include massive cellular necrosis with pyknotic and karyolytic nuclei are also found in the hepatocytes, together with eosinophilic cytoplasmic inclusion bodies (Senapin et al., 2018). Figure 3.
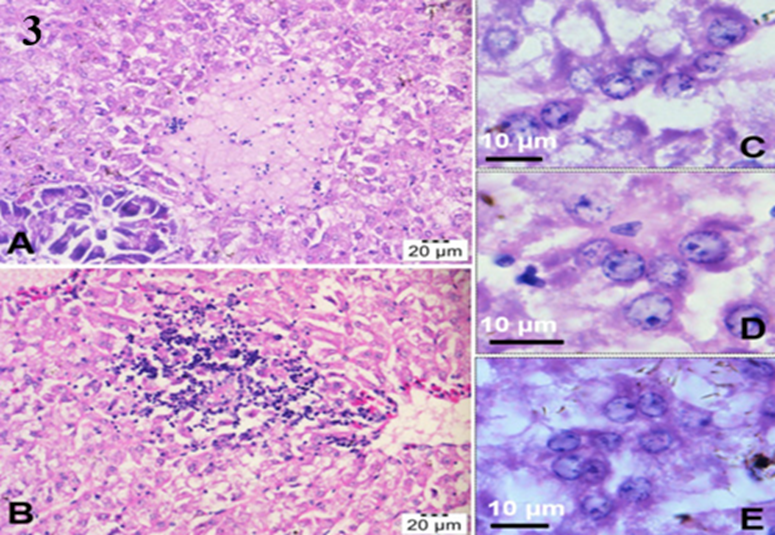
Fig3: H&E stained sections of the fish liver showing focal necrosis of hepatocytes and infiltration of lymphocytic inflammatory cells (A, B). Presence of hepatocytes resembling giant cells which contained multiple nuclei (C–E) was also observed. Senapin et. al (2018).
➡️Transmission electron microscope
Diagnosis of TiLV can be confirmed by Transmission electron micrographs (TEM), which reveals the presence of spherical virus particles in the cytoplasmic area (Dong et al., 2017b) Figure 4.
NB. Samples which are used for (TEM) must be fixed in 2 % glutaraldehyde and stored at 4 °C
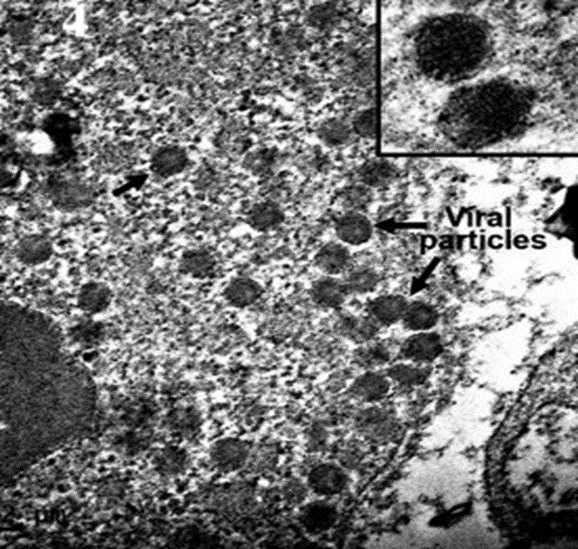
Fig 4: TEM ultrastrcutre of TiLV infected liver, with intracytoplasmic virions observed at higher magnification showing round-shaped particles with diameter around 100 nm, Dong et al. (2017b).
Molecular Techniques
- RT-PCR
Several techniques of RT-PCR for accurate and sensitive detection of TiLV in samples even in a symptomtic or apparent healthy fish. Single RT-PCR, nested RT-PCR, SYBR green-based qPCR, probed-based qPCR, semi-nested RT-PCR, in situ hybridization (ISH) using DIG-labelled probe. The scientists have developed commercial kits based on RT-PCR and RT-qPCR.
- RT-LAMP
This approach enables the identification of TiLV at the pond site using inexpensive equipment such as a water bath.
- Cell culture
TiLV can be cultured on many cell lines including E-11 (derived from Channa striata, a species of snakehead fish), CFF (from Pristolepis fasciatus, a species of Malayan leaffish), OmB (from Oreochromis mossambicus), OnlB (from Nile tilapia brain), OnlL (from Nile tilapia liver), or CAMB (from hybrid snakehead brain). Following the introduction of the virus into the cells, the cells should be placed in an incubator at a temperature range of 25 to 27 ºC for a period of 4 to 12 days. During this time, the cells should be monitored daily for any cytopathic effects (CPE). Infected cells exhibit CPE such as shrinkage, aggregation, rounding up, and detachment from the culture surface (Figure, 5) Tattiyapong et al., (2017)
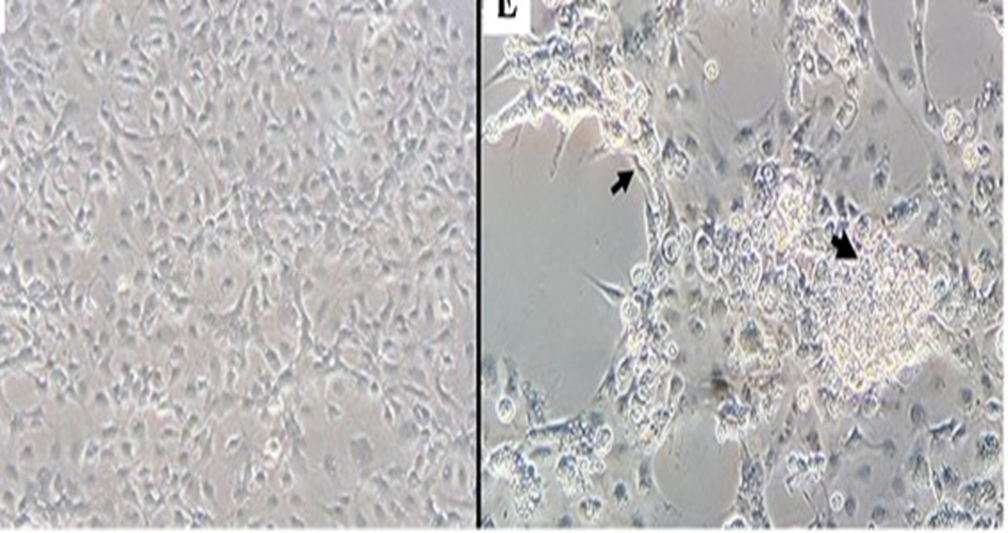
Fig 5: (A) E-11cells inoculated with tissue homogenate prepared from normal tilapia brain; (B) E-11 cells inoculated with tissue homogenate prepared from TiLV-positive brain, CPE with cell shrinkage and syncytial formation (black arrow), four days post-inoculation. Tattiyapong, et al, (2017).
- Prevention and control
o Tilapia lake virus free tilapia
To limit the spread of TiLVD, it is crucial to use SPF tilapia that are devoid of TiLV to avoid the introduction of infected tilapia (Machimbirike et al., 2019).
o Tilapia lake virus resistant tilapia
Selective breeding techniques to develop tilapia species with resistance to the disease can prove highly successful in disease control (Barría et al., 2020).
o Vaccination
No commercial vaccine is currently available. Tilapia produce anti-TiLV antibodies within 7–10 days of virus inoculation (Tattiyapong et al., 2020). Many TiLV immunizations have shown efficacy, however survival rates vary. The TiLV vaccine, produced using β-propiolactone (BPL) inactivation and Montanide IMS 1312 adjuvant, is an example (Zeng et al., 2021). This vaccination has 85.7% survival rate in lab challenge testing. Two alternative vaccines, injectable heat-killed and formalin-killed, are effective against Nile tilapia TiLV infection. Mai et al. (2021) found that the heat-killed vaccination had a relative survival rate of 71.3% and the formalin-killed vaccine 79.6%.
o Immunstimulants and probiotics
Immunostimulants can be used as an alternative to vaccines. They have broad-spectrum activity and are relatively cost-effective than vaccines. They are more suitable for younger fish as adaptive immunity is not fully developed. Probiotic bacteria can enhance fish immunity by promoting phagocytic activity, complement responses, lysozyme levels, and cytokine production. Probiotics must demonstrate efficacy and safety.
Zoonotic importance
No zoonotic importance was documented.
- Refrences
1. Adams, M. J., Lefkowitz, E. J., King, A. M., Harrach, B., Harrison, R. L., Knowles, N. J., et al. (2017). Changes to taxonomy and the international code of virus classification and nomenclature ratified by the international committee on taxonomy of viruses. Archives of Virology, 162(8), 2505–2538.
2. Barría, A., Trinh, T. Q., Mahmuddin, M., Benzie, J. A., Chadag, V. M., & Houston, R. D. (2020). Genetic parameters for resistance to Tilapia lake virus (TiLV) in nile tilapia (Oreochromis niloticus). Aquaculture, 522, 735126.
3.
Del-Pozo, J., Mishra, N., Kabuusu, R., Cheetham, S.,
Eldar, A., Bacharach, E., &
Ferguson, H. W. (2017). Syncytial hepatitis of tilapia (Oreochromis niloticus
L.) is
associated with orthomyxovirus-like virions in hepatocytes. Veterinary Pathology,
54
(1), 164–170.
4. Dong, H. T., Ataguba, G. A., Khunrae, P., Rattanarojpong, T., & Senapin, S. (2017a). Evidence of TiLV infection in tilapia hatcheries from 2012 to 2017 reveals probable global spread of the disease. Aquaculture, 479, 579–583.
5.
Dong, H. T., Siriroob, S., Meemetta, W.,
Santimanawong, W., Gangnonngiw, W.,
Pirarat, N., & Senapin, S. (2017b). Emergence of tilapia lake virus in
Thailand and an
alternative semi-nested RT-PCR for detection. Aquaculture, 476, 111–118.
6. Dong, H. T., Senapin, S., Gangnonngiw, W., Nguyen, V. V., Rodkhum, C., Debnath, P. P., ... & Mohan, C. V. (2020). Experimental infection reveals transmission of tilapia lake virus (TiLV) from tilapia broodstock to their reproductive organs and fertilized eggs. Aquaculture, 515, 734541.
7. Eyngor, M., Zamostiano, R., Tsofack, J. E. K., Berkowitz, A., Bercovier, H., Tinman, S., & Eldar, A. (2014). Identification of a novel RNA virus lethal to tilapia. Journal of Clinical Microbiology, 52(12), 4137–4146.
8. Fathi, M., Dickson, C., Dickson, M., Leschen, W., Baily, J., Muir, F., & Weidmann, M. (2017). Identification of Tilapia Lake Virus in Egypt in Nile tilapia affected by ‘summer mortality’syndrome. Aquaculture, 473, 430–432.
9. Ferguson, H. W., Kabuusu, R., Beltran, S., Reyes, E., Lince, J. A., & Del Pozo, J. (2014). Syncytial hepatitis of farmed tilapia, Oreochromis niloticus (L.): A case report. Journal of Fish Diseases, 37(6), 583–589.
10. Machimbirike, V. I., Jansen, M. D., Senapin, S., Khunrae, P., Rattanarojpong, T., & Dong, H. T. (2019). Viral infections in tilapines: More than just tilapia lake virus. Aquaculture, 503, 508-518.
11. Mai, T. T., Kayansamruaj, P., Taengphu, S., Senapin, S., Costa, J. Z., del‐Pozo, J., ... & Dong, H. T. (2021). Efficacy of heat‐killed and formalin‐killed vaccines against Tilapia tilapinevirus in juvenile Nile tilapia (Oreochromis niloticus). Journal of fish diseases, 44(12), 2097-2109.
12. Pierezan, F., Yun, S., Surachetpong, W., & Soto, E. (2019). Intragastric and intracoelomic injection challenge models of tilapia lake virus infection in Nile tilapia (Oreochromis niloticus L.) fingerlings. Journal of Fish Diseases, 42(9), 1301–1307.
13. Senapin, S., Shyam, K. U., Meemetta, W., Rattanarojpong, T., & Dong, H. T. (2018). Inapparent infection cases of tilapia lake virus (TiLV) in farmed tilapia. Aquaculture, 487, 51–55.
14. Tang, K.F.J., Bondad-Reantaso, M.G., Surachetpong, W., Dong, H.T., Fejzic, N., Wang, Q., Wajsbrot, N. & Hao, B. (2021).Tilapia lake virus disease strategy manual. FAO Fisheries and Aquaculture Circular No. 1220. Rome, FAO.https://doi.org/10.4060/cb7293en
15.
Tattiyapong, P., Dachavichitlead, W., &
Surachetpong, W. (2017). Experimental
infection of Tilapia lake virus (TiLV) in nile tilapia (Oreochromis
niloticus) and red
tilapia (Oreochromis spp.). Veterinary Microbiology, 207, 170–177
16. Tattiyapong, P., Dechavichitlead, W., Waltzek, T. B., & Surachetpong, W. (2020). Tilapia develop protective immunity including a humoral response following exposure to tilapia lake virus. Fish & Shellfish Immunology, 106, 666–674.
17. Zeng, W., Wang, Y., Hu, H., Wang, Q., Bergmann, S.M., Wang, Y., Li, B., et al. 2021. Cell culture-derived tilapia lake virus-inactivated vaccine containing Montanide adjuvant provides high protection against viral challenge for Tilapia. Vaccines, 9(2): 86.