the Management of Benign Liver Lesions
| Site: | EHC | Egyptian Health Council |
| Course: | General surgery Guidelines |
| Book: | the Management of Benign Liver Lesions |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:52 AM |
Description
"last update: 12 Jan 2026" Download Guideline
- Executive summary
These guidelines offer evidence-based recommendations on the targeted levels of clinical practice guidelines and provide healthcare professionals with practice guidance on the management of benign hepatic lesions.
· In patients with a focal liver lesion of uncertain aetiology, we recommend multiphasic contrast-enhanced imaging, preferably MRI or CT, performed with late arterial, portal venous, and delayed phases, (Strong recommendation).
· In patients with a normal or healthy liver, a hyperechoic lesion, in ultrasonography, is very likely to be a liver haemangioma. With typical radiology (homogeneous hyperechoic, sharp margin, posterior enhancement, and absence of halo sign) in a lesion less than 3 cm, we advise performing ultrasound for the diagnosis, (Conditional recommendation).
· We recommend the diagnosis of haemangioma by contrast enhanced imaging, (CT and/or MRI). It is based on a typical vascular profile characterized by peripheral and globular enhancement on arterial phase followed by a central enhancement on delayed phases. MRI provides additional findings such as lesion signal on T1-, T2- weighted sequences, and diffusion imaging, (Strong recommendation).
· Pregnancy and oral contraceptives are not contraindicated, (Conditional recommendation).
· We recommend conservative management for typical cases of haemangioma, (Strong recommendation).
· In patients with asymptomatic haemangioma < 10 cm we suggest no imaging follow-up, due to its benign course, (Conditional recommendation).
· In symptomatic patients or with haemangiomas > 10 cm we advise follow-up, due to possible complications, (Conditional recommendation).
· When indicated, we advise to follow-up with ultrasound in view of non-invasiveness, low costs, and absence of biological risk, (Conditional recommendation).
· In haemangiomas > 15 cm, due to the difficulty of assessing dimensional variations by means of ultrasound, we advise performing magnetic resonance, (Conditional recommendation).
· In patients with symptomatic haemangiomas (Kasabach- Merritt syndrome or bulk symptoms) or pedunculated haemangiomas or haemangiomas with a diameter of 10 cm or more, we advise performing surgical treatment, (Conditional recommendation).
· For symptomatic haemangiomas less than 10 cm, we advise performing loco-regional ablation techniques initially. In case of unfavourable clinical evolution or volume increase after treatment, we advise performing resection, (Conditional recommendation).
· In symptomatic patients with unresectable giant hepatic haemangioma or multiple haemangiomas, we advise performing liver transplantation as a feasible treatment, (Conditional recommendation).
· We recommend CEUS, CT, or MRI for diagnosing FNH with nearly 100% specificity. MRI has the highest diagnostic performance overall, (Strong recommendation).
· We advise evaluating patients with focal liver lesions that are suspicious for focal nodular hyperplasia using multiphase MRI with contrast agents to distinguish focal nodular hyperplasia from hepatocellular adenoma, (Conditional recommendation).
· For a lesion typical of FNH, we advise against routine follow-up, (Conditional recommendation).
· We do not advise routinely discontinuing oral contraceptives in patients diagnosed with focal nodular hyperplasia, (Conditional recommendation).
· We recommend against treatment, if imaging is typical, or the patient is asymptomatic, (Strong recommendation).
· In symptomatic adults with focal nodular hyperplasia, we advise performing surgical treatment of the lesion, as it might improve the quality of life. However, follow-up does not appear associated with the occurrence of major complications, (Conditional recommendation).
· We recommend MRI for diagnosing HCA as it is superior to all other imaging modalities and due to its intrinsic properties to detect fat and vascular spaces. It offers an opportunity to subtype HCA up to 80%, (Strong recommendation).
· Treatment decisions should be based on gender, and size and pattern of progression of adenoma, (Strong recommendation).
· We recommend discontinuation of oral contraceptives or intrauterine devices that are hormone impregnated in patients with hepatic adenomas, (Strong recommendation)
· We advise encouraging weight loss in overweight or obese patients with hepatic adenomas, (Conditional recommendation, very low certainty of evidence, (6)).
· HCA resection is advised in any instance of proven β-catenin mutation in both sexes, (Conditional recommendation).
· HCA resection is advised irrespective of size in men, (Conditional recommendation).
· In women with lesions less than 5 cm, we advise annual imaging reassessment. (Conditional recommendation).
· In women with nodules equal or greater than 5 cm and those continuing to grow, we advise resection, (Conditional recommendation).
· In bleeding HCA with hemodynamic instability, we advise performing embolization. Resection is advised for residual viable lesions on follow-up imaging, (Conditional recommendation).
· In patients with hepatic adenomas requiring treatment who are unable to undergo surgical resection, we advise performing embolization or ablation as alternative treatment approaches, (Conditional recommendation).
· In patients with ruptured hepatic adenomas, we advise hemodynamic stabilization followed by embolization and/or surgical resection, (Conditional recommendation).
· In patients with multiple HCA, management is advised to be based on the size of the largest tumor, (Conditional recommendation).
· We advise performing hepatic resection in unilobular disease. In those cases with more widespread HCAs, resection of the largest adenomas may be an option, (Conditional recommendation).
· Liver transplantation is not advised in multiple HCA, but may be advised in individuals with underlying liver disease, (Conditional recommendation).
· In patients with asymptomatic simple hepatic cysts, regardless of size, we recommend expectant management without need for routine surveillance or intervention, (Strong recommendation).
· In patients with simple hepatic cysts with specific high-risk features seen on ultrasound (e.g. septations, calcifications, mural thickening or nodularity, heterogeneity, and presence of daughter cysts), we recommend further investigation with CT or MRI, (Strong recommendation).
· We advise performing surgical cyst fenestration or aspiration with sclerotherapy for management of patients with symptomatic simple hepatic cysts, (Conditional recommendation).
· We recommend chemical and cytological examination for aspirated fluid, (Good practice statement).
· We advise discontinuation of exogenous oestrogen use in women with polycystic liver disease, (Conditional recommendation).
· For patients with PCLD with numerous small- to medium-sized cysts throughout the liver not amenable to surgical resection, we advise performing cyst fenestration, or aspiration sclerotherapy, (Conditional recommendation).
· For patients with symptomatic ADPKD with concurrent PCLD, we advise medical management using somatostatin analogues, (Conditional recommendation).
· Antihelmenthic treatment is recommended for all viable cysts disregarding any further surgical or non-surgical modality of treatment, (Good practice statement).
· We advise surgical management in patients with complicated hydatid cysts (i.e., those with biliary fistulas or cysts communicating with the biliary tree, multiseptated cysts, rupture or haemorrhage, secondary infection, or percutaneously inaccessible cysts) provided there is no contraindication to surgery, (Conditional recommendation).
· In patients with uncomplicated hydatid cysts in whom surgery is not an option, we advise performing percutaneous treatment with PAIR with adjunct antihelminthic therapy, (Conditional recommendation).
- Recommendations
Section1: General:
· In patients with a focal liver lesion of uncertain aetiology, we recommend multiphasic contrast-enhanced imaging, preferably MRI or CT, performed with late arterial, portal venous, and delayed phases, (Strong recommendation, low certainty evidence, (6)).
Section 2: Hepatic haemangioma:
· In patients with a normal or healthy liver, a hyperechoic lesion, in ultrasonography, is very likely to be a liver haemangioma. With typical radiology (homogeneous hyperechoic, sharp margin, posterior enhancement, and absence of halo sign) in a lesion less than 3 cm, we advise performing ultrasound for the diagnosis, (Conditional recommendation, moderate certainty evidence, (4)).
· We recommend the diagnosis of haemangioma by contrast enhanced imaging, (CT and/or MRI). It is based on a typical vascular profile characterized by peripheral and globular enhancement on arterial phase followed by a central enhancement on delayed phases. MRI provides additional findings such as lesion signal on T1-, T2- weighted sequences, and diffusion imaging, (Strong recommendation, moderate certainty evidence, (4)).
· Pregnancy and oral contraceptives are not contraindicated, (Conditional recommendation, low certainty evidence, (4)).
· We recommend conservative management for typical cases of haemangioma, (Strong recommendation, moderate certainty evidence, (4)).
· In patients with asymptomatic haemangioma < 10 cm we suggest no imaging follow-up, due to its benign course, (Conditional recommendation, moderate certainty evidence, (4)).
· In symptomatic patients or with haemangiomas > 10 cm we advise follow-up, due to possible complications, (Conditional recommendation, very low certainty evidence, (7)).
· When indicated, we advise to follow-up with ultrasound in view of non-invasiveness, low costs, and absence of biological risk, (Conditional recommendation, moderate certainty evidence, (4)).
· In haemangiomas > 15 cm, due to the difficulty of assessing dimensional variations by means of ultrasound, we advise performing magnetic resonance, (Conditional recommendation, very low certainty evidence, (7)).
· In patients with symptomatic haemangiomas (Kasabach- Merritt syndrome or bulk symptoms) or pedunculated haemangiomas or haemangiomas with a diameter of 10 cm or more, we advise performing surgical treatment, (Conditional recommendation, very low certainty evidence, (7)).
· For symptomatic haemangiomas less than 10 cm, we advise performing loco-regional ablation techniques initially. In case of unfavourable clinical evolution or volume increase after treatment, we advise performing resection, (Conditional recommendation, very low certainty evidence, (4 &7).
· In symptomatic patients with unresectable giant hepatic haemangioma or multiple haemangiomas, we advise performing liver transplantation as a feasible treatment, (Conditional recommendation, very low certainty evidence, (7)).
Section 3: Focal nodular hyperplasia:
· We recommend CEUS, CT, or MRI for diagnosing FNH with nearly 100% specificity. MRI has the highest diagnostic performance overall, (Strong recommendation, moderate certainty evidence, (4)).
· We advise evaluating patients with focal liver lesions that are suspicious for focal nodular hyperplasia using multiphase MRI with contrast agents to distinguish focal nodular hyperplasia from hepatocellular adenoma, (Conditional recommendation, low certainty evidence, (6)).
· For a lesion typical of FNH, we advise against routine follow-up, (Conditional recommendation, low certainty evidence, (4)).
· We do not advise routinely discontinuing oral contraceptives in patients diagnosed with focal nodular hyperplasia, (Conditional recommendation, low certainty evidence, (7)).
· We recommend against treatment, if imaging is typical, or the patient is asymptomatic, (Strong recommendation, low certainty evidence, (4)).
· In symptomatic adults with focal nodular hyperplasia, we advise performing surgical treatment of the lesion, as it might improve the quality of life. However, follow-up does not appear associated with the occurrence of major complications, (Conditional recommendation, very low certainty evidence, (7)).
Section 4: Hepatocellular adenoma:
· We recommend MRI for diagnosing HCA as it is superior to all other imaging modalities and due to its intrinsic properties to detect fat and vascular spaces. It offers an opportunity to subtype HCA up to 80%, (Strong recommendation, moderate certainty evidence, (4)).
· Treatment decisions should be based on gender, and size and pattern of progression of adenoma, (Strong recommendation, moderate certainty evidence, (4)).
· We recommend discontinuation of oral contraceptives or intrauterine devices that are hormone impregnated in patients with hepatic adenomas, (Strong recommendation, low certainty evidence, (4, 6 &7)).
· We advise encouraging weight loss in overweight or obese patients with hepatic adenomas, (Conditional recommendation, very low certainty of evidence, (6)).
· HCA resection is advised in any instance of proven β-catenin mutation in both sexes, (Conditional recommendation, low certainty evidence, (4)).
· HCA resection is advised irrespective of size in men, (Conditional recommendation, low certainty evidence, (4)).
· In women with lesions less than 5 cm, we advise annual imaging reassessment. (Conditional recommendation, very low certainty of evidence, (4 & 6)).
· In women with nodules equal or greater than 5 cm and those continuing to grow, we advise resection, (Conditional recommendation, low certainty evidence, (4)).
· In bleeding HCA with hemodynamic instability, we advise performing embolization. Resection is advised for residual viable lesions on follow-up imaging, (Conditional recommendation, very low certainty of evidence, (4)).
· In patients with hepatic adenomas requiring treatment who are unable to undergo surgical resection, we advise performing embolization or ablation as alternative treatment approaches, (Conditional recommendation, low certainty of evidence, (4)).
· In patients with ruptured hepatic adenomas, we advise hemodynamic stabilization followed by embolization and/or surgical resection, (Conditional recommendation, very low certainty of evidence, (6)).
· In patients with multiple HCA, management is advised to be based on the size of the largest tumor, (Conditional recommendation, very low certainty of evidence, (6)).
· We advise performing hepatic resection in unilobular disease. In those cases with more widespread HCAs, resection of the largest adenomas may be an option, (Conditional recommendation, very low certainty of evidence, (6)).
· Liver transplantation is not advised in multiple HCA, but may be advised in individuals with underlying liver disease, (Conditional recommendation, very low certainty of evidence, (6)).
Section 6: Simple hepatic cysts:
· In patients with asymptomatic simple hepatic cysts, regardless of size, we recommend expectant management without need for routine surveillance or intervention, (Strong recommendation, low certainty evidence, (6)).
· In patients with simple hepatic cysts with specific high-risk features seen on ultrasound (e.g. septations, calcifications, mural thickening or nodularity, heterogeneity, and presence of daughter cysts), we recommend further investigation with CT or MRI, (Strong recommendation, low certainty evidence, (6)).
· We advise performing surgical cyst fenestration or aspiration with sclerotherapy for management of patients with symptomatic simple hepatic cysts, (Conditional recommendation, low certainty evidence, (6)).
· We recommend chemical and cytological examination for aspirated fluid, (Good practice statement).
· We advise discontinuation of exogenous oestrogen use in women with polycystic liver disease, (Conditional recommendation, very low certainty evidence, (6)).
· For patients with PCLD with numerous small- to medium-sized cysts throughout the liver not amenable to surgical resection, we advise performing cyst fenestration, or aspiration sclerotherapy, (Conditional recommendation, moderate certainty evidence, (6)).
· For patients with symptomatic ADPKD with concurrent PCLD, we advise medical management using somatostatin analogues, (Conditional recommendation, moderate certainty evidence, (6)).
Section 7: Hydatid (echinococcal) cysts:
· Antihelmenthic treatment is recommended for all viable cysts disregarding any further surgical or non-surgical modality of treatment, (Good practice statement).
· We advise surgical management in patients with complicated hydatid cysts (i.e., those with biliary fistulas or cysts communicating with the biliary tree, multiseptated cysts, rupture or haemorrhage, secondary infection, or percutaneously inaccessible cysts) provided there is no contraindication to surgery, (Conditional recommendation, very low certainty evidence, (6)).
· In patients with uncomplicated hydatid cysts in whom surgery is not an option, we advise performing percutaneous treatment with PAIR with adjunct antihelminthic therapy, (Conditional recommendation, low certainty evidence, (6)).
- Acknowledgement
We would like to acknowledge the Guideline Development Group, (GDG) committee for developing this guideline.
Chair of GDG: Mostafa Abdel-Hamed Soliman, Professor of Surgery, Cairo University.
Moderator of GDG: Mohamed Ali Mohamed Nada, Professor of Surgery, Ain-Shams University.
Members of GDG (Alphabetically):
Abdel-Wahab Mohamed Ezzat, Professor of Surgery, Ain -Shams University.
Ahmed Abdel-Raouf Elgeidie, Professor of Digestive Surgery, Mansoura University.
Alaa Abdallah, Professor of Surgery, Ain -Shams University.
Atef Abdel-Ghani Salem, Professor of Surgery, Benha University.
Hesham Abdel-Raouf El-Akkad, Professor of Surgery, Ain-Shams University.
Ibrahim El-Zayat, Head of Surgery Department, Aswan University.
Khaled Abdallah El-Fiky, Professor of Surgery, Ain-Shams University.
Khaled Amer, Professor of Surgery, Military Medical Academy.
Khaled Safwat, Professor of Surgical Oncology and Endoscopy, Zagazig University.
Mohamed Ibrahim Abdel-Hamed Al-Said, Professor of Surgery, Zagazig University.
Tarek Ibrahim, Professor of Surgery, National Liver Institute, Menofia University.
- Abbreviations
|
ADPKD |
Autosomal Dominant Polycystic Kidney Disease |
|
AGREEII |
Appraisal of Guidelines for Research and Evaluation II |
|
β-HCA |
β-catenin Activated Hepatocellular Adenoma |
|
CRP |
C-reactive Protein |
|
CT |
Computed Tomography |
|
CEUS |
Contrast-Enhanced Ultrasound |
|
FNA |
Fine Needle Aspiration |
|
FNH |
Focal Nodular Hyperplasia |
|
GDG |
Guidelines Development Group |
|
GRADE |
Grading of Recommendations, Assessment, Development and Evaluation |
|
HCA |
Hepatocellular Adenoma |
|
HCC |
Hepatocellular Carcinoma |
|
H_HCA |
Hepatocyte Nuclear Factor _1a Hepatocellular Adenoma |
|
HNF-1a |
Hepatocyte Nuclear Factor-1a |
|
I-HCA |
Inflammatory Hepatocellular Adenomas |
|
MRI |
Magnetic Resonance Imaging |
|
OCP |
Oral Contraceptive Pills |
|
PAIR PCLD |
Puncture, Aspiration, Injection of Scolicidal Agent, and Reaspiration Polycystic Liver Disease |
- Glossary
Hemangioma:
Hemangiomas are the most common benign non-cystic liver lesions, occurring in up to 20% of the population, with a reported preponderance in women at a 4:1 ratio. They are benign mesenchymal vascular lesions consisting of clusters of blood- filled cavities lined by endothelial cells, ranging in size from a few millimeters to greater than 20 cm. Hemangiomas are believed to arise from a congenital abnormality in vasculogenesis, growing slowly from birth. Increase in size of hemangiomas can occur and is favored to be due to progressive ectasia of the vasculature and not related to hypertrophy of the lesion, (1).
Focal nodular hyperplasia:
FNH is the second most common solid liver lesion, with 0.3%–3% of people having FNH on autopsy. FNH is favored to arise as a local reaction to vascular abnormalities, specifically aberrant hemodynamics within the liver, usually secondary to an aberrant dystrophic artery or a vascular injury, resulting in a disturbance of local blood flow, which can result in hyper-perfusion, oxidative stress from hypoxia, and hepatic stellate cell response, all of which result in a hyperplastic microenvironment and FNH development, (2).
Hepatocellular adenoma:
HCAs represent a benign proliferation of mature hepatocytes, which can develop in a background of an otherwise normal liver, or one affected by steatosis or glycogenosis. This tumor is usually well-defined but rarely encapsulated, highly vascular, variable in size, and solitary or multifocal. The presence of multifocal nodules has been defined as adenomatosis, (3).
HCA molecular classification, (4):
Based on genomic analysis, three main molecular subtypes of HCA have been clearly identified so far, with a fourth class presently uncharacterized.
1. HCA inactivated for HNF-1a (H-HCA), accounting for 30 to 40% of HCA.H-HCA are defined by inactivation of HNF-1a, a transcription factor involved in hepatocyte differentiation and metabolism control. Morphologically, H-HCAs are characterized by prominent steatosis.
2. Inflammatory adenomas (I-HCA), accounting for 40 to 55% of HCA. I-HCAs represent a heterogeneous subgroup of HCA regarding the variety of gene mutations, I-HCA are more often observed in patients with obesity and/or metabolic syndrome, as well as in the context of a high alcohol consumption. Systemic inflammatory syndrome, demonstrated by increased serum C-reactive protein (CRP) and fibrinogen levels, can regress following HCA removal. Morphologically, I-HCA, initially described as ‘‘telangiectatic form of FNH”, are characterized by the presence of clusters of small arteries surrounded by extracellular matrix and inflammatory infiltrates associated with foci of sinusoidal dilatation.
3. β-catenin activated HCA (β-HCA), accounting for 10 to 20% of HCA. β-HCA are defined by β-catenin activation within the tumours. β-HCA are over represented in males and display a higher risk of malignant transformation towards hepatocellular carcinoma (HCC). Morphologically, β-HCAs are characterized by the presence of cellular atypias, pseudoglandular formations and cholestasis.
4. Unclassified HCA, accounting for 5% to 10% of HCA. A small subset of HCA do not display any specific morphological features nor do they have any of the gene mutations previously described.
Hepatic cysts:
Hepatic cysts are thin-walled structures lined by cuboidal bile duct epithelium and filled with isotonic fluid. They are the result of ductal plate malformation without communication with the biliary tree, (4).
- Introduction
Benign liver lesions are a heterogeneous group of lesions with different cellular origins, as summarized by an international panel of experts sponsored by the World Congress of Gastroenterology in 1994,(5). Some of these lesions are of greater clinical relevance than others, and the aim of these recommendations is to
provide a contemporary aid for the practical diagnosis and management of the more common benign lesions. These include haemangiomas, focal nodular hyperplasia (FNH), hepatocellular adenoma (HCA) and hepatocystic disease including poly-cystic liver, (4 & 6).
There is no justification to recommend histopathology of liver lesions or molecular subtyping of HCA as routine clinical practice. As evidence accumulates and methodologies improve with respect to risk and sensitivity, this may change. (4).
- Scope and purpose
The scope of this guideline is to set recommendations for the diagnosis and treatment of benign liver lesions. The main purpose of these guidelines is to minimize malpractice and poor surgical decision, to improve the quality of medical care and surgical service, to provide the good surgical practice to our patients, and finally to be cost effective.
- Target audience
The principle targeted candidates are the practicing surgeons, however endoscopists and radiologists and all specialists involved in the treatment of benign liver lesions are also included.
- Methods
A comprehensive search for guidelines was undertaken to identify the relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting only national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated in 2016 and later)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in:
• Developing organization/authors
• Date of publication, posting, and release
• Country/language of publication
• Date of posting and/or release
• Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point (any guideline scoring above 50% on the rigor dimension was retained). The GDG decided to adapt the ACG Clinical Guideline: Focal Liver Lesions, 2024, EASL Clinical Practice Guidelines on the management of benign liver tumours, 2016 and Benign liver lesions: Guideline for clinical practice, 2022
➡️Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
· GRADE working group: https://www.gradeworkinggroup.org
· GRADE online training modules: http://cebgrade.mcmaster.ca/
Specifically, the quality of evidence was graded as ‘High’, ‘Moderate’, ‘Low’ or ‘Very low’, (table 1& 2).
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
these are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Research needs
1. Role of FNA in diagnosis of focal hepatic lesions.
2. Management of multiple hepatic lesions.
3. Application of artificial intelligence in radiological diagnosis of hepatic lesions.
- Clinical indicators for monitoring
1. Hepatic imaging, (ultrasonography, CT, MRI).
2. Documentation of management of solid hepatic lesion.
3. Documentation of management of cystic hepatic lesion.
- Updating of the guideline
The GDG committee for guidelines development is responsible for the continuous evaluation of evidence available about benign liver lesions. The present guidelines will be updated in case of significant changes based on new evidence.
- Annexes
Table 1: Quality and Significance of the Four Levels of Evidence in GRADE

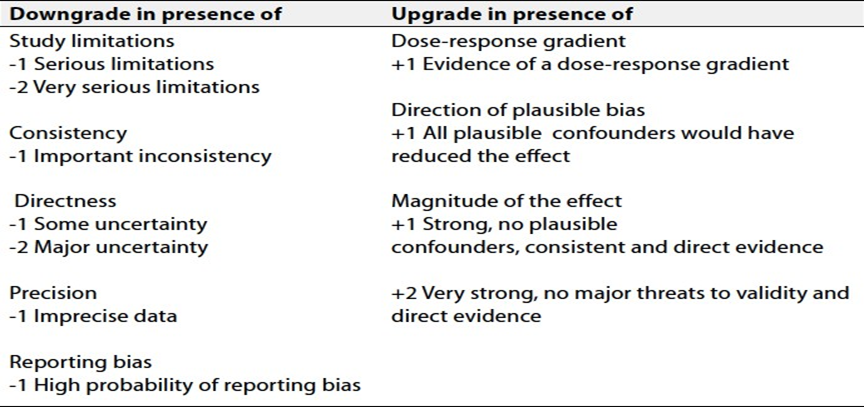
Table 2: Factors that Determine How to Upgrade or Downgrade the Quality of Evidence
- References
1. Klotz T, Montoriol PF, Da Ines D, et al. Hepatic haemangioma: Common and uncommon imaging features. Diagn Interv Imaging 2013; 94(9):849–59.
2. Libbrecht L, Cassiman D, Verslype C, et al. Clinicopathological features of focal nodular hyperplasia-like nodules in 130 cirrhotic explant livers. Am J Gastroenterol 2006;101(10):2341–6.
3. Barbier L, Nault JC, Dujardin F, et al. Natural history of liver adenomatosis: A long-term observational study. J Hepatol 2019;71(6): 1184–92.
4. 4.Colombo M, Forner A, Ijzermans J, Paradis V, Reeves H, Vilgrain V, et al. EASL Clinical Practice Guidelines on the management of benign liver tumours. J Hepatol. 2016 Aug 1;65(2):386–98.
5. Wanless IR, Party IW. Terminology of nodular hepatocellular lesions. Hepatology [Internet]. 1995;22(3). Available from: https://journals.lww.com/hep/fulltext/1995/09000/terminology of nodular hepatocellular_lesions.40.aspx
6. Frenette C, Mendiratta-Lala M, Salgia R, Wong RJ, Sauer BG, Pillai A. ACG Clinical Guideline: Focal Liver Lesions. American Journal of Gastroenterology. 2024 Jul 1;119(7):1235–71.
7. Pompili M, Ardito F, Brunetti E, Cabibbo G, Calliada F, Cillo U, et al. Benign liver lesions 2022: Guideline for clinical practice of Associazione Italiana Studio del Fegato (AISF), Società Italiana di Radiologia Medica e Interventistica (SIRM), Società Italiana di Chirurgia (SIC), Società Italiana di Ultrasonologia in Medicina e Biologia (SIUMB), Associazione Italiana di Chirurgia Epatobilio-Pancreatica (AICEP), Società Italiana Trapianti d’Organo (SITO), Società Italiana di Anatomia Patologica e Citologia Diagnostica (SIAPEC-IAP) – Part II - Solid lesions. Digestive and Liver Disease. 2022 Dec 1;54(12):1614–22.