Branchiomycosis (Gill Rot Disease)
| الموقع: | EHC | Egyptian Health Council |
| المقرر الدراسي: | Aquatic animal medicine Guidelines |
| كتاب: | Branchiomycosis (Gill Rot Disease) |
| طبع بواسطة: | Guest user |
| التاريخ: | الأربعاء، 6 مايو 2026، 3:45 AM |
الوصف
"last update: 22 Jun 2026" Download Guideline
- Acknowledgment
We would like to acknowledge the committee of National Egyptian Guidelines for Veterinary Medical Interventions, Egyptian Health Council for adapting this guideline.
Executive Chief of the Egyptian Health Council: Prof. Mohamed Mustafa Lotief.
Head of the Committee: Prof. Ahmed M Byomi
The rapporteur of the Committee: Prof. Mohamed Mohamedy Ghanem.
Scientific Group Members: Prof. Gamal A. Sosa, Prof. Nabil Yassien, Prof. Ashraf Aldesoky Shamaa, Prof. Amany Abbass, Prof. Dalia Mansour, Dr. Essam Elmarakby, Dr. Mohamed Elsharkawy , Dr. Naglaa Radwan, Dr. Hend El Sheikh
Authors: Mohamed Faisal; Adel A. Shaheen, Amany A. Abbass; Amel M. El Asely1; Eman A. Abd El-Gawad; Hiam S. Elabd; Aya F. Matter; Hadeer A. Youssef, and Amira M. El-Daim.
- Scope
The disease is generally known as a bad-management disease since it flourishes up in poor water quality with high levels of organic matter. It is an acute, subacute and chronic mycotic disease that affects cultured fresh and brackish water fish during summer. It is characterized by gill degenerative changes (marbling appearance), gill necrosis (gill rot) and high morbidity (100%) with a 50-65% mortality rate.
- Summary
This disease is characterized by a marbling appearance in the gills either due to intravascular growth of Branchiomyces sanguinis (B. sanguinis) inside the gill blood vessels or constriction of blood vessels in the case of Branchiomyces demingrans (B. demingrans). Histologically, gills showed hyperplasia, fusion of gill lamellae, and areas of infarctive necrosis resulting from thrombosis of vessels by fungal hyphae. The prognosis of this disease is bad due to its sudden occurrence and the rapid course of mortality that reaches up to 60% within 48 hrs. The infected fish suffer mainly from respiratory distress. The disease may be predisposed by bad water quality, high water temperatures and high load of organic matters. Preventative measures are much more effective than treatment which could be achieved by strict hygienic measures, water quality improvement, removal of dead and dying fish, and avoidance of overfeeding, especially at high water temperatures.
- Introduction
Branchiomycosis is globally distributed in regions where summer water temperatures are above 20°C. In Egypt, Branchiomyces spp. was isolated from naturally infected Nile tilapia with a prevalence of 92% from cultured fish in Edco farms and revealed high mortality (Khalil et al., 2015, Mahboub and Shaheen, 2021). Branchiomycosis most causes respiratory manifestations, lethargy, and high mortalities (Sen and Mandal, 2018). Gills necrosis of infected fish renders the fish to be susceptible to predation and other infections from opportunistic pathogens and mortality.
- Etiological agents
o Branchiomycosis is caused by two species, B. sanguinis and B. demingrans. They are currently in the Kingdom Chromista, a diverse group that includes diatoms, brown algae, and other water molds including Saprolegnia spp.
o The species B. sanguinis and B. demingrans have been differentiated based on differences in hyphae and spore diameter and the location of hyphae within fish gills. The B. sanguinus species has hyphae that are 8-30 μm in diameter, and spores that are 5-9 μm in diameter, and it is found only in the blood vessels of the gills. The other species, B. demingrans, is found in gill tissues outside of blood vessels and its spores are larger in diameter (12-17 μm) (Goodwin, 2012).
o Branchiomyces spp has been reported in a broad taxonomic range of fish species. Given this broad host range, it is likely that many more species of freshwater cultured fish only are susceptible to branchiomycosis (Goodwin, 2012).
o Poor-quality water at the sites of infection (environmental stress including ammonia, nitrite and organic matter) could be a major predisposing cause of this fungal infection. As expected, water reuse systems (Closed systems) are more susceptible to its infection than other culture systems (El-sayed, 2020).
o The fungus invades the gill via branchial vessels or the epithelium and can cause massive destruction of respiratory surfaces (El-Sayed, 2020). The invasive fungal hyphae obstruct the circulation of the blood through the gills. Outbreaks appear may be unpredictable although mortalities up to 50% have been recorded.
- Diagnosis
o Common Clinical signs
The infected fish suffered weakness, lethargy and respiratory distress resulting from gill tissue damage, represented by swimming in a vertical position to gasp the air, surfacing, and accumulating at the water inlet with rapid operculum movement and eventually, dying with an open mouth. Primarily, appeared congested in the early stage and then showed the beginning of tissue paleness due to loss of oxygenation. The color of the gills began to turn white because of necrosis, and eventually, bright, white-colored gills in advanced cases of infection. The disease progressed as a result of severe necrosis of gill tissue which gave gills the marbling appearance (Mahboub and Shaheen, 2021) (Fig.1; A, B, C).

Fig. 1: A) Gills showed severe congestion in the acute course. B) Gills showed a marbling appearance in the acute course. C) Gills look bright white colored in advanced cases of infection with severe necrosis.
- Lab Diagnosis
Samples are taken from infected gills.
· Presumptive diagnosis
o Isolation, Identification
Branchiomyces spp. could grow on Sabouraud dextrose agar (SDA) media (with duck extract decoction 10%+ citric acid 10%); the primary growth was observed from 2 to 3 days post-incubation at room temperature characterized by whitish raised hairy growth. Microscopically, through a wet mount preparation from the culture growth, the hyphae appeared broad, non-septated, intermingled with each other, and branched at their tips (Fig, 2; A, B, C, D, E).

Fig.2: A) primary growth of Branchiomyces sanguinis growth on SDA media observed from 2 to 3 days post-incubation. B) Typical growth of colonies was noticed from 7 to 9 days showing creamy velvety heaped at the center with hairy in the periphery and firmly attached to the media. C) Wet mount preparation from the culture growth, the hyphae appeared broad, non-septated, intermingled with each other, and branched at their tips. D) A portion of the colony stained with lactophenol cotton blue (LPCB) reveals thick hyphae that are branching at their tips (arrowhead) (Mahboub and Shaheen, 2021). Showed E) Thick hyphae with opalescence organelles (Shaheen unpublished data).
· Histopathological examination
o Gills specimens were examined for histopathological changes. The specimen was rapidly fixed in 10% natural formalin buffered phosphate for 24 hours, washed under running tap water and dehydrated through ascending grads of ethanol. It was cleaned in chloroform and embedded in paraffin wax at 60°C. The sections of 5 μm thickness were stained by Hematoxylin and eosin stain (Eyarefe et al., 2008; Adeshina et al., 2019). Using special stains for fungi (periodic acid Schiff’s (PAS) or silver stains), oomycete hyphae and spores can be easily seen in the gill vasculature and in extravascular gill tissues, (Fig.3, A, B).
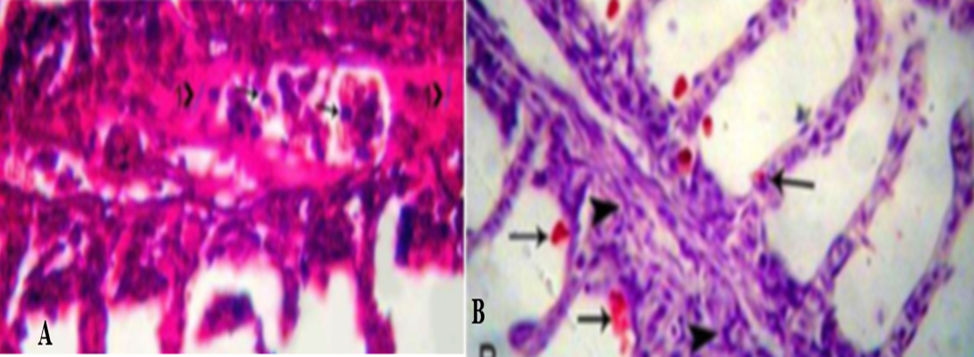
Fig.3: A) Periodic Acid-Schiff -stained paraffin section, s pores (arrowhead) and hyphae (head 1) stain purple and present inside the gill vessels. Also, some secondary lamellae show shortening, others reveal desquamation in the epithelium (head 2). B) H&E-stained section of gills Infected with B. demingrans reveal bright red spores (arrowhead) locate in-between the gill filaments, with aggregation of macrophages, lymphocytes and fibroblasts inside the gill vessels (head) (Mahboub and Shaheen, 2021).
· Confirmatory Diagnosis
· Molecular diagnosis
PCR tests for detection of DNA in samples (a pure isolate of Branchiomycosis sp. On SDA with 10% duck excrement decoction) were performed using two universal primers to ensure that Branchiomyces sp. is related to fungi.
· Control
o Prevention measures
- Quarantine for the newly introduced fish
- Proper drainage, drying, and disinfectant of the pond (Perfect disinfections)
- Increasing water exchange in the Pond.
- Feed well balanced ration.
- Controlling the addition of fertilizers(chemical).
- Addition of copper sulphate in four treatments 2-3 kg/hectare, at monthly intervals starting in mid-May and ending in mid-August.
- Addition of CaO (quick lime) 150-200 kg/hectare, at 2-week intervals during the summer.
o Control measures and treatment
- The dead and dying fish must be removed and burned in a lime pit (burying and burning).
- Avoid moving infected fish to prevent spreading of infection.
- Feeding fish should be stopped.
- Increase the pond water exchange to ensure maximum oxygenation.
- Addition of copper sulfate 2-3 kg/hectare, at monthly intervals starting in mid-May and ending in mid-August (0.3 mg/L),
- Using CaO (quick lime) 150-200 kg/hectare daily and the pH should not exceed 9.0
- The ponds must be drained, dried and disinfected with quick lime.
· Zoonotic importance
No zoonotic importance
- References
1. Andrew Goodwin., 2012. Branchiomycosis. University of Arkansas at Pine Bluff Aquaculture/Fisheries Center.
2. Bocci, V., 2005. Ozone Anew Medical Drug. Springer, Netherlands.
3. El-Bouhy, Z.M; Shaheen, A.A; Hassanin, M.E and Mahboub, H.H (2014): Branchiomycosis in Nile tilapia (Oreochromis niloticus) in Behiera Governorate with trials for treatment. Zag. Vet. J. (ISSN.1110-148) Vol.42, No.3.pp.29-42.
4. El-sayed A.M., 2020. Tilapia culture. Book second edition 2020.
5. Ibrahim, K.S., 2011. Isolation and pathological study of branchiomycosis from the commercial pond of common carp (Cyprinus carpio) fish, in Governorate of Duhok/Iraq. Iraqi J. Vet. Med. 35 (1), 1–9
6. Khalil RH, Saad TT, Selema TA and Abdel-Latif HM. 2015. Branchiomyces demigrans infection in farm-reared Common Carp (Cyprinus carpio L.) and Nile Tilapia (Oreochromis niloticus) at different localities in Egypt, with special emphasis to the role of environmental stress factors. Int. J. Innovative Stud. Aquat. Biol. Fish., 1: 15-23.
7. Mahboub HH and Shaheen AA. 2021. Mycological and histopathological identification of potential fish pathogens in Nile tilapia. Aquaculture 530.
8. Melaku, H., Lakew, M., Alemayehu, E., Wubie, A., 2017. Isolation and identification of pathogenic fungus from African Catfish (Clarias gariepinus) eggs and adults in National Fishery and Aquatic Life Research Center Hatchery, Ethiopia. FAJ 8 (3), 1–5.https://doi.org/10.4172/2150-3508.1000213.
9. Noga, E.J. (2010): Fish Disease Diagnosis and Treatment. Mosby. 'New York. pp. 367.
10. Patel, A.S., Patel, S.J., Bariya, A.R., Pata, B.A., Ghodasara, S.N., 2018. Fungal diseases of fish: a review. J. Vet. Sci. Res. 3 (3) (ISSN: 2474-9222).
11. Ramaiah, N., 2006. A review on fungal diseases of algae, marine fishes, shrimps and corals. Indian J. Mar. Sci. 35 (4), 380–387.
12. Sen, K., and Mandal, R., 2018. Fresh-water fish diseases in west Bengal, India. IJFAS 6 (5), 356–362.
13. Shawer R, Gomaa S, Saleh W, Soliman MK, Khalil R and Zaki MS. 2011. Some studies on fish deformity in freshwater fish in Egypt. Life Science Journal; 8(3): 415-422.