THE MANAGEMENT OF MASSIVE BLEEDING IN POLYTRAUMA PATIENTS
| Site: | EHC | Egyptian Health Council |
| Course: | Emergency Medicine Guidelines |
| Book: | THE MANAGEMENT OF MASSIVE BLEEDING IN POLYTRAUMA PATIENTS |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:52 AM |
Description
"last update: 9 Feb 2026" Download Guideline
- Executive Summary
This guideline provides standardized, evidence‑based recommendations for the recognition and management of massive bleeding in trauma patients.
Key Principles
- Early Recognition & Immediate Control: Rapid identification of haemorrhage and prompt surgical or interventional bleeding control is essential.
- Damage Control Resuscitation (DCR): Adopted globally over the past decade, DCR emphasizes:
- Permissive hypotension until bleeding is controlled.
- Balanced blood‑product transfusion (plasma, platelets, red cells).
- Restricted crystalloid use to avoid dilutional coagulopathy.
- Early correction of coagulopathy.
- Multidisciplinary Approach: Integration of emergency medicine, surgery, anaesthesia, and intensive care teams to deliver coordinated haemostatic resuscitation tailored to patient physiology.
1. Minimize Time to Bleeding Control
We recommend that the time between injury and bleeding control be minimized. (Strong recommendation)
2. Local Compression of Open Wounds
3. Tourniquet Use for Extremity Injuries
4. Follow the C-ABCDE Approach
5. Clinical Assessment of Traumatic Haemorrhage
6. Use of Shock Index (SI) and narrowed pulse pressure (PP)
We recommend that the Shock Index (SI) and/or Pulse Pressure (PP) be used to assess the degree of hypovolemic shock and transfusion requirements. (Strong recommendation)
7. Immediate Bleeding Control
We recommend that patients with an obvious bleeding source and those presenting with haemorrhagic shock in extremities and a suspected source of bleeding undergo an immediate bleeding control procedure, if not available transfer patient to the nearest appropriate facility after stabilization. (Strong recommendation)
8. Investigation of Unidentified Bleeding Source
We recommend
that patients with an unidentified source of bleeding should undergo immediate
further investigation to determine the bleeding source.
(Strong recommendation)
9. Use of Point-of-Care Ultrasonography (POCUS)
We suggest the use of point-of-care ultrasonography (POCUS), including eFAST, in patients with thoracoabdominal injuries if feasible (Conditional recommendation)
10. Early Whole-Body CT (WBCT)
We suggest early imaging using contrast-enhanced whole-body CT (WBCT) for detection and identification of injury type and bleeding source after patient stabilization, if available. (Conditional recommendation)
11. Repeated Haemoglobin/Haematocrit Monitoring
during resuscitation, we recommend repeating Hb and/or Hct measurements within 30 – 60 minutes, as initial normal values may mask early bleeding. (Strong recommendation)
12. Lactate and Base Deficit Monitoring
We suggest measurement of blood lactate as a sensitive test to estimate and monitor the extent of bleeding and tissue hypoperfusion; In the absence of lactate measurements, base deficit may represent a suitable alternative. If available. (Conditional recommendation)
13. Monitoring of Haemostasis
We recommend the early and repeated monitoring of haemostasis, using an international normalised ratio (INR), and platelet count. (Strong recommendation)
14. Restricted Volume Replacement and Blood Pressure Targets
We recommend the use of a restricted volume replacement strategy in the absence of clinical evidence of brain injury with a target systolic blood pressure of 80–90 mmHg (mean arterial pressure 50–60 mmHg) In the initial phase following trauma, until major bleeding has been stopped. (Strong recommendation)
15. In patients with severe TBI (GCS ≤ 8)
we recommend maintaining mean arterial pressure ≥ 80 mmHg. (Strong recommendation)
16. Use of Noradrenaline When Restricted Volume Replacement Fails
we recommend the administration of noradrenaline to maintain target arterial blood pressure, if a restricted volume replacement strategy does not achieve the target blood pressure. (Strong recommendation)
17. Choice of Crystalloid Solutions
18. Avoidance of Hypotonic Solutions in TBI
19. Restriction of Colloid Use
20. Target Haemoglobin After Bleeding Control
We recommend a target haemoglobin of 7–9 g/dL after controlling the source of bleeding. (Strong recommendation)
21. Prevention and Management of Hypothermia
We recommend early application of measures such covering the patient and warm fluids to reduce heat loss and warm the hypothermic patient to achieve and maintain normothermia. (Strong recommendation)
22. Damage Control Surgery
We suggest damage control surgery in the severely injured patient if the definitive surgery to control the source of bleeding is complicated and time-consuming (>90 minutes) in the presence of severe persistent coagulopathy, severe acidosis with base deficit >15 mmol/L or lactate >5 mmol/L, hypothermia <34°C, or signs of ongoing bleeding despite the initial attempts of bleeding control with systolic BP persistently <90 mmHg. (Conditional recommendation)
23. Pelvic Binder Use
We recommend the adjunct use of a pelvic binder or pelvic sheet to limit life threatening bleeding in the presence of a suspected pelvic fracture. (Strong recommendation)
24. Topical Haemostatic Agents
We suggest the use of topical haemostatic agents with packing for venous or moderate arterial bleeding associated with parenchymal injuries. (Conditional recommendation)
25. Tranexamic Acid (TXA)
We recommend TXA
administration in trauma patients who are bleeding or at risk of significant
bleeding, as soon as possible and within 3 hours of injury at a loading dose of
1 g IV over 10 min, followed by 1 g IV infusion over 8 h.
(Strong recommendation)
26. Balanced Blood Product Transfusion
We recommend transfusion of pRBCs: FFP: Platelets in ratio following the massive transfusion protocols, In the initial management of patients with suspected massive haemorrhage. (Strong recommendation)
27. Calcium Monitoring and Supplementation
We recommend that ionised calcium levels be monitored and maintained within the normal range following major trauma and especially during massive transfusion. We recommend the administration of calcium to correct hypocalcaemia. (Strong recommendation)
28. Reversal of Vitamin K Antagonists (VKA)
We recommend the emergency reversal of vitamin K-dependent oral anticoagulants in the bleeding trauma patient with the early use of 5–10 mg I.V. phytomenadione (vitamin K1) in addition to FFP. (Strong recommendation)- Recommendations
1. Minimize Time to Bleeding Control
We recommend that the time between injury and bleeding control be minimized.
Strength of recommendation: Strong
Level of evidence: Moderate
Rationale:
Trauma
Systems and the Chronology of Care
Regionalization and Trauma System Maturity
The implementation of regionalized trauma systems has fundamentally improved outcomes for severely injured patients globally. These systems utilize a tiered network of designated trauma centers (Levels I through IV) that collaborate with pre-hospital Emergency Medical Services (EMS). A meta-analysis of over 1.1 million patients confirmed that the establishment of such systems significantly reduces mortality, with survival rates continuing to climb as these systems mature and integrate quality improvement programs and trauma registries [8].
Evidence from multicenter cohort studies indicates that patients in hemorrhagic shock (SBP < 90 mmHg) have significantly higher survival rates when treated at Level I Trauma Centers compared to Level III or IV facilities [9].
This underscores the necessity of a systemized approach that matches patient acuity to the appropriate facility based on vital status, injury patterns, and available hospital resources.
The Impact of Time on Survival
Uncontrolled hemorrhage is the leading cause of potentially preventable trauma deaths, with data suggesting that approximately 34.5% of early hemorrhagic fatalities could be avoided through more rapid bleeding control [10].
Pre-Hospital Time Factors
Time lost during the pre-hospital phase is directly linked to increased mortality. Extensive analyses of EMS data demonstrate that:
- Penetrating Trauma: Every additional minute of response time correlates with a 2% increase in mortality, while every extra minute spent on the scene correlates with a 1% increase [11, 12].
- Hemodynamic Instability: For unstable patients, particularly those with penetrating injuries, "scoop and run" strategies (minimizing scene time) are significantly more beneficial than prolonged on-scene stabilization [13].
- Functional Outcomes: Even in hemodynamically stable patients where mortality might not be immediately affected by time, longer pre-hospital durations are associated with an increased risk of poor long-term functional recovery [14].
In-Hospital Delay: "Door-to-Needle"
The mandate to minimize time-to-intervention extends beyond the hospital doors. For patients with ongoing hemorrhage, "door-to-intervention" time—whether for surgery or angioembolization is a critical survival factor [15, 16].
Delays in initiating these procedures are associated with worsened outcomes, reinforcing that both swift pre-hospital transport and streamlined in-hospital trauma protocols are essential to patient salvage.
2. Local Compression of Open Wounds
We recommend local compression of open wounds to limit life-threatening bleeding.
Strength of recommendation: Strong
Level of evidence: Moderate
Rationale:
Local Haemorrhage Control Techniques
In civilian trauma, most life-threatening hemorrhages resulting from open extremity injuries can be managed effectively through local compression. This is typically achieved via direct manual pressure or the application of specialized pressure dressings to the site of injury [17].
Additional compression to the source of bleeding can also be achieved for some penetrating injuries by Foley catheter inserting directly into the wound, initially described in bleeding penetrating neck injuries. [18].
Furthermore, the efficacy of external hemorrhage control in pre-hospital environments is significantly enhanced using compression bandages impregnated with or used in conjunction with topical hemostatic agents [19].
3. Tourniquet Use for Extremity Injuries
Strength of recommendation: Strong
Level of evidence: Moderate
Rationale:
Rationale for Tourniquet Application
In cases of catastrophic extremity trauma, such as traumatic amputations or deep penetrating injuries, manual direct pressure may be insufficient to achieve definitive hemostasis. For these life-threatening scenarios, the application of a tourniquet is a critical, life-saving intervention [19].
Originally validated in military medicine where it has saved numerous lives, the efficacy of tourniquets is now strongly supported by civilian data demonstrating a significant decrease in hemorrhage-related mortality [19–22].
While potential complications—such as nerve injury or localized tissue damage have been documented, these risks are statistically rare and are heavily outweighed by the immediate necessity of preventing exsanguination [23–25].
A tourniquet acts as a vital bridge, controlling the bleeding just long enough to get the patient to definitive surgical care."[19].
4. Follow the C-ABCDE Approach
We recommend Following the C-ABCDE approach in the management of polytraumatized patients with massive bleeding.
Strength of
recommendation: Strong
Level of evidence: Low
Rationale:
The <C>ABCDE Paradigm
In the acute management of severe trauma, the most immediate threat to patient survival is exsanguination from catastrophic hemorrhage, which can lead to death within minutes. Consequently, the traditional "ABCDE" resuscitation sequence has been refined to the "<C>ABCDE" framework [26].
By prioritizing "<C>" (Catastrophic External Hemorrhage) at the start of the primary survey, clinicians are directed to achieve definitive bleeding control even before addressing airway management. This fundamental shift in trauma priority ensures that the most time-critical physiological threat is mitigated immediately, thereby enhancing the efficacy of subsequent resuscitation and stabilization efforts [26, 27].
5. Clinical Assessment of Traumatic Haemorrhage
We recommend that the physician should clinically assess the extent of traumatic haemorrhage using a combination of patient vital signs, anatomical injury pattern, mechanism of injury and the patient response to initial resuscitation.
Strength of
recommendation: Strong
Level of evidence: Low
Rationale:
Assessment
and Prediction of Traumatic Haemorrhage
Limitations of Traditional Assessment and Scoring Systems
Although the current ATLS classification system for hypovolemic shock incorporates physiological markers like base excess to estimate blood loss and transfusion requirements, its precision remains a subject of clinical debate [26, 27].
While numerous predictive models and clinical scores have been engineered to forecast hemorrhage, traumatic coagulopathy, or the necessity for Massive Transfusion (MT), their performance is inconsistent. Reported Area Under the Receiver Operating Characteristics (AUROC) values range widely from 0.73 to 0.95, and a lack of robust prospective validation has prevented any single tool from achieving universal clinical adoption [28].
A meta-analysis of 84 studies identified 35 distinct predictive variables, with systolic blood pressure (SBP), age, heart rate, and injury mechanism being the most frequently scrutinized [28].
However, methodological rigor varies; of the 47 multivariate models analyzed, only 21 adhered to the standard of 10 events per predictor. The most reliable variables identified across multiple models include:
- Mechanism of injury
- Systolic Blood Pressure (SBP) and Heart Rate
- Haemoglobin and Lactate levels
- Focused Assessment with Sonography in Trauma (FAST) findings [28].
The Role of Injury Mechanism and Critical Haemorrhage
Evaluating the mechanism of trauma is essential for accurate risk stratification. A fall exceeding the "critical height" of 6 meters (20 feet) is strongly correlated with major internal injuries and significant blood loss [29].
Furthermore, patients who are physically trapped following an incident often present with time-critical injuries that demand immediate intervention due to the high probability of occult hemorrhage [30].
Other high-risk mechanisms include high-energy deceleration impacts and wounds from high-velocity projectiles. Notably, modern clinical shifts toward low-volume resuscitation and permissive hypotension have changed how practitioners interpret a patient’s physiological response to fluid challenges, making traditional assessment even more complex.
6. Use of Shock Index (SI) and narrowed
pulse pressure (PP)
We recommend that the Shock Index (SI) and/or Pulse Pressure (PP) be used to
assess the degree of hypovolemic shock and transfusion requirements.
Strength of recommendation: Strong
Level of evidence: Low
Rationale:
Predictive
Value of Physiological Ratios
Shock Index (SI)
The Shock Index (SI), defined as the ratio of heart rate to systolic blood pressure, serves as a robust predictor of occult hemorrhage. While a normal SI in healthy adults it ranges from 0.5 to 0.7, retrospective data indicates that values between 0.9 and 1.0 are associated with a 25% requirement for Massive Transfusion (MT), a 14.7% need for surgical intervention, and a 6.2% requirement for interventional radiology [31].
Across various retrospective studies, thresholds between 0.8 and 1.0 have consistently predicted the need for MT, yielding AUROC values between 0.73 and 0.89 [32–35].
Prospective analysis of 1,402 patients suggested that an SI of 0.8 provides higher sensitivity than 0.9 [32].
Specifically, a cut-off of 0.81 demonstrated 85% sensitivity and a 98% negative predictive value for MT [36], while a threshold of 0.91 achieved 81% sensitivity and 87% specificity [35].
Even after adjusting for age, sex, and injury severity (ISS/GCS), the SI remains an independent predictor for mortality and transfusion requirements (OR 3.57) [36].
Notably, an SI of 1.0 has been shown to outperform the ABC score in predicting MT and is more effective than simple hypotension in identifying patients requiring emergency operative intervention [33, 37].
Pulse Pressure (PP)
A narrowed Pulse Pressure (PP) the difference between systolic and diastolic blood pressure is a recognized hallmark of Class II hemorrhage according to ATLS guidelines. Current literature defines a narrow PP as less than 40 mmHg, or in some clinical contexts, less than 30 mmHg.
Evidence confirms that a narrowed PP is independently linked to an increased likelihood of requiring blood transfusions, resuscitative thoracotomy, and emergent surgical control of bleeding [38–40].
Multivariate analyses further substantiate this, revealing that a PP below 30 mmHg is a significant predictor for both MT (OR 3.74) and the necessity for immediate operative intervention [41].
7. Immediate Bleeding Control
We recommend that patients with an obvious bleeding source and those presenting with haemorrhagic shock in extremities and a suspected source of bleeding undergo an immediate bleeding control procedure, if not available transfer patient to the nearest appropriate facility after stabilization.
Strength of recommendation: Strong
Level of evidence: Moderate
Rationale:
Rationale for Immediate Surgical Intervention
Patients presenting in profound hemorrhagic shock have typically sustained critical blood loss. In "agonal" patients where active hemorrhage persists, mortality is imminent unless the source of bleeding is controlled with extreme urgency.
Evidence from a study of 271 patients undergoing immediate laparotomy for gunshot wounds indicates that the combination of penetrating trauma and severe hypovolemic shock necessitates rapid, definitive surgical hemostasis [42].
Furthermore, a large-scale analysis of 16,113 trauma admissions identified a specific subset of 628 patients requiring "direct-to-operating-room" resuscitation. This study concluded that the most accurate predictors for the necessity of immediate surgical intervention include:
- Mechanism of Injury: Penetrating truncal trauma.
- Anatomical/Physical Findings: Traumatic amputations or other obvious major vascular disruptions.
- Physiological Derangement: Profound shock (systolic blood pressure < 90 mmHg) or a requirement for pre-hospital CPR [43].
Ultimately, for patients meeting these criteria, bypassing the traditional emergency department workup in favor of direct operative resuscitation optimizes outcomes when every minute is critical to survival [43].
8. Investigation of Unidentified Bleeding Source
We recommend that patients with an
unidentified source of bleeding should undergo immediate further investigation
to determine the bleeding source.
Strength of recommendation: Strong
Level of evidence: Low
Rationale:
Diagnostic Evaluation of the Stabilized Patient
For trauma patients who remain hemodynamically stable or achieve stabilization during initial resuscitation, a systematic diagnostic investigation is required to identify occult sources of hemorrhage. In cases where immediate surgical control is not indicated, the primary survey should be augmented with laboratory analysis—specifically blood gas and coagulation profiles and advanced imaging modalities, including ultrasonography and Computed Tomography (CT) [26, 44].
The Evolution of Trauma Imaging
In modern trauma care, the widespread accessibility of CT scanners has largely superseded traditional radiographic imaging for the definitive assessment of internal injuries [45].
Research indicates that the physical proximity of the CT scanner to the resuscitation bay significantly improves survival probabilities for severely injured patients by minimizing transport times and delays in diagnosis [46].
Whole-Body CT in Unstable Patients
When a CT scanner is located outside the emergency department, clinicians must carefully weigh the diagnostic benefits of imaging against the risks associated with transporting a critically ill patient. Continuous monitoring and ongoing resuscitation must be maintained throughout the transfer process. However, evidence suggests that within a well-structured environment and a highly organized trauma team, whole-body CT (WBCT) is both safe and clinically justified, even in the management of hemodynamically unstable patients [47].
9. Use of Point-of-Care Ultrasonography (POCUS)
We advise the use of point-of-care ultrasonography (POCUS), including eFAST, in patients with thoracoabdominal injuries IF FEASIABLE
Strength of
recommendation: Conditional
Level of evidence: Low
Rationale:
The Role
of Point-of-Care Ultrasound (POCUS)
Pre-hospital Ultrasound (PHUS)
The diagnostic accuracy of pre-hospital ultrasound (PHUS) is considered adequate for identifying pneumothorax, free intra-abdominal fluid, and hemoperitoneum. A systematic review involving 2,889 trauma patients demonstrated high sensitivity and specificity, with several studies noting that PHUS findings led to direct changes in clinical management [48].
While a more recent review of 3,317 patients confirmed the feasibility of PHUS and its impact on transport decisions, significant inconsistencies in protocols and outcome measures currently prevent a formal meta-analysis of its global efficacy [49].
In-Hospital POCUS and the FAST Protocol
In the hospital setting, the Focused Assessment with Sonography in Trauma (FAST) remains a cornerstone of the primary ATLS survey for detecting hemorrhage in the plural, pericardial, and peritoneal cavities [50].
While FAST exhibits high specificity (0.96), its sensitivity is variable (0.74), often depending on the patient population and the anatomical region affected [50]. Notably, a negative FAST examination cannot definitively exclude internal injury and must be validated against a reference standard, such as CT, especially in symptomatic patients.
This limitation is particularly critical in hemodynamically unstable patients. An analysis of the PROMMTT trial revealed that nearly 7% of hypotensive patients with a negative FAST still required a laparotomy within six hours of admission [51].
Consequently, in the presence of unexplained hypotension, significant intra-abdominal hemorrhage must be suspected regardless of ultrasound findings [51].
Advanced Techniques and Regional Accuracy
POCUS appears to yield higher diagnostic sensitivity for thoracic and cardiac injuries compared to abdominal assessments [50, 52, 53].
To improve the utility of ultrasound in trauma, several specialized techniques have been proposed:
- FAST-PLUS Protocol: Incorporating a transverse scan of the pubic symphysis can identify unstable pelvic fractures with high correlation to CT findings [54].
- Patient Positioning: Rolling a patient to the right lateral position during the exam may increase sensitivity by shifting fluid into more visible acoustic windows, potentially converting false-negative results into true positives [55].
10. Early Whole-Body CT (WBCT)
We suggest early
imaging using contrast-enhanced whole-body CT (WBCT) for detection and identification
of injury type and bleeding source after patient stabilization, if available.
Strength of recommendation: Conditional
Level of evidence: Moderate
Rationale:
Efficacy
of Whole-Body Computed Tomography (WBCT)
Diagnostic Accuracy and Clinical Utility
Observational and retrospective evidence consistently supports the use of Whole-Body Computed Tomography (WBCT) for its superior diagnostic accuracy, time-saving capabilities, and ability to localize hemorrhage sources rapidly [45, 56].
In multicenter studies, CT has demonstrated 100% sensitivity for identifying retroperitoneal hematomas and intra-abdominal injuries in patients presenting with the "seat belt sign" (abrasions or ecchymosis) [57, 58].
Evidence from the REACT-2 Trial
The REACT-2 trial, the only prospective randomized controlled trial (RCT) in this field, compared immediate WBCT to conventional selective CT. While the trial found no significant survival difference between the two groups for general polytrauma or traumatic brain injury (TBI) [59], a secondary analysis yielded critical insights for bleeding patients. For the subset of patients requiring emergency hemorrhage control, immediate WBCT was associated with an absolute risk reduction in mortality of 11.2% [60].
Time-Critical Outcomes
The implementation of WBCT significantly reduces the total time spent in the emergency department [61].
Furthermore, evidence suggests a direct correlation between the speed of imaging and survival; a median time of 19 minutes from hospital admission to CT has been significantly associated with a decrease in mortality caused by exsanguination [62].
Refined Clinical Criteria
Based on secondary data from the REACT-2 study, a set of 10 clinical criteria with high positive predictive value for severe injury has been developed to assist in identifying candidates for immediate WBCT [63].
However, practitioners should note that these criteria are derived from post hoc analyses and may not be universally applicable. A targeted diagnostic approach is often warranted, as hemodynamic instability can occasionally impair the sensitivity of contrast-enhanced CT in detecting active extravasation [64].
11. Repeated Haemoglobin/Haematocrit Monitoring
During
resuscitation, we recommend repeating Hb and/or Hct measurements within 30 – 60 minutes, as initial normal values
may mask early bleeding.
Strength of recommendation: Strong
Level of evidence: Moderate
Rationale:
Haemoglobin
and Haematocrit Monitoring
Limitations of Initial Measurements
In the acute phase of trauma, a single, baseline hemoglobin (Hb) measurement is often an unreliable indicator of the extent of blood loss. This diagnostic limitation stems from the physiological lag in fluid equilibration and the confounding effects of early crystalloid resuscitation, which may artificially dilute or temporarily mask the severity of the hemorrhage [66, 72].
Because initial Hb levels often remain within the normal range during the early stages of catastrophic bleeding, relying solely on admission values can lead to a dangerous underestimation of injury severity [73, 74].
Serial Monitoring and Emerging Technologies
Current evidence emphasizes the necessity of serial Hb or hematocrit (Hct) measurements to accurately track the progression of bleeding and the patient's response to resuscitation [67–69].
Clinical guidelines suggest repeating these tests every 30 to 60 minutes during the initial assessment phase, as significant downward trends in Hct even during active fluid administration are highly predictive of ongoing hemorrhage [70, 71].
Furthermore, recent advancements in non-invasive Hb monitoring have demonstrated high precision and close correlation with traditional laboratory-based results, offering a potential tool for continuous, real-time assessment in trauma settings [65].
Predictive Value in Trauma
Longitudinal changes in Hb and Hct levels are essential for identifying patients who require blood transfusions or urgent surgical intervention [68, 69]. These markers, when combined with other laboratory indicators such as base excess and fibrinogen levels, assist in forming a comprehensive picture of the patient's coagulation status and overall injury severity [72, 73].
Ultimately, the "stop the bleed" priority is best supported by frequent reassessment rather than a single static measurement [74].
12. Lactate and Base Deficit Monitoring
We recommend
measurement of blood lactate as a sensitive test to estimate and monitor the
extent of bleeding and tissue hypoperfusion; In the absence of lactate
measurements, base deficit may represent a suitable alternative. if available.
Strength of recommendation: Conditional
Level of evidence: Moderate
Rationale:
Lactate and Base Deficit as Diagnostic Tools
For the initial assessment of patients with traumatic hemorrhage, the measurement of serum lactate and base deficit is essential. These markers serve as highly sensitive indicators for estimating the severity of blood loss and monitoring the degree of tissue hypoperfusion [75, 76].
Unlike traditional vital signs, such as systolic blood pressure, which may remain compensated in the early stages of shock, lactate and base deficit provide an objective window into cellular dysfunction and the adequacy of oxygen delivery (DO_2) [75, 76].
Pathophysiological Rationale
The clinical utility of these markers is rooted in cellular metabolic shifts during hemorrhagic shock. When catastrophic bleeding reduces systemic oxygen delivery, tissues are forced to transition from aerobic to anaerobic metabolism to sustain energy production. This shift results in the accumulation of lactate, a metabolic byproduct that often signals critical hypoperfusion before the onset of overt clinical hypotension [76].
Lactate Clearance and Resuscitation Endpoints
In the context of trauma resuscitation, the primary objective is to reverse tissue debt and restore metabolic homeostasis. Consequently, "lactate clearance" the observed reduction of lactate levels over time—is considered a superior prognostic indicator and a more definitive endpoint for resuscitation than the mere normalization of arterial blood pressure [77].
Base Deficit and Metabolic Acidosis
Base deficit, derived from arterial blood gas (ABG) analysis, quantifies the concentration of base required to restore the blood to a physiological pH. It is a direct reflection of metabolic acidosis in trauma patients [78, 79].
As lactate a strong acid accumulates, it releases hydrogen ions (H+) into circulation. These ions are neutralized by the body’s bicarbonate (HCO3-) buffering system; the subsequent consumption of bicarbonate stores results in an increasingly negative base deficit, signaling worsening physiological status [80].
13. Monitoring of Haemostasis
We recommend the
early and repeated monitoring of haemostasis, using a traditional laboratory
determination such international normalised ratio (INR), and platelet count.
Strength of recommendation: Strong
Level of evidence: Low
Rationale:
Conventional
Coagulation Testing (CCT)
Prognostic Value and Mortality Correlation
Conventional coagulation tests (CCTs) including International Normalized Ratio (INR), Prothrombin Time (PT), activated Partial Thromboplastin Time (aPTT), platelet count, and fibrinogen concentration remain foundational in the early detection of trauma-induced coagulopathy. Because coagulopathy is a primary driver of mortality in trauma victims, the widespread availability and diagnostic reach of CCTs are critical [81].
Research indicates that even in patients with moderate injuries, abnormal CCT results upon admission are strongly associated with increased mortality [81].
Recent evidence suggests that CCTs may offer superior predictive power for mortality compared to viscoelastic hemostatic assays (VHAs), such as Rotational Thromboelastometry (ROTEM) or Thromboelastography (TEG). Specifically, a 2025 prospective study demonstrated that prolonged INR and clotting times are more reliable prognostic indicators for survival than VHA parameters alone [82].
Integration into Massive Transfusion Protocols (MTP)
CCTs are essential components of massive transfusion protocols, particularly in resource-constrained environments where advanced assays may not be available. While VHAs like ROTEM may exhibit higher sensitivity in diagnosing the specific nuances of trauma-induced coagulopathy (TIC), the clinical relevance of CCTs remains undisputed; patients presenting with abnormal CCT values experience significantly higher mortality rates [83].
Consequently, these tests remain a primary guide for the targeted administration of blood products during acute resuscitation [82, 83].
14. Restricted Volume Replacement and Blood Pressure Targets
We recommend the use of a restricted volume replacement strategy in the absence of clinical evidence of brain injury with a target systolic blood pressure of 80–90 mmHg (mean arterial pressure 50–60 mmHg) In the initial phase following trauma, until major bleeding has been stopped.
Strength of
recommendation: Strong
Level of evidence: Moderate
Rationale:
Permissive
Hypotension in Haemorrhagic Shock
Rationale and Mechanism
Permissive hypotension is a resuscitation strategy utilized in trauma patients with active bleeding, characterized by maintaining systolic blood pressure at sub-normal levels until definitive surgical or radiological hemorrhage control is secured. This is achieved through restricted volume replacement. The physiological objective of this approach is to prevent the "popping of the clot" the disruption of nascent hemostatic plugs which is a common complication of aggressive fluid resuscitation and high-pressure flow [84].
Clinical Evidence and the "Lethal Triad"
Conventional resuscitation models that prioritize the rapid restoration of normotension are frequently associated with the development of dilutional coagulopathy, hypothermia, and metabolic acidosis. These three conditions comprise the "lethal triad," which significantly drives mortality in severely injured patients [85].
Clinical evidence, most notably the landmark trial by Bickell et al., has demonstrated a distinct survival advantage in patients with penetrating truncal trauma when fluid resuscitation is limited or delayed until the point of surgical intervention [86].
Contraindications and Implementation
Although permissive hypotension is a cornerstone of contemporary trauma management, its application requires careful clinical judgment. This strategy is generally contraindicated in patients with concomitant traumatic brain injury (TBI), where maintaining cerebral perfusion pressure is paramount, and in scenarios involving prolonged transport times where the risk of protracted tissue ischemia may outweigh the benefits of restricted filling [84, 85].
15- In patients with severe TBI (GCS ≤ 8)
We recommend
maintaining mean arterial pressure ≥ 80 mmHg.
Strength of recommendation: Strong
Level of evidence: Low
Rationale:
Restrictive
Fluid Resuscitation and Permissive Hypotension
Clinical Evidence and Mortality Benefits
The contemporary management of trauma-induced hypotension has shifted from aggressive fluid administration toward restrictive volume replacement and permissive hypotension. This paradigm was largely established by a seminal randomized controlled trial (RCT) in the 1990s, which demonstrated improved survival in patients with penetrating injuries [86].
Recent meta-analyses of RCTs have since confirmed that in trauma patients without traumatic brain injury (TBI), restrictive fluid strategies significantly decrease mortality compared to traditional high-volume resuscitation [87, 88].
These findings are further corroborated by several meta-analyses of retrospective and mixed-methodology studies, which consistently show superior outcomes when normotension is not the immediate target [89–92].
Complications of Aggressive Resuscitation
Retrospective data indicate that aggressive resuscitation—often initiated in the pre-hospital setting is associated with a higher incidence of adverse outcomes. These include increased mortality, a greater necessity for damage control laparotomy, and the development of the "lethal triad" (coagulopathy, acidosis, and hypothermia). Furthermore, aggressive fluid protocols are linked to higher rates of multi-organ failure, nosocomial infections, increased transfusion requirements, and prolonged intensive care and hospital stays [93–95].
Recent evidence suggests these risks extend to pediatric populations, where higher initial crystalloid volumes are also associated with increased mortality [96].
Contraindications and Clinical Precautions
Despite its benefits, permissive hypotension is strictly contraindicated in patients with TBI or spinal cord injuries. In these cases, maintaining adequate mean arterial pressure is vital to ensure cerebral and spinal cord perfusion and oxygenation. The optimal balance between fluid administration and vasopressor use in these scenarios remains a subject of ongoing research, making rapid hemorrhage control the highest priority [97]. Additionally, this strategy should be applied with caution in elderly patients and those with a history of chronic arterial hypertension, as they may have a higher baseline requirement for organ perfusion.
Summary and Future Directions
Current literature supports a damage control resuscitation strategy that targets a reduced systolic blood pressure of 80–90 mmHg in the absence of CNS injury. However, the existing evidence base contains limitations, including small sample sizes in RCTs and potential selection bias in retrospective cohorts. While the shift toward restrictive volume replacement is clinically justified by the available data, further confirmation through adequately powered, high-quality prospective RCTs is necessary to refine these protocols.
16. Use of Noradrenaline When Restricted Volume Replacement Fails
We recommend the administration of noradrenaline
to maintain target arterial blood pressure, if a restricted volume replacement
strategy does not achieve the target blood pressure.
Strength of recommendation: Strong
Level of evidence: Low
Rationale:
Role of
Vasopressors and Inotropes in Haemorrhagic Shock
Noradrenaline and Early Resuscitation
The use of noradrenaline and other vasopressors during the acute phase of trauma remains controversial. Several retrospective analyses and systematic reviews have associated vasopressor use with increased mortality or a lack of clinical benefit [98–102, 104].
One study noted that mortality was not independently linked to vasopressors unless epinephrine was administered [103]. However, these findings are often limited by significant selection bias, as patients receiving vasopressors are typically more critically ill than those who are not [104].
Current evidence supports a strategy of restricted volume replacement and permissive hypotension (targeting a systolic blood pressure of 80–90 mmHg) without vasopressors in the initial stages of resuscitation. There is a theoretical concern that early vasopressor use may worsen organ ischemia by inducing excessive vasoconstriction. However, if these conservative measures fail and life-threatening hypotension persists (SBP < 80 mmHg), the transient use of noradrenaline is recommended to sustain tissue perfusion and prevent cardiovascular collapse.
Arginine Vasopressin (AVP)
Hemorrhagic shock involves a biphasic physiological response: an initial sympathoexcitatory phase followed by a sympathoinhibitory, vasodilatory phase that can severely reduce vascular tone [105]. Based on the hypothesis that profound shock leads to a deficiency in endogenous arginine vasopressin, clinical trials have investigated its supplementation.
A well-designed randomized controlled trial (RCT) involving 100 patients demonstrated that low-dose arginine vasopressin (4 IU bolus followed by 0.04 IU/min) significantly reduced the requirement for blood product transfusions [106]. These results align with earlier data showing that adding vasopressin to resuscitation fluids decreases the total volume of fluid required over five days without increasing adverse events or 30-day mortality [107]. While promising, further high-powered research is necessary to confirm the impact of AVP on long-term morbidity and mortality.
Managing Cardiac Dysfunction
Cardiac dysfunction in trauma patients may result from myocardial contusion, pericardial effusion, or secondary to increased intracranial pressure from brain injury. In these instances, inotropic agents such as dobutamine or epinephrine are indicated. In the absence of advanced hemodynamic monitoring, cardiac dysfunction should be suspected if a patient remains hypotensive despite adequate fluid expansion and noradrenaline administration.
17. Choice of Crystalloid Solutions
We recommend that fluid therapy using a 0.9% sodium chloride and/or ringer lactate or balanced crystalloid solution be initiated in the hypotensive bleeding trauma patient.
Strength of
recommendation: Strong
Level of evidence: Moderate
Rationale:
Crystalloid
Selection and Fluid Volume
Balanced Solutions vs. Normal Saline
While the use of crystalloids is fundamental to a restrictive fluid strategy in trauma, the optimal type of solution remains a subject of active debate. Historically, 0.9% sodium chloride (Normal Saline) was the standard; however, its use is increasingly scrutinized due to the risk of inducing hyperchloremic acidosis and acute kidney injury (AKI), both of which may negatively impact survival [108].
In contrast, balanced electrolyte solutions (such as Ringer’s Lactate or Plasma-Lyte) feature chloride concentrations closer to physiological levels. A large-scale RCT involving 15,802 patients suggested that balanced solutions reduce a composite outcome of death, new renal-replacement therapy, and persistent renal dysfunction [109].
However, subsequent high-quality evidence including the BaSICS trial and recent meta-analyses found no significant differences in 30-day mortality or hospital length of stay when comparing balanced solutions to saline in broader critically ill populations [110–112].
Clinical Recommendations
Despite the lack of a definitive survival benefit in all trials, current trauma guidelines favor balanced electrolyte solutions for initial management. If 0.9% sodium chloride is utilized, it should be restricted to a maximum volume of 1.0–1.5 L. Saline is specifically contraindicated in patients with severe metabolic acidosis, particularly when hyperchloremia is present.
Role of Colloids and Coagulation
The preference for crystalloids as the primary resuscitation fluid is also driven by the known inhibitory effects of synthetic colloids (such as hydroxyethyl starch and gelatin) on platelet function and the coagulation cascade.
Research indicates that while fibrinogen concentrates can partially mitigate this dilutional and functional coagulopathy, the effect depends heavily on the type and concentration of the colloid used [113, 114]. Therefore, colloid infusions should be reserved as a secondary option for cases of excessive hemorrhage where crystalloids and vasopressors are insufficient to maintain essential tissue perfusion.
18. Avoidance of Hypotonic Solutions in TBI
We recommend that hypotonic solutions be avoided during resuscitation of patients with haemorrhagic shock in addition to Traumatic brain injury.
Strength of
recommendation: Strong
Level of evidence: Moderate
Rationale:
Fluid Management in Traumatic Brain Injury (TBI)
In patients suffering from Traumatic Brain Injury, the selection of resuscitation fluids is restricted by the need to maintain cerebral osmotic pressure. Hypotonic solutions, including Ringer’s lactate and hypotonic albumin, should be strictly avoided. The administration of these fluids can lower serum osmolality, potentially driving a fluid shift into damaged cerebral tissues and exacerbating life-threatening cerebral edema [115].
This clinical caution is supported by secondary analysis from the PROMMTT study, which evaluated the impact of pre-hospital fluid choice. The data revealed that the use of Ringer’s lactate was associated with a higher adjusted mortality rate in the TBI population when compared to the use of isotonic normal saline [115].
19. Restriction of Colloid Use
We advise that
the use of colloids to be restricted due to the adverse effects on haemostasis.
Strength of recommendation: Conditional
Level of evidence: Low
Rationale:
Adverse
Effects of Colloids on Haemostasis
Mechanisms of Coagulopathy
The clinical rationale for restricting the use of synthetic colloids, particularly Hydroxyethyl Starches (HES) and Dextrans, is primarily based on their deleterious impact on the coagulation system [116].
These agents impair hemostasis through a dual mechanism:
- Dilutional Coagulopathy: Like all large-volume intravenous fluids, they reduce the systemic concentration of platelets and essential clotting factors.
- Specific Anticoagulant Effects: Colloids exert direct biochemical interference with the coagulation cascade, distinct from simple dilution [117–119].
Direct Haemostatic Interference
Colloid molecules directly compromise the integrity of the blood clot in several ways:
- Platelet Dysfunction: They interfere with both platelet adhesion and aggregation, reducing the effectiveness of the initial platelet plug.
- Factor Inhibition: They decrease the activity of critical coagulation components, specifically Factor VIII and Factor XIII [120].
- Fibrin Stability: Colloids compromise the polymerization and cross-linking of fibrin, resulting in a fragile, unstable clot that is highly susceptible to premature lysis.
This impairment of the coagulation system is dose-dependent, meaning the risk of exacerbated hemorrhage increases significantly as larger volumes of synthetic colloids are administered [120, 121].
While albumin-induced coagulopathy is documented, it is generally considered less severe and more readily reversible with fibrinogen concentrates than the coagulopathy induced by synthetic alternatives [121].
20. Target Haemoglobin After Bleeding Control
We recommend a target haemoglobin of 7–9 g/dL after controlling the source of bleeding.
Strength of
recommendation: Strong
Level of evidence: Low
Rationale:
Post-Operative
Haemoglobin Targets and Transfusion Strategy
The Restrictive Transfusion Model
Once definitive hemorrhage control is achieved, a restrictive red blood cell (RBC) transfusion strategy is recommended, targeting a hemoglobin (Hb) concentration of 7–9 g/dL. This threshold is supported by high-quality evidence indicating that a restrictive approach is non-inferior to more liberal strategies regarding patient morbidity and mortality [122].
The landmark TRICC trial established the safety of this lower threshold for the majority of critically ill and stable patients [122].
Adopting a restrictive range is clinically advantageous as it mitigates the risks of transfusion-related complications, such as:
- Transfusion-Associated Circulatory Overload (TACO)
- Nosocomial infections
- Immunomodulation and suppression [123]
By maintaining this target, clinicians utilize the body's physiological compensatory mechanisms to ensure tissue oxygenation while avoiding the adverse effects of excessive blood product administration [123, 124].
Clinical Exceptions
While the 7–9 g/dL target is the standard for most patients, specific populations require individualized management. Patients with acute ischemic conditions, most notably Acute Coronary Syndrome (ACS) or Traumatic Brain Injury (TBI) may necessitate a more liberal transfusion threshold to optimize cerebral and myocardial oxygen delivery and prevent secondary injury [124].
21. Prevention and Management of Hypothermia
We recommend
early application of measures such covering the patient and warm fluids to
reduce heat loss and warm the hypothermic patient to achieve and maintain
normothermia.
Strength of recommendation: Strong
Level of evidence: Low
Rationale:
Hypothermia
in Trauma Management
Impact on Mortality and Coagulopathy
Hypothermia is a critical independent predictor of poor outcomes in trauma, consistently correlating with increased mortality rates and a higher demand for blood products [125, 126].
A core body temperature falling below 35 °C often triggers a cascade of metabolic acidosis, hypotension, and impaired hemostasis, serving as a primary driver of trauma-induced coagulopathy [127].
The prognostic implications of heat loss are severe; logistic regression analyses indicate that once core temperatures drop below 34 °C, the probability of mortality exceeds 80%. This elevated risk persists regardless of the patient’s injury severity score (ISS), degree of shock, or the volume of transfusions received [128].
Hypothermia in Traumatic Brain Injury (TBI)
In the context of severe TBI, temperature regulation is particularly vital. Both spontaneous hypothermia upon hospital admission and therapeutically induced hypothermia have been significantly linked to increased mortality rates, suggesting that maintaining normothermia is essential for neurological preservation and survival in these patients [129, 130].
Prevention and Rewarming Strategies
Aggressive measures to maintain normothermia should begin in the prehospital phase and continue through definitive care.
Primary strategies include:
- Passive Warming: Removal of wet clothing and insulation to prevent ongoing heat loss.
- Active Warming: Increasing the ambient temperature of the resuscitation room and utilizing forced-air warming blankets.
- Internal Warming: Infusion of intravenous fluids warmed to 39–40 °C and, in extreme or refractory cases, the use of extracorporeal rewarming [131].
Furthermore, the use of specialized hypothermia management kits—originally developed for military tactical combat casualty care—is increasingly recommended for civilian use. These compact kits provide sustained dry heat for up to 10 hours and represent a practical, cost-effective adjunct for preventing the lethal progression of trauma-associated cooling [131].
22. Damage Control Surgery
We suggest damage control surgery in the severely injured patient if the definitive surgery to control the source of bleeding is complicated and time-consuming (>90 minutes) in the presence of severe persistent coagulopathy, severe acidosis with base deficit >15 mmol/L or lactate >5 mmol/L, hypothermia <34°C, or signs of ongoing bleeding despite the initial attempts of bleeding control with systolic BP persistently <90 mmHg.
Strength of
recommendation: Conditional
Level of evidence: Low
Rationale:
Damage
Control Surgery (DCS)
Definition and Historical Context
In the management of severe hemorrhagic shock, the delay of definitive hemorrhage control significantly increases morbidity and mortality. To address the needs of exsanguinating patients, Damage Control Surgery (DCS) was formalized by Rotondo et al. in 1993. Originally defined for abdominal trauma, the approach prioritizes the initial control of active bleeding and contamination, followed by intraperitoneal packing and rapid temporary closure [132, 133].
This strategy intentionally shifts the focus from definitive anatomical repair to the abbreviation of the initial operation. This allows for a period of intensive resuscitation in the ICU, where the patient's hemodynamic profile, temperature, and coagulation status can be stabilized prior to returning to the operating room for final management [133].
Benefits and Clinical Risks
The primary advantage of DCS is the stabilization of the "lethal triad," providing clinicians with the necessary time to reverse physiological insults before complex reconstructive surgery is attempted [134].
However, DCS carries a high risk of post-operative complications, including:
- Surgical site infections and sepsis.
- Incisional hernias and wound dehiscence.
- Abdominal compartment syndrome.
- The inherent physiological burden of multiple subsequent surgical procedures [134].
Reducing the Need for Damage Control
While DCS remains a life-saving intervention, recent shifts in Damage Control Resuscitation (DCR) including the early correction of coagulopathy, rapid hemorrhage control, and the avoidance of excessive crystalloid administration have been shown to improve physiological stability earlier in the treatment timeline. These adjunctive strategies may potentially reduce the necessity for full damage control procedures in a subset of severely injured patients [135].
23. Pelvic Binder Use
We recommend the
adjunct use of a pelvic binder or pelvic sheet to limit life threatening
bleeding in the presence of a suspected pelvic fracture.
Strength of recommendation: Strong
Level of evidence: Low
Rationale:
Emergency
Pelvic Stabilization
Role of External Stabilization
For patients presenting with unstable pelvic ring fractures, emergency external stabilization is a critical component of initial hemorrhage control. These techniques are widely utilized because they reduce pelvic volume and provide mechanical stability, which facilitates the tamponade of venous bleeding and protects nascent clots from disruption [136].
Clinical Application and DCO Principles
The use of non-invasive pelvic binders has become a standard of care in the pre-hospital and early clinical phases of resuscitation. However, the decision to apply such a device must be tailored to the patient's specific injury morphology. In alignment with the principles of Damage Control Orthopaedics (DCO), the primary objective is to prioritize rapid physiological stabilization and hemorrhage control over definitive anatomical fixation [137–139].
Data from major registries indicate that while binders are highly effective for "open-book" (anteroposterior compression) patterns, their appropriateness depends on a careful assessment of the fracture pattern to ensure that the compression does not exacerbate certain types of lateral compression injuries [137].
24. Topical Haemostatic Agents
We consider the use of topical haemostatic agents with packing for venous or moderate arterial bleeding associated with parenchymal injuries.
Strength of recommendation: Conditional
Level of evidence: Moderate
Rationale:
Local Haemostatic
Agents
Role as Surgical Adjuncts
A diverse array of local hemostatic agents is currently available to supplement traditional surgical methods, such as suturing or cauterization. These topical adjuncts are particularly advantageous in scenarios where anatomical access is restricted or when dealing with diffuse, non-compressible bleeding from parenchymal organs, bone, or vascular anastomoses [140, 142].
Clinical Evidence and Survival
The efficacy of these agents is well-documented in high-acuity settings. A retrospective review of the UK Joint Trauma Registry demonstrated that the early application of hemostatic dressings in severely injured military casualties was significantly associated with increased survival rates [140].
Similarly, in the prehospital setting, a systematic review corroborated the value of these dressings in managing catastrophic external hemorrhage before definitive surgical intervention could be reached [148].
Specialized Applications
Extensive clinical experience in humans has validated the use of various formulations across multiple surgical disciplines:
- Gelatine-Thrombin Matrices: Highly effective in vascular, renal, and spinal surgeries for managing active oozing [141, 145, 146].
- Fibrin Sealants: Proven to improve haemostasis in peripheral vascular procedures [142].
- Oxidized Cellulose and Collagen-Based Composites: Utilized effectively in thoracic surgery and for controlling bone-surface bleeding at donor sites [143, 144, 147].
- Intra-abdominal Packing: The combination of traditional laparotomy pads with haemostatic gauze (e.g., QuikClot) during damage control laparotomy has been analysed for safety and efficacy in stabilizing patients with profound intra-abdominal haemorrhage [149].
25. Tranexamic Acid (TXA)
We recommend TXA
administration in trauma patients who are bleeding or at risk of significant bleeding,
as soon as possible and within 3 hours of injury at a loading dose of 1 g IV
over 10 min, followed by 1 g IV infusion over 8 hours.
Strength of recommendation: Strong
Level of evidence: High
Rationale:
Tranexamic
Acid (TXA) in Trauma Resuscitation
Clinical Efficacy and Mortality Benefit
Tranexamic acid (TXA) has established itself as a cornerstone in the management of trauma patients at risk of significant hemorrhage. Large-scale clinical trials have demonstrated that its administration can reduce overall mortality by 1.5% and, more significantly, decrease deaths specifically attributed to bleeding by approximately one-third [150, 151]. Its mechanism as an antifibrinolytic helps stabilize formed clots, preventing their premature breakdown during the hyperfibrinolytic state often seen in trauma-induced coagulopathy.
Pre-hospital Administration and Dosing Strategies
The transition of TXA administration to the pre-hospital setting has been a major focus of recent research. In a randomized controlled trial of 927 trauma patients, a 1 g pre-hospital dose of TXA administered within two hours of injury resulted in a 30-day mortality rate of 8.1%, compared to 9.9% in the placebo group [155].
The impact of subsequent in-hospital dosing appears to follow a dose-response relationship. Data indicates that following a pre-hospital dose, 30-day mortality rates were lowest (7.3%) in patients who received a follow-up 1 g bolus plus a 1 g infusion, compared to those receiving an infusion only (7.8%) or no additional in-hospital TXA (9.3%) [155].
The "Golden Hour" and Patient Selection
The survival benefit of TXA is highly time dependent. Secondary analysis confirms that patients treated within one hour of injury particularly those with a shock index < 0.9 experienced a 65% lower likelihood of 30-day mortality [155].
Furthermore, early administration is associated with a lower incidence of multiorgan failure and reduced 24-hour transfusion requirements. Conversely, delayed treatment (> 3 hours from injury) has been shown in related literature to potentially increase the risk of death, reinforcing the necessity of early intervention [150, 155].
Recent applications have also expanded into Traumatic Brain Injury (TBI), where TXA has been shown to reduce the progression of intracranial hemorrhagic masses and improve survival in cases of mild-to-moderate TBI, provided it is administered promptly [151–154].
26. Balanced Blood Product Transfusion
We recommend
transfusion of pRBCs: FFP: Platelets in ratio following the massive transfusion
protocols, In the initial management of patients with suspected massive
haemorrhage.
Strength of recommendation: Strong
Level of evidence: Low
Rationale:
Initial
Transfusion Strategies: Fixed-Ratio Protocols
Empirical Ratio-Driven Resuscitation
During the critical interval between hospital arrival and the availability of laboratory coagulation results, many trauma centers employ an empirical, ratio-driven transfusion strategy. This approach involves the administration of Fresh Frozen Plasma (FFP), platelets, and packed Red Blood Cells (pRBCs) in fixed ratios to pre-emptively manage trauma-induced coagulopathy. While the precise benefit of fixed ratios remains a subject of debate, this strategy aims to mimic the composition of whole blood and prevent the dilution of clotting factors [157].
The PROPPR Trial and Haemostatic Outcomes
The definitive evidence for ratio-driven resuscitation comes from the PROPPR trial, which randomized 680 severely injured patients to receive blood products in either a 1:1:1 ratio (where platelets are included in the first transfusion pack) or a 1:1:2 ratio (where platelets are delayed until the second pack) [156].
The study found that while overall 24-hour and 30-day mortality rates were comparable between the two groups.
The 1:1:1 ratio offered significant physiological advantages:
- Improved Haemostasis: Patients in the 1:1:1 group achieved formal haemostasis significantly faster than those in the 1:1:2 group.
- Reduced Exsanguination: There was a notable reduction in deaths specifically attributed to early exsanguination in the 1:1:1 cohort [156].
Consequently, fixed-ratio protocols, particularly the 1:1:1 approach are widely integrated into Massive Transfusion Protocols (MTPs) to stabilize patients during the hyperacute phase of bleeding until goal-directed therapy can be initiated [157].
27. Calcium Monitoring and Supplementation
We recommend that
ionised calcium levels be monitored and maintained within the normal range
following major trauma and especially during massive transfusion. We recommend
the administration of calcium to correct hypocalcaemia.
Strength of recommendation: Strong
Level of evidence: Low
Rationale:
Calcium Homeostasis in Major Haemorrhage
In the management of patients with major trauma, maintaining normocalcaemia is a critical yet often overlooked component of resuscitation. It is strongly advised to perform frequent monitoring of ionized calcium levels, particularly during the activation of massive transfusion protocols.
Impact of Transfusion on Calcium Levels
The primary driver of hypocalcemia during resuscitation is the administration of large volumes of blood products. These products contain citrate, an anticoagulant that binds to free ionized calcium, rapidly lowering its systemic concentration. Since ionized calcium is a vital cofactor (Factor IV) in the coagulation cascade essential for the conversion of prothrombin to thrombin and the stabilization of fibrin low levels can severely exacerbate trauma-induced coagulopathy [158].
Clinical Intervention
Clinicians should maintain ionized calcium within normal physiological limits to support both hemostasis and myocardial contractility. Calcium supplementation (typically via calcium chloride or calcium gluconate) must be administered promptly whenever hypocalcemia is identified or empirically during rapid transfusion to prevent the "citrate toxicity" effect [158].
28. Reversal of Vitamin K Antagonists (VKA)
We recommend the
emergency reversal of vitamin K-dependent oral anticoagulants in the bleeding
trauma patient with the early use of 5–10 mg intravenous phytomenadione
(vitamin K1) in addition to FFP".
Strength of recommendation: Strong
Level of evidence: High
Rationale:
Reversal
of Vitamin K Antagonist (VKA) Therapy
Multi-Modal Reversal Strategy
In trauma patients presenting significant hemorrhage while on Vitamin K Antagonist (VKA) therapy (e.g., Warfarin), emergency reversal of anticoagulation is a critical priority. Current guidelines recommend a dual-action approach: the immediate administration of four-factor Prothrombin Complex Concentrate (PCC) supplemented with 5–10 mg of intravenous phytomenadione (Vitamin K1) [159, 160].
Physiological Rationale
The necessity for this combined therapy is rooted in the distinct pharmacokinetics of the reversal agents:
- Intravenous Vitamin K: Provides the essential substrate for the hepatic synthesis of functional Factors II, VII, IX, and X. While vital for sustained hemostasis, the onset of action is delayed, typically requiring 6 to 12 hours for the liver to produce sufficient new clotting factors [161].
- Prothrombin Complex Concentrate (PCC): Offers a concentrated source of the Vitamin K-dependent factors, bypassing the need for hepatic synthesis and providing immediate correction of the International Normalized Ratio (INR).
PCC vs. Fresh Frozen Plasma (FFP)
Historically, Fresh Frozen Plasma (FFP) was utilized for VKA reversal; however, its use in trauma is increasingly restricted by volume limitations. Achieving effective reversal with FFP often necessitates dosages of 15–30 mL/kg, which equates to 1 to 2 liters of fluid for an average adult. This high-volume requirement carries significant risks, including Transfusion-Associated Circulatory Overload (TACO) and acute cardiac strain [161].
Furthermore, high-quality randomized evidence, such as the INCH trial, has demonstrated that PCC is superior to FFP in achieving rapid normalization of the INR and limiting the expansion of life-threatening bleeding, particularly in cases of intracranial hemorrhage [161].
- Acknowledgement
Chairperson of the Guideline Development Group (GDG):
· Prof. Dr. Jehan Ali ElKholy, Head of Scientific Committee of the Egyptian Board of the Emergency Medicine. Head of Anaesthesia, SICU & Pain Management Cairo University. Provided overall supervision and coordination of the guideline development process. Conducted comprehensive revision of the introduction and all recommendations
Members of the Guideline Development Group (GDG):
· Prof. Dr. Jehan Ali ElKholy, Head of Scientific Committee of the Egyptian Board of the Emergency Medicine. Head of Anaesthesia, SICU & Pain Management Cairo University.
· Prof. Dr. Maha Mohamed Ismail Youssef
Professor of anaesthesia, Surgical ICU, and Pain Management, Faculty of medicine, Cairo University (GLN 5,6, 15,16,21,23, references distribution and revision)
· Dr. Ahmed Abouelyazeid Aly Abouelyazeid
Consultant of Emergency medicine and Traumatology, Director of Planning at Central Administration for Training; Egyptian Ambulance Organization, Chapter director of International Trauma Life Support ITLS at EAO. (GLN 9,10)
· Dr. Omar Mohamed Mattar
Assistant lecturer of Emergency Medicine, Faculty of Medicine, Cairo University
(GLN 24)
· Heba Ali Abdelaziz Eldash
Methodologist, Head of clinical pharmacy department in Banha Teaching Hospital (Final revision and format editing)
· Dr. Ahmad Majed Abdul Mohsen Abdulaziz Younis
Assistant lecturer of Emergency Medicine & Traumatology, Faculty of Medicine, Mansoura University (introduction, GLN 1,2)
· Dr. Ahmed Mounir Sorour
Emergency Officer / Public Health & Disaster Medicine Specialist, World Health Organization in Egypt (GLN 3,4)
· Dr. Bassel Adel Elkhouly
Assistant Lecturer of Emergency Medicine, Faculty of Medicine, Cairo University (GLN 7,8 )
· Dr. Eman Mohamed Gaber
Emergency Medicine consultant - Deputy Manager of Critical and Urgent Care Administration, Alexandria Health Directorate (GLN 11, 12)
· Dr. Fatma Abdelsalam Abdalla Ali
Lecturer of Emergency Medicine & Traumatology, Faculty of Medicine, Tanta University (GLN 13, 14)
· Dr. Mohamed Elsayed Elsherbiny
Emergency medicine consultant, Chief of Training central administration, Egyptian Ambulance Organization (GLN 17, 18)
· Dr. Mohamed Rashad Abdelaziz
Emergency and disaster medicine consultant, International Federation for red cross and red crescent (GLN 19, 20)
· Dr. Mostafa Abdelnaby
Assistant lecturer of Anaesthesia, surgical ICU, and pain management, Faculty of medicine, Cairo University (GLN 22)
· Dr. Omar Nasr
Emergency Medicine Consultant -Head of the Emergency Department, Central Administration of Emergency & Critical Care – MOHP
· Dr. Sally Wassfi
Lecturer of Emergency Medicine, Faculty of Medicine, Alexandria University, ERC course director and educator (GLN 26)
· Dr. Shaimaa El Sayed Mohamed
Clinical pathology, Infection control consultant, National methodologist
(GLN 27)
· Dr. Noha Ahmed
Manager of Critical and Urgent Care administration Alexandria Health Affairs Directorate, Neonatology consultant (GLN 28)
· Dr A'laa Atef Shalaby
Critical Care and Healthcare Quality Assurance Specialist - Central Administration of Emergency and Critical care - MOHP
· Ph. Heba Abdelkader Hussein
Methodologist - Pharmacy department manager in Mataria Teaching Hospital, GOTHI
We would like to acknowledge the efforts of Supervisors of the guidelines:
· Prof. Dr. Jehan Ali ElKholy
· Prof. Dr. Maha Mohamed Ismail Youssef
Coordinators & Editing Supervisors
1. Prof. Dr. Maha Mohamed Ismail Youssef
2. Dr. Ahmed Abouelyazeid Aly Abouelyazeid
3. Dr. Omar Mohamed Mattar
- Abbreviations
ABCDE Approach: Airway, Breathing, Circulation, Disability and Exposure approach
ACS: Acute Coronary Syndrome
aPTT: Activated Partial Thromboplastin Time
ATLS: Advanced Trauma Life Support
AUROC: Area Under the Receiver Operating Characteristics
CCT: Conventional coagulation tests
DCR: Damage Control Resuscitation
eFAST: extended Focused Assessment with Sonography for Trauma
EHC: Egyptian Health Council
GCS: Glascow Coma Scale
GDG: Guideline Development Group
GRADE: Grading of Recommendations, Assessment, Development and Evaluation
HES: Hydroxyethyl Starches
ICD-11: International Classification of Diseases, 11th Edition
INR: International Normalized ratio
MOHP: the Ministry of Health and Population
MT: massive transfusion
NGOs: Non-Governmental Organizations
PCC: Prothrombin Complex Concentrate
POCUS: Point-of-Care Ultrasonography
PP: Pulse Pressure
PT: Prothrombin Time
ROTEM: Rotational Thromboelastometry
SBP: Systolic Blood Pressure
SI: Shock Index
TACO: Transfusion-Associated Circulatory Overload
TBI: Traumatic Brain Injury
TEG: Thromboelastography
TIC: Trauma-Induced Coagulopathy
TXA: Tranexamic Acid
VHAs: Viscoelastic Haemostatic Assays
VKA: Vitamin K Antagonists
WBCT: Whole-Body Computed Tomography
WHO: World Health Organization.
- Glossary
Damage Control Resuscitation (DCR): It emphasizes Permissive hypotension until bleeding is controlled, Balanced blood‑product transfusion (plasma, platelets, red cells), Restricted crystalloid use to avoid dilutional coagulopathy and Early correction of coagulopathy.
Multidisciplinary Approach: Integration of emergency medicine, surgery, anaesthesia, and intensive care teams to deliver coordinated haemostatic resuscitation tailored to patient physiology.
- Introduction
The Global and National Challenge
Massive hemorrhage remains the primary cause of preventable mortality in polytrauma patients, presenting a formidable challenge to healthcare systems both globally and within Egypt. The clinical progression from uncontrolled bleeding to hypovolemic shock, coagulopathy, and multi-organ failure occurs with high velocity, necessitating immediate recognition and definitive intervention.
Central to the pathophysiology of traumatic death is the "lethal triad"—the synergistic effect of hypothermia, metabolic acidosis, and coagulopathy. These factors are the key determinants of survival during the critical "golden hour" of resuscitation, and their mitigation is the primary goal of early trauma care [1, 2].
Evolution of Trauma Care: Damage Control Resuscitation
Over the last decade, the management of major haemorrhage has shifted toward Damage Control Resuscitation (DCR). This evidence-based multidisciplinary approach prioritizes:
- Permissive Hypotension: Maintaining limited perfusion until surgical control is achieved.
- Balanced Transfusion: Utilizing fixed-ratio blood product administration (e.g., 1:1:1 pRBC, plasma, and platelets) to mimic whole blood [3].
- Restricted Crystalloids: Minimizing the use of clear fluids to avoid dilutional coagulopathy and tissue oedema [4, 5].
- Scope and Purpose
Egypt faces a high trauma burden, placing emergency physicians at the forefront of haemorrhage management. This chapter provides standardized, evidence‑based recommendations for the recognition and management of massive bleeding in trauma patients to be used by policymakers, health professionals, and emergency health care providers to offer better treatment strategies.
The objective of this chapter is to standardize practice to decrease variation of practice and improve outcomes for management of massive bleeding in trauma patients.
Strategic Objectives
- Standardize clinical practice across emergency departments and trauma centres.
- Optimize resource utilization by standardizing transfusion and resuscitation protocols.
- Strengthen coordination among multidisciplinary teams for rapid haemorrhage control.
- Enhance quality, safety, and equity of emergency medical care nationwide.
- Reduce preventable trauma‑related mortality through evidence‑informed practice.
Impact
By institutionalizing an evidence‑based approach to trauma resuscitation, this guideline contributes to Egypt’s broader vision of improving emergency care systems, reducing mortality, and aligning national practice with international standards.
- Target Audience
This guideline targets emergency physicians, general practitioners, policy makers, as well as non-governmental organizations (NGOs) and other stakeholders to offer the most appropriate treatment guidelines for trauma patients with massive bleeding.
- Methodology
This national guideline was developed under the auspices of the Egyptian Health Council (EHC), the Ministry of Health and Population (MOHP), and the World Health Organization (WHO) Egypt Country Office. To ensure international standards of transparency and clinical rigor while maintaining local contextual relevance, the GRADE-ADOLOPMENT methodology was employed for the adaptation of global evidence into the Egyptian clinical landscape [6,7].
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria were followed in the search and retrieval of guidelines to be adapted:
- Selecting only evidence-based guidelines (guidelines must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
· Selecting only national and/or international guidelines
· Selecting peer-reviewed publications only
· Selecting guidelines written in the English language
· Excluding guidelines written by a single author, not on behalf of an organization, to be valid and comprehensive, a guideline ideally requires multidisciplinary input
· Excluding guidelines published without references, as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in:
· Developing organization/authors
· Date of publication, posting, and release
· Country/language of publication
· Date of posting and/or release
· Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point or ranked the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The GDG decided to adapt the Guidelines for the management of conditions specifically related to stress by the WHO and the Guidelines of the International Society for the Study of Trauma and Dissociation (ISSTD). (2,6)
- Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of
Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because they represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
• GRADE working group: https://www.gradeworkinggroup.org/
• GRADE online training modules: http://cebgrade.mcmaster.ca/
Table 1 Quality and Significance of the four levels of evidence in GRADE:
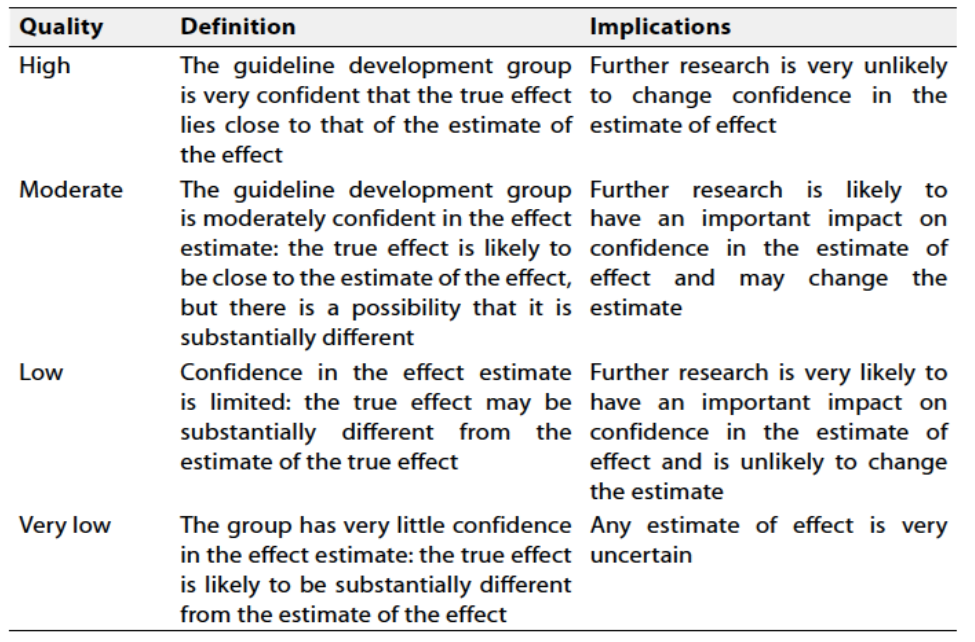
Table 2 Factors that determine How to upgrade or downgrade the quality of evidence
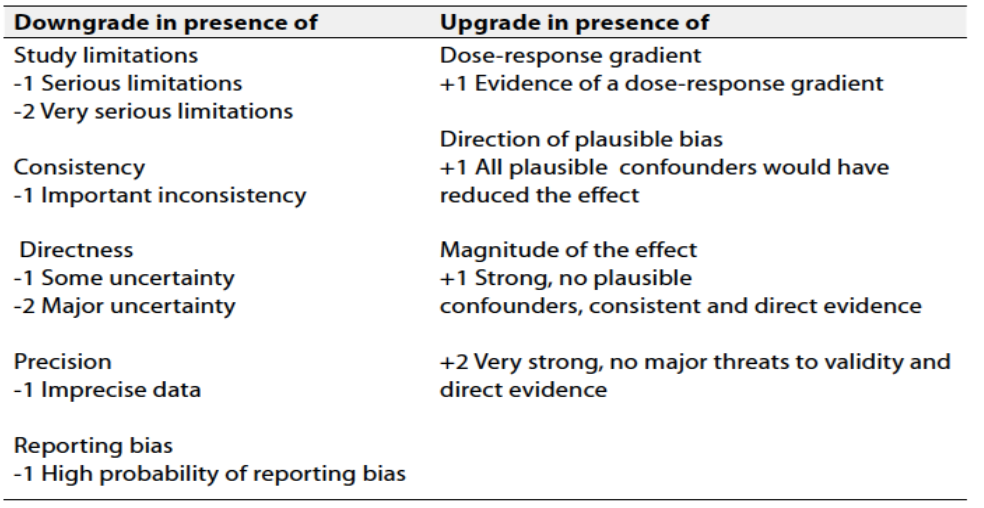
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
These are made when there is greater uncertainty about the four factors above or if local adaptation should account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.- Research Needs
- The study of coagulation parameters in polytrauma patients and their effects on outcome.
- Resuscitation of polytrauma patients: the management of massive extremity bleeding.
- Traumatic coagulopathy and massive transfusion: improving outcomes.
- Monitoring and Evaluating the Impact of the Guideline
- Hb and/or Hct measurements.
- International Normalized Ratio (INR)
- Platelet Count.
- Whole-Body CT (WBCT)
- Lactate and Base Deficit Monitoring
- Point-of-Care Ultrasonography (POCUS)
- Update of the Guideline
This guideline will be updated whenever there is new evidence
- References
- Kauvar DS, Lefering R, Wade CE. Impact of hemorrhage on trauma outcome: an overview of epidemiology, clinical presentations, and therapeutic considerations. J Trauma. 2006;60(6 Suppl):S3–11.
- Brohi K, Singh J, Heron M, Coats T. Acute traumatic coagulopathy. J Trauma. 2003;54(6):1127–33.
- Holcomb JB, Tilley BC, Baraniuk S, et al. Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs 1:1:2 ratio and mortality in patients with severe trauma (PROPPR trial). JAMA. 2015;313(5):471–82.
- Spahn DR, Bouillon B, Cerny V, et al. The European guideline on management of major bleeding and coagulopathy following trauma: sixth edition. Crit Care. 2023;27(1):87.
- Cannon JW. Hemorrhagic shock. N Engl J Med. 2018;378(4):370–9.
- Schünemann HJ, Wiercioch W, Brozek J, et al. GRADE Evidence-to-Decision (EtD) frameworks for adoption, adaptation, and de novo development of trustworthy recommendations: GRADE-ADOLOPMENT. J Clin Epidemiol. 2017;81:101–10.
- World Health Organization (WHO) Country Office – Egypt; Egyptian Health Council (EHC); Ministry of Health and Population (MOHP). Emergency Medicine Guideline Development Training Workshop Report: Using GRADE Methodology for Guideline Adaptation. Alexandria, Egypt: WHO; 2025.
- Alharbi RJ, Lewis V, Shrestha S, Miller C. Effectiveness of trauma care systems at different stages of development in reducing mortality: a systematic review and meta-analysis protocol. BMJ Open. 2021;11(6):e047439.
- Dufresne P, Moore L, Tardif P, Razek T, Boutin A, Clément J. Impact of trauma centre designation level on outcomes following haemorrhagic shock: a multicentre cohort study. Can J Surg. 2017;60(1):45–52.
- Kalkwarf KJ, Drake SA, Yang Y, Thetford C, Myers L, Brock M, Wolf DA, Persse D, Wade CE, Holcomb JB. Bleeding to death in a big city: an analysis of all trauma deaths from haemorrhage in a metropolitan area during 1 year. J Trauma Acute Care Surg. 2020;89(4):716–22.
11. Ruelas OS, Tschautscher CF, Lohse CM, Sztajnkrycer MD. Analysis of prehospital scene times and interventions on mortality outcomes in a national cohort of penetrating and blunt trauma patients. Prehosp Emerg Care. 2018;22(6):691–7.
12. Nasser AAH, Nederpelt C, El Hechi M, Mendoza A, Saillant N, Fagenholz P, Velmahos G, Kaafarani HMA. Every minute counts: the impact of pre-hospital response time and scene time on mortality of penetrating trauma patients. Am J Surg. 2020;220(1):240–4.
13. Harmsen AMK, Giannakopoulos GF, Moerbeek PR, Jansma EP, Bonjer HJ, Bloemers FW. The influence of prehospital time on trauma patients’ outcome: a systematic review. Injury. 2015;46(4):602–9.
14. Chen C, Shin SD, Sun J, Jamaluddin SF, Tanaka H, Song KJ, Kajino K, Kimura A, Huang EP, Hsieh M, et al. Association between prehospital time and outcome of trauma patients in four Asian countries: a cross-national, multicentre cohort Chehab study. PLoS Med. 2020;17(10):e1003360.
15. M, Afaneh A, Bible L, Castanon L, Hanna K, Ditillo M, Khurrum M, Asmar S, Joseph B. Angioembolization in intra-abdominal solid organ injury: does delay in angioembolization affect outcomes? J Trauma Acute Care Surg. 2020;89(4):723–9.
16. Matsushima K, Khor D, Berona K, Antoku D, Dollbaum R, Khan M, Demetriades D. Double jeopardy in penetrating trauma: get FAST, get it right. World J Surg. 2018;42(1):99–106.
17. Singh RA, Asprou F, Patel A, Trickett RW. Haemorrhage control in extremity stab injury. J Surg Case Rep. 2013;2013(12):rjt093.
18. Van Waes OJ, Cheriex KCAL, Navsaria PH, van Riet PA, Nicol AJ, Vermeulen J. Management of penetrating neck injuries. Br J Surg. 2012;99(Suppl 1):149–54.
19. Bulger EM, Snyder D, Schoelles K, Gotschall C, Dawson D, Lang E, Sanddal ND, Butler FK, Fallat M, Taillac P, et al. An evidence-based prehospital guideline for external haemorrhage control: American College of Surgeons Committee on Trauma. Prehosp Emerg Care. 2014;18(2):163–73.
20. Lakstein D, Blumenfeld A, Sokolov T, Lin G, Bssorai R, Lynn M, Ben-Abraham R. Tourniquets for haemorrhage control on the battlefield: a 4-year accumulated experience. J Trauma. 2003;54(5 Suppl):S221–5.
21. Beekley AC, Sebesta JA, Blackbourne LH, Herbert GS, Kauvar DS, Baer DG, Walters TJ, Mullenix PS, Holcomb JB. Prehospital tourniquet use in Operation Iraqi Freedom: effect on haemorrhage control and outcomes. J Trauma. 2008;64(2 Suppl):S28–37; discussion S37.
22. Kragh JF Jr, Cooper A, Aden JK, Dubick MA, Baer DG, Wade CE, Blackbourne LH. Survey of trauma registry data on tourniquet use in paediatric war casualties. Pediatr Emerg Care. 2012;28(12):1361–5.
23. Eilertsen KA, Winberg M, Jeppesen E, Hval G, Wisborg T. Prehospital tourniquets in civilians: a systematic review. Prehosp Disaster Med. 2021;36(1):86–94.
24. Dayan L, Zinmann C, Stahl S, Norman D. Complications associated with prolonged tourniquet application on the battlefield. Mil Med. 2008;173(1):63–6.
25. Kragh JF Jr, O’Neill ML, Walters TJ, Jones JA, Baer DG, Gershman LK, Wade CE, Holcomb JB. Minor morbidity with emergency tourniquet use to stop bleeding in severe limb trauma: research, history, and reconciling advocates and abolitionists. Mil Med. 2011;176(7):817–23.
26. American College of Surgeons. ATLS®: Advanced Trauma Life Support Student Course Manual. 10th ed. Chicago (IL): American College of Surgeons; 2018. p. 606–3211.
27. Mutschler
M, Nienaber U, Brockamp T, Wafaisade A, Wyen H, Peiniger S, Pafrath T, Bouillon
B, Maegele M. A critical reappraisal of the ATLS clas‑
sifcation of hypovolaemic shock: does it really refect clinical reality?
Resuscitation. 2013;84(3):309–13.vb
28. Tran
A, Matar M, Lampron J, Steyerberg E, Taljaard M, Vaillancourt C.
Early identifcation of patients requiring massive transfusion, emboliza‑
tion or haemostatic surgery for traumatic haemorrhage: a systematic
review and meta-analysis. J Trauma Acute Care Surg. 2018;84(3):505–16.
29. Liu
C, Wang C, Shih H, Wen Y, Wu JJ, Huang C, Hsu H, Huang M, Huang
M. Prognostic factors for mortality following falls from height. Injury.
2009;40(6):595–7.
30. Nutbeam
T, Fenwick R, Smith J, Bouamra O, Wallis L, Stassen W. A
comparison of the demographics, injury patterns and outcome data for
patients injured in motor vehicle collisions who are trapped compared
to those patients who are not trapped. Scand J Trauma Resusc Emerg
Med. 2021;29(1):17.
31. Vandromme
MJ, Grifn RL, Kerby JD, McGwin G Jr, Rue LW 3rd,
Weinberg JA. Identifying risk for massive transfusion in the relatively
normotensive patient: utility of the prehospital shock index. J Trauma.
2011;70(2):384–8 (discussion 388-390).
32. Campos-Serra
A, Montmany-Vioque S, Rebasa-Cladera P, Llaquet-Bayo
H, Gràcia-Roman R, Colom-Gordillo A, Navarro-Soto S. The use of the
shock index as a predictor of active bleeding in trauma patients. Cir Esp
(Engl Ed). 2018;96(8):494–500.
33. Schroll R, Swift D, Tatum D, Couch S, Heaney
JB, Llado-Farrulla M, Zucker
S, Gill F, Brown G, Bufn N, et al. Accuracy of shock index versus ABC
score to predict need for massive transfusion in trauma patients. Injury.
2018;49(1):15–9.
34. Terceros-Almanza
LJ, García-Fuentes C, Bermejo-Aznárez S, Prieto Del
Portillo IJ, Mudarra-Reche C, Domínguez-Aguado H, Viejo-Moreno
R, Barea-Mendoza J, Gómez-Soler R, Casado-Flores I, et al. Prediction
of massive bleeding in a prehospital setting: validation of six scoring
systems. Med Intensiva (Engl Ed). 2019;43(3):131–8.
35. Sorensen
DA, April MD, Fisher AD, Schauer SG. An analysis of the shock
index and pulse pressure as a predictor for massive transfusion and
death in US and coalition Iraq and Afghanistan. Med J (Ft Sam Houst
Tex) 2021(Pb 8-21-07/08/09):63–68.
36. El-Menyar A, Goyal P, Tilley E, Latif R. The
clinical utility of shock index to
predict the need for blood transfusion and outcomes in trauma. J Surg
Res. 2018;227:52–9.
37. Kheirbek
T, Martin TJ, Cao J, Hall BM, Lueckel S, Adams CA. Prehospital
shock index outperforms hypotension alone in predicting signifcant
injury in trauma patients. Trauma Surg Acute Care Open. 2021;6(1):
e000712.
38. Priestley
EM, Inaba K, Byerly S, Biswas S, Wong MD, Lam L, Benjamin E,
Demetriades D. Pulse pressure as an early warning of haemorrhage in
trauma patients. J Am Coll Surg. 2019;229(2):184–91.
39. Bankhead-Kendall
B, Teixeira P, Roward S, Ali S, Ryder A, Sahi S, Card‑
enas T, Aydelotte J, Coopwood B, Brown C. Narrow pulse pressure is
independently associated with massive transfusion and emergent
surgery in haemodynamically stable trauma patients. Am J Surg.
2020;220(5):1319–22.
40. Schellenberg M, Owattanapanich N, Getrajdman
J, Matsushima K, Inaba
K. Prehospital narrow pulse pressure predicts need for resuscitative
thoracotomy and emergent intervention after trauma. J Surg Res.
2021;268:284–90.
41. Warren J, Moazzez A, Chong V, Putnam B,
Neville A, Singer G, Deane
M, Kim DY. Narrowed pulse pressure predicts massive transfusion and
emergent operative intervention following penetrating trauma. Am J
Surg. 2019;218(6):1185–8.
42. Johnson JW, Gracias VH, Schwab CW, Reilly PM, Kauder DR, Shapiro MB, Dabrowski GP, Rotondo MF. Evolution in damage control for exsanguinating penetrating abdominal injury. J Trauma. 2001;51(2):261–9.
43. Johnson A, Rott M, Kuchler A, Williams E, Cole F, Ramzy A, Barbosa R, Long W, Martin MJ. Direct to operating room trauma resuscitation: optimizing patient selection and time‑critical outcomes when minutes count. J Trauma Acute Care Surg. 2020;89(1):160–6.
44. Kinoshita T, Yamakawa K, Matsuda H, Yoshikawa Y, Wada D, Hamasaki T, Ono K, Nakamori Y, Fujimi S. The survival benefit of a novel trauma workflow that includes immediate whole‑body computed tomography, surgery, and interventional radiology, all in one trauma resuscitation room: a retrospective historical control study. Ann Surg. 2019;269(2):370–6.
45. Huber‑Wagner S, Lefering R, Qvick LM, Körner M, Kay MV, Pfeifer KJ, Reiser M, Mutschler W, Kanz KG. Effect of whole‑body CT during trauma resuscitation on survival: a retrospective, multicentre study. Lancet. 2009;373(9673):1455–61.
46. Huber‑Wagner S, Mand C, Ruchholtz S, Kühne CA, Holzapfel K, Kanz K, van Griensven M, Biberthaler P, Lefering R. Effect of the localisation of the CT scanner during trauma resuscitation on survival: a retrospective, multicentre study. Injury. 2014;45(Suppl 3):S76–82.
47. Huber‑Wagner S, Biberthaler P, Häberle S, Wierer M, Dobritz M, Rummeny E, van Griensven M, Kanz KG, Lefering R. Whole‑body CT in haemodynamically unstable severely injured patients: a retrospective, multicentre study. PLoS One. 2013;8(7):e68880.
48. van der Weide L, Popal Z, Terra M, Schwarte LA, Ket JCF, Kooij FO, Exadaktylos AK, Zuidema WP, Giannakopoulos GF. Prehospital ultrasound in the management of trauma patients: systematic review of the literature. Injury. 2019;50(12):2167–75.
- Mercer CB, Ball M, Cash RE, Rivard MK, Chrzan K, Panchal AR. Ultrasound use in the prehospital setting for trauma: a systematic review. Prehosp Emerg Care. 2021;25(4):566–82.
- Stengel D, Leisterer J, Ferrada P, Ekkernkamp A, Mutze S, Hoenning A. Point‑of‑care ultrasonography for diagnosing thoracoabdominal injuries in patients with blunt trauma. Cochrane Database Syst Rev. 2018;12(12):CD012669.
- Rowell SE, Barbosa RR, Holcomb JB, Fox EE, Barton CA, Schreiber MA. The focused assessment with sonography in trauma (FAST) in hypotensive injured patients frequently fails to identify the need for laparotomy: a multi‑institutional pragmatic study. Trauma Surg Acute Care Open. 2019;4(1):e000207.
- Staub LJ, Biscaro RRM, Kaszubowski E, Maurici R. Chest ultrasonography for the emergency diagnosis of traumatic pneumothorax and haemothorax: a systematic review and meta‑analysis. Injury. 2018;49(3):457–66.
- Gonzalez‑Hadad A, García AF, Serna JJ, Herrera MA, Morales M, Manzano‑Nunez R. The role of ultrasound for detecting occult penetrating cardiac wounds in haemodynamically stable patients. World J Surg. 2020;44(5):1673–80.
- Ianniello S, Conte P, Di Serafino M, Miele V, Trinci M, Vallone G, Galluzzo M. Diagnostic accuracy of pubic symphysis ultrasound in the detection of unstable pelvis in polytrauma patients during e‑FAST: the value of FAST‑PLUS protocol. A preliminary experience. J Ultrasound. 2021;24(4):423–8.
- Pigneri DA, Behm RJ, Granet PJ. Rolling a trauma patient onto the right-side increases sensitivity of FAST examination. J Clin Ultrasound. 2020;48(3):152–5.
56. Hajibandeh S, Hajibandeh S. Systematic review: effect of whole‑body computed tomography on mortality in trauma patients. J Inj Violence Res. 2015;7(2):64–74.
57. Caleo O, Bocchini G, Paoletta S, Ierardi AM, Scionti A, Tonerini M, Guida F, Sica G, Perillo A, Carrafiello G, et al. Spontaneous non‑aortic retroperitoneal haemorrhage: etiology, imaging characterization and impact of MDCT on management. A multicentric study. Radiol Med. 2015;120(1):133–48.
58. Shreffler J, Smiley A, Schultz M, Ross A, Baker J, Nash N, Harbrecht B, Huecker M. Patients with abrasion or ecchymosis seat belt sign have high risk for abdominal injury, but initial computed tomography is 100% sensitive. J Emerg Med. 2020;59(4):491–8.
59. Sierink JC, Treskes K, Edwards MJ, Beuker BJ, den Hartog D, Hohmann J, Dijkgraaf MG, Luitse JS, Beenen LF, Hollmann MW, et al. Immediate total‑body CT scanning versus conventional imaging and selective CT scanning in patients with severe trauma (REACT‑2): a randomised controlled trial. Lancet. 2016;388(10045):673–83.
60. Treskes K, Saltzherr TP, Edwards MJR, Beuker BJA, Den Hartog D, Hohmann J, Luitse JS, Beenen LFM, Hollmann MW, Dijkgraaf MGW, et al. Emergency bleeding control interventions after immediate total‑body CT scans in trauma patients. World J Surg. 2019;43(2):490–6.
61. Arruzza E, Chau M, Dizon J. Systematic review and meta‑analysis of whole‑body computed tomography compared to conventional radiological procedures of trauma patients. Eur J Radiol. 2020;129:109099.
62. Murao S, Yamakawa K, Kabata D, Kinoshita T, Umemura Y, Shintani A, Fujimi S. Effect of earlier door‑to‑CT and door‑to‑bleeding control in severe blunt trauma: a retrospective cohort study. J Clin Med. 2021;10(7):1522.
63. Treskes K, Saltzherr TP, Edwards MJR, Beuker BJA, Van Lieshout EMM, Hohmann J, Luitse JSK, Beenen LFM, Hollmann MW, Dijkgraaf MGW, et al. Refining the criteria for immediate total‑body CT after severe trauma. Eur Radiol. 2020;30(5):2955–63.
64. Cieslak JA 3rd, Jazmati T, Patel A, Chaudhry H, Kumar A, Contractor S, Shukla PA. Trauma CT evaluation prior to selective angiography in patients with traumatic injuries: negative predictive power and factors affecting its utility. Emerg Radiol. 2020;27(5):477–86.
65. Gamal M, Abdelhamid B, Zakaria D, Dayem OAE, Rady A, Fawzy M, Hasanin A. Evaluation of noninvasive haemoglobin monitoring in trauma patients with low haemoglobin levels. Shock. 2018;49(2):150–3.
66. Kass
LE, Tien IY, Ushkow BS, Snyder HS. Prospective crossover study of
the efect of phlebotomy and intravenous crystalloid on haematocrit.
Acad Emerg Med. 1997;4(3):198–201.
67. Ryan ML, Thorson CM, Otero CA, Vu T, Schulman CI, Livingstone AS, Proctor KG. Initial haematocrit in trauma: a paradigm shift? J Trauma Acute Care Surg. 2012;72(1):54–9.
68. Knottenbelt JD. Low initial haemoglobin levels in trauma patients: an important indicator of ongoing haemorrhage. J Trauma. 1991;31(10):1396–9.
69. Thorson CM, Van Haren RM, Ryan ML, Pereira R, Olloqui J, Guarch GA, Barrera JM, Busko AM, Livingstone AS, Proctor KG. Admission haematocrit and transfusion requirements after trauma. J Am Coll Surg. 2013;216(1):65–73.
70. Thorson CM, Ryan ML, Van Haren RM, Pereira R, Olloqui J, Otero CA, Schulman CI, Livingstone AS, Proctor KG. Change in haematocrit during trauma assessment predicts bleeding even with ongoing fluid resuscitation. Am Surg. 2013;79(4):398–406.
71. Zehtabchi S, Sinert R, Goldman M, Kapitanyan R, Ballas J. Diagnostic performance of serial haematocrit measurements in identifying major injury in adult trauma patients. Injury. 2006;37(1):46–52.
72. Holstein
JH, Culemann U, Pohlemann T. Working group mortality in
pelvic fracture patients: What are predictors of mortality in patients
with pelvic fractures? Clin Orthop Relat Res. 2012;470(8):2090–7.
73. Schlimp
CJ, Voelckel W, Inaba K, Maegele M, Ponschab M, Schöchl H.
Estimation of plasma fbrinogen levels based on haemoglobin, base
excess and injury severity score upon emergency room admission. Crit
Care. 2013;17(4):R137.
74. Figueiredo
S, Taconet C, Harrois A, Hamada S, Gauss T, Raux M, Duran‑
teau J. Traumabase group: How useful are haemoglobin concentra‑
tion and its variations to predict signifcant haemorrhage in the early
phase of trauma? A multicentric cohort study. Ann Intensive Care.
2018;8(1):76.
75. Broder G, Weil MH. Excess lactate: an index of reversibility of shock in human patients. Science. 1964;143(3613):1457–9.
76. Caputo N, Fraser R, Paliga A, Kanter M, Hosford K, Madlinger R. Triage vital signs do not correlate with serum lactate or base deficit, and are less predictive of operative intervention in penetrating trauma patients: a prospective cohort study. Emerg Med J. 2013;30(7):546–50.
77. Vincent JL, Quintairos e Silva A, Couto L Jr, Taccone FS. The value of blood lactate kinetics in critically ill patients: a systematic review. Crit Care. 2016;20(1):257.
78. Mutschler M, Nienaber U, Brockamp T, Wafaisade A, Fabian T, Paffrath T, Bouillon B, Maegele M, TraumaRegister DGU. Renaissance of base deficit for the initial assessment of trauma patients: a base deficit‑based classification for hypovolemic shock developed on data from 16,305 patients derived from the TraumaRegister DGU®. Crit Care. 2013;17(2):R42.
79. Randolph LC, Takacs M, Davis KA. Resuscitation in the pediatric trauma population: admission base deficit remains an important prognostic indicator. J Trauma. 2002;53(5):838–42.
80. Porter JM, Ivatury RR. In search of the optimal end points of resuscitation in trauma patients: a review. J Trauma. 1998;44(5):908–14.
81. Hess JR, Lindell AL, Stansbury LG, Dutton RP, Scalea TM. The prevalence of abnormal results of conventional coagulation tests on admission to a trauma center. Transfusion. 2009;49(1):34–9. doi:10.1111/j.1537-2995.2008.01944.x.
82. Jonish AP, James JD, Titus DK, Sharma SL, Purushothaman V, Dave RG, Nair SC, Chase S, Nayak S, Kumar V. Conventional coagulation tests are a better predictor of mortality than viscoelastic haemostatic assays in trauma patients with shock: a prospective observational study. Eur J Trauma Emerg Surg. 2025;51:Article 131. doi:10.1007/s00068-025-02808-8.
83. Hutspardol S, Borja T, Kroeker J, Wang XQ, Mi J, Zamar D, Chan G, Smith T, Hawes H, Shih AW. Comparison of conventional coagulation tests and ROTEM in identifying trauma‑induced coagulopathy for massive haemorrhage protocol activation. Transfus Med. 2025. doi:10.1111/tme.13128.
84. Nevin DG, Brohi K. Permissive hypotension for active haemorrhage in trauma. Anaesthesia. 2017;72(12):1443–8.
85. Moy HP, Cosgrove A. Permissive hypotension in trauma: viable strategy in the prehospital setting. EMS World. 2016;45(3):44–7.
86. Bickell WH, et al. Immediate versus delayed fluid resuscitation for hypotensive patients with penetrating torso injuries. N Engl J Med. 1994;331(17):1105–9.
87. Tran A, Yates J, Lau A, Lampron J, Matar M. Permissive hypotension ver‑sus conventional resuscitation strategies in adult trauma patients with haemorrhagic shock: a systematic review and meta-analysis of randomised controlled trials. J Trauma Acute Care Surg. 2018;84(5):802–8.
88. Safejko K, Smereka J, Filipiak KJ, Szarpak A, Dabrowski M, Ladny JR, Jaguszewski MJ, Szarpak L. Effectiveness and safety of hypotension fluid resuscitation in traumatic haemorrhagic shock: a systematic review and meta-analysis of randomised controlled trials. Cardiol J. 2020. Preprint.https://www.ncbi.nlm.nih.gov/pubmed/32648249.
89. Wang C, Hsieh W, Chou H, Huang Y, Shen J, Yeo YH, Chang H, Chen S, Lee C. Liberal versus restricted fluid resuscitation strategies in trauma patients: a systematic review and meta-analysis randomised controlled trials and observational studies. Crit Care Med. 2014;42(4):954–61.
90. Guyette FX, Sperry JL, Peitzman AB, Billiar TR, Daley BJ, Miller RS, Harbrecht BG, Claridge JA, Putnam T, Duane TM, et al. Prehospital blood
product and crystalloid resuscitation in the severely injured patient: a secondary analysis of the prehospital air medical plasma trial. Ann Surg. 2021;273(2):358–64
91. Albreiki M, Voegeli D. Permissive hypotensive resuscitation in adult patients with traumatic haemorrhagic shock: a systematic review. Eur J Trauma Emerg Surg. 2018;44(2):191–202.
92. Owattanapanich N, Chittawatanarat K, Benyakorn T, Sirikun J. Risks and benefits of hypotensive resuscitation in patients with traumatic haemorrhagic shock: a meta-analysis. Scand J Trauma Resusc Emerg Med. 2018;26(1):107.
93. Joseph B, Azim A, Zangbar B, Bauman Z, O’Keefe T, Ibraheem K, Kulvatunyou N, Tang A, Latif R, Rhee P. Improving mortality in trauma laparotomy through the evolution of damage control resuscitation: analysis of 1030 consecutive trauma laparotomies. J Trauma Acute Care Surg. 2017;82(2):328-33.
94. Fischer NJ, Civil ID. Haemorrhagic death from severe liver trauma has decreased in the era of haemostatic resuscitation. ANZ J Surg. 2022;92(1–2):188–94.
95. Kasotakis G, Sideris A, Yang Y, de Moya M, Alam H, King DR, Tompkins R, Velmahos G. Infammation and host response to injury investigators: aggressive early crystalloid resuscitation adversely affects outcomes in adult blunt trauma patients: an analysis of the glue grant database. J Trauma Acute Care Surg. 2013;74(5):1215–21 (discussion 1221-1212).
96. Mbadiwe N, Georgette N, Slidell MB, McQueen A. Higher crystalloid volume during initial pediatric trauma resuscitation is associated with mortality. J Surg Res. 2021;262:93–100.
97. Lou X, Lu G, Zhao M, Jin P. Preoperative fluid management in traumatic shock: a retrospective study for identifying optimal therapy of fluid resuscitation for aged patients. Med (Baltimore). 2018;97(8): e9966.
98. Barmparas G, Dhillon NK, Smith EJ, Mason R, Melo N, Thomsen GM, Margulies DR, Ley EJ. Patterns of vasopressor utilization during the resuscitation of massively transfused trauma patients. Injury. 2018;49(1):8–14.
99. Aoki M, Abe T, Saitoh D, Hagiwara S, Oshima K. Use of vasopressor increases the risk of mortality in traumatic haemorrhagic shock: a nationwide cohort study in Japan. Crit Care Med. 2018;46(12):e1145–51.
100. Uchida K, Nishimura T, Hagawa N, Kaga S, Noda T, Shinyama N, Yama‑moto H, Mizobata Y. The impact of early administration of vasopressor agents for the resuscitation of severe haemorrhagic shock following blunt trauma. BMC Emerg Med. 2020;20(1):26.
101. Fisher AD, April MD, Cunningham C, Schauer SG. Prehospital vasopressor use is associated with worse mortality in combat wounded. Prehosp Emerg Care. 2021;25(2):268–73.
102. Gauss T, Gayat E, Harrois A, Raux M, Follin A, Daban JL, Cook F, Hamada S. TraumaBase group, prehospital traumabase group Ile de France SAMU=service d’Aide Médicale Urgente: effect of early use of noradrenaline on in‑hospital mortality in haemorrhagic shock after major trauma: a propensity‑score analysis. Br J Anaesth. 2018;120(6):1237–44.
103. Van Haren RM, Thorson CM, Valle EJ, Guarch GA, Jouria JM, Busko AM, Namias N, Livingstone AS, Proctor KG. Vasopressor use during emergency trauma surgery. Am Surg. 2014;80(5):472–8.
104. Hylands M, Toma A, Beaudoin N, Frenette AJ, D’Aragon F, Belley‑Côté É, Charbonney E, Møller MH, Laake JH, Vandvik PO, et al. Early vasopressor use following traumatic injury: a systematic review. BMJ Open. 2017;7(11): e017559.
105. Richards JE, Harris T, Dünser MW, Bouzat P, Gauss T. Vasopressors in trauma: A never event? Anesth Analg. 2021;133(1):68–79.
106. Sims CA, Holena D, Kim P, Pascual J, Smith B, Martin N, Seamon M, Shiroff A, Raza S, Kaplan L, et al. Effect of low‑dose supplementation of arginine vasopressin on need for blood product transfusions in patients with trauma and haemorrhagic shock: a randomised clinical trial. JAMA Surg. 2019;154(11):994–1003.
107. Cohn SM, McCarthy J, Stewart RM, Jonas RB, Dent DL, Michalek JE. Impact of low‑dose vasopressin on trauma outcome: prospective randomised study. World J Surg. 2011;35(2):430–9.
108. Semler MW, Kellum JA. Balanced crystalloid solutions. Am J Respir Crit Care Med. 2019;199(8):952–60.
109. Semler MW, Self WH, Wanderer JP, Ehrenfeld JM, Wang L, Byrne DW, Stollings JL, Kumar AB, Hughes CG, Hernandez A, et al. Balanced crystalloids versus saline in critically ill adults. N Engl J Med. 2018;378(9):829–39.
110. Zampieri FG, Machado FR, Biondi RS, Freitas FGR, Veiga VC, Figueiredo RC, Lovato WJ, Amêndola CP, Serpa‑Neto A, Paranhos JLR, et al. Effect of intravenous fluid treatment with a balanced solution vs 0.9% saline solution on mortality in critically ill patients: the BaSICS trial. JAMA. 2021;326(9):1–12.
111. Antequera Martín AM, Barea Mendoza JA, Muriel A, Sáez I, Chico Fernández M, Estrada‑Lorenzo JM, Plana MN. Buffered solutions versus 0.9% saline for resuscitation in critically ill adults and children. Cochrane Database Syst Rev. 2019;7:CD012247.
112. Zhu Y, Guo N, Song M, Xia F, Wu Y, Wang X, Chen T, Yang Z, Yang S, Zhang Y, et al. Balanced crystalloids versus saline in critically ill patients: PRISMA meta‑analysis. Medicine (Baltimore). 2021;100(38):e27203.
113. Kind SL, Spahn‑Nett GH, Emmert MY, Eismon J, Seifert B, Spahn DR, Theusinger OM. Is dilutional coagulopathy induced by different colloids reversible by replacement of fibrinogen and factor XIII concentrates? Anesth Analg. 2013;117(5):1063–71.
114. Groene P, Wiederkehr T, Kammerer T, Möhnle P, Maerte M, Bayer A, Görlinger K, Rehm M, Schäfer ST. Comparison of two different fibrinogen concentrates in an in vitro model of dilutional coagulopathy. Transfus Med Hemother. 2020;47(2):167–74.
115. Rowell SE, Fair KA, Barbosa RR, Watters JM, Bulger EM, Holcomb JB, Cohen MJ, Rahbar MH, Fox EE, Schreiber MA. Impact of prehospital administration of lactated Ringer’s vs normal saline in traumatic brain injury. J Neurotrauma. 2016;33(11):1054–9.
116. Kochanek, M., & Schött, D. (2014). Effects of Hydroxyethyl Starch Solutions on Hemostasis. Journal of Vascular and Interventional Radiology, 25(4), 654–657.
117. Kozen-Langenecker, S. A. (2005). Effects of colloid solution on haemostasis. Best Practice & Research Clinical Anaesthesiology, 19(3), 443–456
118. Pérez-Guzmán, C., Vivas-Balcones, A. E., & Díaz-Domínguez, E. (2021). Intravenous Fluid Administration and the Coagulation System. Frontiers in Veterinary Science, 8. Article 662504.
119. Polotsky, A., Schött, U., & Winstedt, D. (2013). Is dilutional coagulopathy induced by different colloids reversible by replacement of fibrinogen and factor XIII concentrates? An in vitro trial. Anesthesia & Analgesia, 117(5), 1061–1068.
120. Trotman, C. R., & Sandoe, J. R. (2021). Synthetic colloids: Useful tool or potential harm? MSPCA-Angell. Retrieved from https://www.mspca.org/angell_services/synthetic-colloids-useful-tool-or potential-harm/
121. Winstedt, D., Hanna, J., & Schött, U. (2014). Albumin-induced coagulopathy is less severe and more effectively reversed with fibrinogen concentrate than is synthetic colloid-induced coagulopathy. Anesthesiology and Intensive Care, 42(7), 659–665
122. TRICC Trial (The foundation for a restrictive strategy): Hébert, P. C., Wells, G., Blajchman, M. A., Marshall, J., Martin, C., Pagliarello, G., Tweeddale, M., Schweitzer, I., & Yetisir, E. (1999). A multicenter, randomized, controlled clinical trial of transfusion requirements in critical care. New England Journal of Medicine, 340(6), 409-417.
123. Meta-Analysis (Confirming safety across multiple settings): Holst, L. B., Haase, N., Wetterslev, J., Wernerman, J., Guttormsen, A. B., Karlsson, S., Zhukovsky, I., Aneman, A., Johansson, P. I., Rasmussen, B. S., Siegemund, M., Lorenzen, E. L., Krag, M., Bäcklund, M., Perner, A., & Kjaergard, H. (2015). Restrictive versus liberal transfusion strategy for red blood cell transfusion: systematic review of randomised trials with meta-analysis and trial sequential analysis. BMJ, 350, h1354.
124. European Guidelines (Application in Trauma/Hemorrhage): Rossaint, R., Afshari, A., Bouillon, B., Cerny, V., Cimpoesu, D., Curry, N., Duranteau, J., Filipescu, D., Grottke, O., Grønlykke, L., Harrois, A., Hunt, B. J., Kaserer, A., Komadina, R., Madsen, M. H., Maegele, M., Mora, L., Riddez, L., Romero, C. S., ... Spahn, D. R. (2023). The European guideline on management of major bleeding and coagulopathy following trauma: sixth edition. Critical Care, 27(1), 80.
125. Jurkovich GJ, Greiser WB, Luterman A, Curreri PW. Hypothermia in trauma victims: an ominous predictor of survival. J Trauma.1987;27(9):1019–24.
126. Lester ELW, Fox EE, Holcomb JB, Brasel KJ, Bulger EM, Cohen MJ, Cotton BA, Fabian TC, Kerby JD, O’Keefe T, et al. The impact of hypothermia on outcomes in massively transfused patients. J Trauma Acute Care Surg.2019;86(3):458–63.
127. Duque P, Calvo A, Lockie C, Schochl H. Pathophysiology of trauma-induced coagulopathy. Transfus Med Rev. 2021;35(4):80–6.
128. Reynolds BR, Forsythe RM, Harbrecht BG, Cuschieri J, Minei JP, Maier RV, Moore EE, Billiar EE, Peitzman AB, Sperry JL, et al. Hypothermia in massive transfusion: Have we been paying enough attention to it? J Trauma Acute Care Surg. 2012;73(2):486–91.
129. Rubiano AM, Sanchez AI, Estebanez G, Peitzman A, Sperry J, Puyana JC. The effect of admission spontaneous hypothermia on patients with severe traumatic brain injury. Injury. 2013;44(9):1219–25.
130. Cheln H, Wu F, Yang P, Shao J, Chen Q, Zheng R. A meta-analysis of the effects of therapeutic hypothermia in adult patients with traumatic brain injury. Crit Care. 2019;23(1):396.
131. Bennett BL, Giesbrect G, Zafren K, Christensen R, Littlejohn LF, Drew B, Cap AP, Miles EA, Butler FK Jr, Holcomb JB, et al. Management of hypothermia in tactical combat casualty care: TCCC guideline proposed change 20–01 (June 2020). J Spec Oper Med. 2020;20(3):21–35.
- Rotondo MF, Schwab CW, McGonigal MD, Phillips GR 3rd, Fruchterman TM, Kauder DR, Latenser BA, Angood PA. Damage control: an approach for improved survival in exsanguinating penetrating abdominal injury. J Trauma. 1993;35(3):375–82.
- Roberts DJ, Ball CG, Feliciano DV, Moore EE, Ivatury RR, Lucas CE, Fabian TC, Zygun DA, Kirkpatrick AW, Stelfox HT. History of the innovation of damage control for management of trauma patients: 1902–2016. Ann Surg. 2017;265(5):1034–44.
- Duchesne JC, McSwain NE Jr, Cotton BA, Hunt JP, Dellavolpe J, Lafaro K, Marr AB, Gonzalez EA, Phelan HA, Bilski T, et al. Damage control resuscitation: the new face of damage control. J Trauma. 2010;69(4):976–90.
- Roberts DJ, Bobrovitz N, Zygun DA, Kirkpatrick AW, Ball CG, Faris PD, Stelfox HT. Indications for trauma damage control surgery international study group: evidence for use of damage control surgery and interventions in civilian trauma patients: a systematic review. World J Emerg Surg. 2021;16(1):10.
- Cullinane DC, Schiller HJ, Zielinski MD, Bilaniuk JW, Collier BR, Como J, Holevar M, Sabater EA, Sems SA, Vassy WM, et al. Eastern Association for the Surgery of Trauma practice management guidelines for haemorrhage in pelvic fracture-update and systematic review. J Trauma.2011;71(6):1850–68.
137. Höch A, Zeidler S, Pieroh P, Josten C, Stuby FM, Herath SC. German pelvic trauma registry: trends and efcacy of external emergency stabilisation of pelvic ring fractures: results from the German Pelvic Trauma Registry. Eur J Trauma Emerg Surg. 2021;47(2):523–31.
138. Naseem H, Nesbitt PD, Sprott DC, Clayson A. An assessment of pelvic binder placement at a UK major trauma centre. Ann R Coll Surg Engl. 2018;100(2):101–5.
139. Audretsch CK, Mader D, Bahrs C, Trulson A, Höch A, Herath SC, Küper
MA. Working group on pelvic fractures of the German Trauma Society:
comparison of pelvic C-clamp and pelvic binder for emergency stabi‑
lisation and bleeding control in type-C pelvic ring fractures. Sci Rep.
2021;11(1):2338.
140. Winstanley M, Smith JE, Wright C. Catastrophic haemorrhage in military trauma: retrospective analysis of haemostatic agents. J R Army Med Corps. 2019;165(6):405–9.
- Pursifull NF, Morris MS, Harris RA, Morey AF. Damage control management of experimental grade 5 renal injuries: further evaluation of FloSeal gelatin matrix. J Trauma. 2006;60(2):346–50.
- Schenk WG 3rd, Burks SG, Gagne PJ, Kagan SA, Lawson JH, Spotnitz WD. Fibrin sealant improves haemostasis in peripheral vascular surgery: a randomised prospective trial. Ann Surg. 2003;237(6):871–6.
- Sherman R, Chapman WC, Hannon G, Block JE. Control of bone bleeding at sternum and iliac crest donor sites using a collagen‑based composite with autologous plasma: RCT results. Orthopedics. 2001;24(2):137–41.
- Testini M, Marzaioli R, Lissidini G, Lippolis A, Logoluso F, Gurrado A, Lardo D, Poli E, Piccinni G. Effectiveness of FloSeal matrix haemostatic agent in thyroid surgery: prospective RCT. Langenbecks Arch Surg. 2009;394(5):837–42.
145. Weaver FA, Hood DB, Zatina M, Messina L, Badduke B. Gelatin‑thrombin haemostatic sealant for intraoperative bleeding in vascular surgery. Ann Vasc Surg. 2002;16(3):286–93.
146. Witte B, Kroeber SM, Hillebrand H, Wolf M, Huertgen M. Cotton‑derived oxidised cellulose in minimally invasive thoracic surgery: clinico‑pathological study. Innovations (Phila). 2013;8(4):296–301.
147. Woodworth BA, Chandra RK, LeBenger JD, Ilie B, Schlosser RJ. Gelatin‑thrombin matrix for haemostasis after endoscopic sinus surgery. Am J Otolaryngol. 2009;30(1):49–53.
148. Welch M, Barratt J, Peters A, Wright C. Systematic review of prehospital haemostatic dressings. BMJ Mil Health. 2020;166(3):194–200.
149. Choron RL, Hazelton JP, Hunter K, Capano‑Wehrle L, Gaughan J, Chovanes J, Seamon MJ. Intra‑abdominal packing with laparotomy pads and QuikClot™ during damage control laparotomy: safety analysis. Injury. 2017;48(1):158–64.
150. CRASH‑2 trial collaborators, Shakur H, Roberts I, Bautista R, Caballero J, Coats T, Dewan Y, El‑Sayed H, Gogichaishvili T, Gupta S, et al. Effects of tranexamic acid on death, vascular occlusive events, and transfusion in trauma patients with haemorrhage (CRASH‑2): RCT. Lancet. 2010;376(9734):23–32.
151. CRASH‑3 trial collaborators. Effects of tranexamic acid on death, disability, vascular occlusive events and morbidities in acute TBI (CRASH‑3): RCT. Lancet. 2019;394(10210):1713–23.
152. Rowell SE, Meier EN, McKnight B, Kannas D, May S, Sheehan K, Bulger EM, Idris AH, Christenson J, Morrison LJ, et al. Out‑of‑hospital TXA vs placebo on 6‑month neurologic outcomes in moderate/severe TBI. JAMA. 2020;324(10):961–74.
153. Mojallal F, Nikooieh M, Hajimaghsoudi M, Baqherabadi M, Jafari M, Esmaeili A, Karimi NM, Zarepur E. IV TXA preventing cerebral haemorrhage progression in TBI: RCT. Med J Islam Repub Iran. 2020;34:107.
154. Fakharian E, Abedzadeh‑Kalahroudi M, Atoof F. TXA effect on prevention of haemorrhagic mass growth in TBI. World Neurosurg. 2018;109:e748–53.
155. Li SR, Guyette F, Brown J, Zenati M, Reitz KM, Eastridge B, Nirula R, Vercruysse GA, O’Keeffe T, Joseph B, et al. Early prehospital tranexamic acid following injury is associated with a 30‑day survival benefit: a secondary analysis of a randomised clinical trial. Ann Surg. 2021;274(3):419–26.
156. Holcomb JB, Tilley BC, Baraniuk S, Fox EE, Wade CE, Podbielski JM, del Junco DJ, Brasel KJ, Bulger EM, Callcut RA, et al. Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs a 1:1:2 ratio and mortality in patients with severe trauma: the PROPPR randomised clinical trial.JAMA. 2015;313(5):471–82.
157. Meneses E, Boneva D, McKenney M, Elkbuli A. Massive transfusion protocol in adult trauma population. Am J Emerg Med. 2020;38(12):2661–6.
158. Kragh JF Jr, O’Neill ML, Walters TJ, Jones JA, Baer DG, Gershman LK, Wade CE, Holcomb JB. Minor morbidity with emergency tourniquet use to stop bleeding in severe limb trauma: research, history, and reconciling advocates and abolitionists. Mil Med. 2011;176(7):817–23.
159. Levi M, Eerenberg E, Kamphuisen PW. Bleeding risk and reversal strategies for old and new anticoagulants and antiplatelet agents. J Thromb Haemost. 2011;9(9):1705–12.
160. Dowlatshahi D, Butcher KS, Asdaghi N, Nahirniak S, Bernbaum ML, Giulivi A, Wasserman JK, Poon MC, Coutts SB. Canadian PCC registry (CanPro) investigators: poor prognosis in warfarin-associated Rossaintet al. Critical Care (2023) 27:80 Page 44 of 45 intracranial haemorrhage despite anticoagulation reversal. Stroke. 2012;43(7):1812–7
161. Steiner T, Poli S, Griebe M, Hüsing J, Hajda J, Freiberger A, Bendszus M, Bösel J, Christensen H, Dohmen C, et al. Fresh frozen plasma versus prothrombin complex concentrate in patients with intracranial haemorrhage related to vitamin K antagonists (INCH): a randomised trial. Lancet Neurol. 2016;15(6):566–73.