Diagnosis of Neonatal and Infantile Cholestasis
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | Diagnosis of Neonatal and Infantile Cholestasis |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 24 Feb 2026" Download Guideline
- Executive Summary
Cholestasis is defined as an impairment in bile formation and/or flow (with all its constituents). Neonatal and infantile cholestasis (NIC) has diverse set of etiologies, some of which are treatable while others are not. Its clinical presentation also varies from a self-limited illness to acute liver failure. Timely detection, being often recognized in our health care settings late, identification of the etiology, particularly treatable ones, and proper management are crucial for patient outcome. The most common recognizable cause of NIC is biliary atresia (BA), in which an early surgical referral is needed to optimize the success rate. A timely diagnosis also allows for prompt specific treatment and a better outcome in other treatable inherited metabolic conditions, such as galactosemia or the rare inborn errors of bile acid synthesis.
This guideline focuses on diagnosing neonatal and infantile cholestasis in Egypt. It provides evidence-based guidance to help in early detection of NIC and in the diagnosis of its etiology.
After revising inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guideline (CPG):
- Ranucci G, Della Corte C, Alberti D, Bondioni MP, Boroni G, Calvo PL, Cananzi M, Candusso M, Clemente MG, D'Antiga L, Degrassi I, De Ville De Goyet J, Di Dato F, Di Giorgio A, Vici CD, Ferrari F, Francalanci P, Fuoti M, Fusaro F, Gaio P, Grimaldi C, Iascone M, Indolfi G, Iorio R, Maggiore G, Mandato C, Matarazzo L, Monti L, Mosca F, Nebbia G, Nuti F, Paolella G, Pinon M, Roggero P, Sciveres M, Serranti D, Spada M, Vajro P, Nicastro E. Diagnostic approach to neonatal and infantile cholestasis: A position paper by the SIGENP liver disease working group. Dig Liver Dis. 2022 Jan;54(1):40-53. doi: 10.1016/j.dld.2021.09.011. Epub 2021 Oct 20. PMID: 34688573.
We conducted an adolopment for this position paper (Adoption, Adaptation, and Development)
- Adoption for most of the recommendations.
- Adaptation for 2 recommendations according to GRADE criteria to be suitable to our Economic implications (Evidence-to-Decision (EtD) table was done)
- Development of Good Practice Statements
This version of the CPG includes recommendations and good practice statements on the following sub-sections:
A. Definition and Etiology:
Neonatal/infantile cholestasis (NIC) is defined as an impairment in bile formation and/or flow, presenting during the first year of life—usually within the first three months—and resulting in retention of bile and biliary substances within the liver that cause liver damage. Strength of recommendation: Strong
Serum conjugated bilirubin greater than 1 mg/dl (17 μmol/L) when total bilirubin is less than 5 mg/dl, or greater than 20% of total bilirubin when total bilirubin exceeds 5 mg/dl, is considered diagnostic of cholestasis and warrants further evaluation. Strength of recommendation: Strong
Once cholestasis has been documented in an infant, achieving a rapid etiological definition is of primary importance. Given the wide spectrum of potential causes, a complex diagnostic approach is required, guided by the accompanying clinical picture. For practical purposes, causes of cholestasis are divided into surgical and medical conditions. Strength of recommendation: Good practice statement
B. Emergency treatment of Neonatal and Infantile cholestasis
In jaundiced infants with suspected or confirmed cholestasis, a detailed pre‑, peri‑, and postnatal medical and family history should be obtained. Strength of recommendation: Strong
Infants with jaundice should receive a complete physical examination, in particular searching for the presence of hepatomegaly and/or splenomegaly, dysmorphic features, growth and nutritional status, skin lesions, cardiac murmurs, and neurologic examination. Strength of recommendation: Strong
Direct visualization of stool color and its monitoring should be part of the clinical evaluation of any infant with jaundice. Strength of recommendation: Strong
Infants with jaundice persisting after 2 weeks of age should be evaluated for cholestasis by measurements of serum total and direct reacting (conjugated) bilirubin. Strength of recommendation: Strong
Upon evaluating causes of cholestasis, a stepwise approach is important to rule out first diseases with available treatment such as sepsis, congenital hypothyroidism, galactosemia, and surgical causes including biliary atresia and choledochal cyst. Strength of recommendation: Strong
In infants with cholestasis, inborn errors of metabolism (IEM) should always be considered in the differential diagnosis. Clinicians should be aware of which IEM causing cholestasis are included in the expanded newborn screening of their own region or country. Strength of recommendation: Strong
A careful clinical evaluation, combined with first‑level laboratory testing, may raise the suspicion of specific IEM, indicating second‑level laboratory analyses for confirmatory diagnosis. Strength of recommendation: Strong
Infants with cholestasis should undergo liver ultrasound to exclude biliary obstructive conditions other than biliary atresia, such as choledochal malformations or gallstone disease. Strength of recommendation: Strong
Findings such as the absence of the gallbladder or its dysmorphic aspect, along with other minor features suggestive of biliary atresia, should always be searched for at liver ultrasound. Strength of recommendation: Strong
Endoscopic or direct surgical exploration and evaluation of the biliary tract by cholangiogram is considered the gold standard to positively and definitively establish the diagnosis of biliary atresia (as well as neonatal sclerosing cholangitis). Strength of recommendation: Strong
Magnetic Resonance Cholangio‑Pancreatography (MRCP) is not reliable as the normal biliary tract is not consistently visualized in children under 3 months of age; false negative results are thus common. Strength of recommendation: Strong
Hepatobiliary scintigraphy is not recommended anymore. Strength of recommendation: Strong
Liver biopsy is useful to discriminate between extrahepatic and intrahepatic causes of NIC and may provide important clues for specific underlying etiologies, when performed at the appropriate timing and evaluated by an experienced pathologist. Liver biopsy should only be done after consultation with a pediatric hepatologist. Strength of recommendation: Strong / Good practice statement
Infants with cholestatic liver disease may present acutely with complications such as bleeding due to vitamin K deficiency. Indicators of complications in cholestatic neonates and infants include episodes of acute metabolic decompensation (hypoglycemia, hyperammonemia, acidosis, or acute liver failure), recurrent or chronic vomiting, neurological signs, multi‑organ involvement, ill appearance suggesting infection or metabolic disease, lethargy, and poor feeding. These are managed in parallel to the rapid diagnostic work‑up. Strength of recommendation: Strong / Good practice statement
At first encounter with a cholestatic infant (proven or suspected), 2 mg Vitamin K1 should be given intramuscularly. Strength of recommendation: Good practice statement
All age‑appropriate immunizations should be given to infants with cholestasis if there are no other contraindications. Strength of recommendation: Good practice statement
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: ((submitted and in process)). Link: http://www.guidelines-registry.org/
- Recommendations
|
Table 3. Recommendations |
|
|
|||
|
A. Definition and etiologies |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
A1
|
What is the definition of neonatal cholestasis?
|
(2) |
1-Neonatal/ Infantile cholestasis (NIC) is defined as an impairment in bile formation and/or flow (with all its constituents) presenting during the first year of life, usually in the first three months, and resulting in the retention of bile and biliary substances within the liver that cause liver damage.
2- Serum conjugated bilirubin > 1mg/dl (17 mmol/L) if total bilirubin is < 5 mg/dl, or > 20% if total bilirubin > 5 mg/dl, is considered diagnostic of cholestasis and warrants further evaluation.
|
High
|
Good practice statement
Strong
|
|
A2 |
What are the causes of cholestasis? |
(1) |
3- Once cholestasis has been documented in an infant, achieving a rapid etiological definition is of primary importance. The wide spectrum of causes potentially underlying NIC entails a complex diagnostic approach that depends on the accompanying clinical picture. For practical purposes, causes of cholestasis are divided into surgical and medical conditions. |
|
Good practice statement
|
|
Table 4. Recommendations |
|
|
|||
|
B. Diagnosis and emergency treatment |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
B1 |
What is the diagnostic approach to different etiologies of cholestasis in neonates and infants?
|
(1) |
4- In jaundiced infants with suspected or confirmed cholestasis a detailed pre-, peri-, and postnatal medical and family history should be obtained. |
Moderate
|
Strong
|
|
5- Infants with jaundice should receive a complete physical examination in particular searching for the presence of hepatomegaly and / or splenomegaly, dysmorphic features, growth and nutritional status, skin lesions, cardiac murmurs and neurologic examination. |
High
|
Strong
|
|||
|
6- Direct visualization of stools color and its monitoring should be part of the clinical evaluation of any infant with jaundice. |
High
|
Strong
|
|||
|
7- Infants with jaundice persisting after 2 weeks of age should be evaluated for cholestasis by measurements of serum total and direct reacting (conjugated) bilirubin. |
High
|
Strong
|
|||
|
8- Upon evaluating causes of cholestasis, a stepwise approach is important to rule out first diseases with available treatment as sepsis, congenital hypothyroidism, galactosemia and surgical causes as biliary atresia and choledochal cyst |
Moderate |
Strong
|
|||
|
9- In infants with cholestasis, inborn errors of metabolism (IEM) should always be considered in the differential diagnosis. Clinicians should be aware of which IEM causing cholestasis is included in the expanded newborn screening of their own region/country. |
High
|
Strong
|
|||
|
10- A careful clinical evaluation, combined with first-level laboratory testing, may raise the suspicion of specific IEM, indicating second level laboratory analyses for confirmatory diagnosis |
High
|
Strong
|
|||
|
B2 |
What is The Role of Ultrasound in Diagnosis of Cholestasis?
|
|
11- Infants with cholestasis should undergo liver ultrasound to exclude biliary obstructive conditions other than biliary atresia, such as choledochal malformations or gallstone disease. 12- Findings such as the absence of the gallbladder or its dysmorphic aspect, along with other minor features suggestive of biliary atresia, should always be searched for at liver ultrasound. |
High
High
|
Strong
Strong
|
|
B3 |
What is The Role of other imaging in Diagnosis of Cholestasis?
|
(1)
|
13- Endoscopic or direct surgical exploration and evaluation of biliary tract by cholangiogram is considered the gold standard to positively and definitively establish the diagnosis of biliary atresia (as well as neonatal sclerosing cholangitis) 14-Magnetic Resonance Cholangio-Pancreatography (MRCP) is not reliable as the normal biliary tract is not consistently visualized in children under 3 months of age; false negative results are thus common. 15-Hepatobiliary scintigraphy is not recommended anymore. |
High
High
|
Strong
Strong
|
|
B4 |
When to Refer a Cholestatic Neonate or Infant for Liver Biopsy?
|
(1)
|
16- Liver biopsy is useful to discriminate between extrahepatic and intrahepatic causes of NIC and may provide important clues for specific underlying etiologies, when performed at the appropriate timing and evaluated by an experienced pathologist. Liver biopsy should be only done after the consultation of a pediatric hepatologist. |
Moderate
|
Strong
Good practice statement
|
|
B5 |
How to Detect Complications in Cholestatic Neonates & Infants?
|
(1)
|
17- Infants with Cholestatic liver disease may present acutely with complications such as bleeding due to vitamin K deficiency. 18- The following could be indicators of complications in cholestatic neonates and infants: - Episodes of acute metabolic decompensation such as hypoglycemia, hyperammonemia, acidosis, or acute liver failure/involvement. - Recurrent/chronic vomiting, - Neurological signs. - Multi-organ involvement. - Ill appearance may indicate infection or metabolic disease - Lethargy, and poor feeding. These are managed in parallel to the rapid diagnostic work-up. |
High
|
Good practice statement
Strong
|
|
B6 |
What is The Emergency Treatment of Cholestasis In Neonates And Infants? |
(10)
|
19- At first encounter with cholestatic infant (proven or suspected) 2 mg Vitamin K1 should be given intramuscularly |
|
Good practice statement
|
|
B7 |
What about vaccination of cholestatic neonates and infants? |
(11) |
All age-appropriate immunizations should be given to infants with cholestasis if there are no other contraindications.
|
|
Good practice statement
|
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location |
Contribution |
|||
|
Prof. Mortada El-Shabrawi |
Professor of pediatrics, pediatric hepatology, Cairo University, Egypt |
Supervise and coordinate the guidelines development process, share in collecting published guidelines from databases, attending online meetings of GDG, sharing in writing up the manuscript and revising its final draft. |
|||
|
Prof. Tawhida Yassin Abdel Ghaffar |
Professor of pediatrics, pediatric hepatology, Ain Shams University, Egypt |
Supervise and share in collecting published guidelines from databases, attending online meetings of GDG, sharing in writing up the manuscript and revising its final draft. |
|||
|
Prof. Nehal El Koofy |
Professor of pediatrics, pediatric hepatology and nutrition, Cairo University, Egypt |
Collecting published guidelines from databases, attending online meetings of GDG, sharing in writing up the manuscript and revising its final draft. |
|||
|
Prof Lerine Bahy |
Professor of pediatrics, pediatric hepatology, Ain Shams University, Egypt |
Collecting published guidelines from databases, attending online meetings of GDG, sharing in writing up the manuscript and revising its final draft. |
|||
|
Prof, Manal Abd El Gawad |
Professor of pediatrics, Alexandria University, Egypt |
Collecting published guidelines from databases, attending online meetings of GDG, sharing in writing up the manuscript and revising its final draft. |
|||
|
Prof Nagla Abu Faddan |
Professor of pediatrics, pediatric hepatology, Assiut University, Egypt |
Collecting published guidelines from databases, attending online meetings of GDG, sharing in writing up the manuscript and revising its final draft. |
|||
|
Prof. Hanaa Elaraby |
Professor of pediatrics, pediatric hepatology, National Liver Institute, Menoufia University, Egypt |
Collecting published guidelines from databases, attending online meetings of GDG, sharing in writing up the manuscript and revising its final draft. |
|||
|
Prof. Ahmed Megahed |
Professor of pediatrics, pediatric gastroenterology and hepatology, Mansoura university, Egypt. |
Collecting published guidelines from databases, attending online meetings of GDG, sharing in writing up the manuscript and revising its final draft. |
|||
|
Ass. Prof. Mohammad EL Adawy |
Ass. Prof. of pediatrics, pediatric hepatology, Ain Shams University, Egypt |
Collecting published guidelines from databases, attending online meetings of GDG, sharing in writing up the manuscript and revising its final draft. |
|||
|
Ass. Prof. Hanan Mina |
Ass. Professor of pediatrics, pediatric hepatology, Helwan university, Egypt. |
Collecting published guidelines from databases, attending online meetings of GDG, shares also in the guidelines appraisal, and she was the main organizer of the guidelines draft, and revising its final draft. |
|||
|
Dr. Sherif Baroudy |
Lecturer of pediatrics, pediatric hepatology, Cairo University, Egypt |
Collecting published guidelines from databases, attending online meetings of GDG, sharing in writing up the manuscript and revising its final draft. |
|||
|
Prof Gamal EL Tagy |
Professor of pediatric surgery, Cairo University, Egypt |
Attending online meetings of GDG, sharing in writing up the manuscript and revising its final draft. |
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
1. Pediatrics department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Associate Professor of pediatrics Ain Shams University, Egypt |
Developing evidence to decision (EtD) frameworks, participating in search and guideline appraisal |
||||
|
- Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. - Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. - Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
||||
|
Dr. Lamis Mohsen Elsholkamy
|
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
Dr. Ahmed Mahmoud Youssef
|
Fellow of Pediatrics, General Organization For Teaching Hospitals and Institutes (GOTHI). Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
Dr. Mona Saber |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Prof. Ahmed Abdallah |
Prof. of pediatrics, pediatric hepatology, Mansoura University |
||||
|
Prof. Mona El Raziky |
Prof. of pediatrics, pediatric hepatology, Cairo University |
||||
|
Prof. Ahmed Sira |
Prof. of pediatrics, pediatric hepatology, National Liver Institute, Menoufia University. |
||||
|
International Peer Reviewers |
|||||
|
Prof Lorenzo D'Antiga |
University of Milano – Bicocca. Child Health Department. Paediatric Hepatology, Gastroenterology and Transplantation Hospital Papa Giovanni XXIII, Bergamo, Italy Piazza Oms 1, 24127, Bergamo. Italy ERN TransplantChild, ERN Rare Liver, ERN ERNICA |
||||
|
Prof. Anil Dhawan |
Prof of Paediatric at Liver GI and Nutrition Center and MowatLabs. Director Research and Innovation. King’s College Hospital. London |
||||
|
Prof. Hesham Hassan |
Emeritus Professor of Pediatrics University of Arizona, USA |
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
||||
· The GDG/ GAG acknowledge EPG for its help in completing this project.
· We acknowledge the SIGENP liver disease working group (Diagnostic approach to neonatal and infantile cholestasis, 2022), (the source original position paper) for their cooperation in providing the permission for adapting our guidelines.
· Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.- Abbreviations
|
Adolopment |
Adoption-Adaptation-Development |
|
AGREE II |
Appraisal of Guidelines for Research and Evaluation Instrument |
|
ALT |
Alanine aminotransferase |
|
ALP |
alkaline phosphatase |
|
AST |
Aspartate aminotransferase |
|
BA |
Biliary atresia |
|
BASM |
Biliary atresia with splenic malformations |
|
CBD |
Common bile duct |
|
CM |
Choledochal malformation |
|
CMV |
Cytomegalovirus |
|
CPG/ CPGs |
Clinical Practice Guideline (s) |
|
DHS |
Demographic and Health Survey |
|
ERCP |
Endoscopic retrograde cholangiopancreatography |
|
EPG |
Egyptian Pediatrics Clinical Practice Guidelines Committee |
|
EPG CPG |
EPG Clinical Practice Guideline |
|
ERG |
External Review Group |
|
GAG |
Guideline Adaptation Group |
|
GALD |
Gestational alloimmune liver disease |
|
GB |
Gall bladder |
|
GDG |
Guideline Development Group |
|
GGT |
Gamma-glutamyl transferase |
|
GPS |
Good Practice Statement |
|
GRADE |
Grading of Recommendations Assessment, Development and Evaluation |
|
HIV |
Human Immunodeficiency virus |
|
IEM |
Inborn errors of metabolism |
|
MRCP |
Magnetic resonance cholangiopancreatography |
|
NIC |
Neonatal and infantile cholestasis |
|
PFIC |
Progressive familial intrahepatic cholestasis |
|
PIBD |
Paucity of intrelobular bile ducts |
|
PICO |
Population, intervention, comparison, and outcomes |
|
PIPOH |
Patient population, intervention, professionals, outcomes, and healthcare context |
|
PNALD |
Parenteral nutrition-associated cholestasis |
|
PT |
Prothrombin time |
|
RIGHT |
A Reporting Tool for Practice Guidelines in Health Care |
|
SCC |
Stool color card |
|
TSH |
Thyroid stimulating hormone |
- Introduction
➡️Definition and causes (1-4)
Neonatal and infantile cholestasis (NIC) is defined as impaired bile formation, flow, or both and usually manifests in the first year of life, particularly in the first three months. Its clinical presentation varies from self-limited illness to acute liver failure. It results in hepatic retention of bile and biliary substances, causing severe liver damage.
The prevalence of NIC in infants is 1 out of 2,500. Its causes are due to either surgical or medical disorders. Such differentiation is crucial for the proper management and a better outcome. If biliary atresia (BA) is the cause, early referral for surgical intervention is the key to successful management. Timely diagnosis and management of treatable metabolic conditions, such as galactosemia, provide a better outcome.
Classically, infants present with prolonged jaundice (conjugated), dark urine, and pale stools. Prolonged jaundice, defined as jaundice lasting for more than two weeks of age, should raise the suspicion of cholestasis among health practitioners. Prolonged jaundice necessitating serum-conjugated bilirubin assessment, is observed in about 15% of infants. In the neonatal period, hyperbilirubinemia occurs in about 50%, thus the expected occurrence of physiological jaundice usually delays the diagnosis of cholestasis. Infants may rarely present with a recurrence of jaundice (conjugated), after the complete disappearance of the neonatal physiological jaundice.
When cholestasis is suspected, its definition in infants is as follows: a serum conjugated (direct) bilirubin of more than 1 mg/dl if the total bilirubin is less than 5 mg/dl, or a conjugated component of more than 20% of the total when total bilirubin exceeds 5 mg/dl.
As NIC has a diverse set of etiologies, some of which are treatable while others are not, the second step, after documenting cholestasis, is to recognize its etiology. Generally, causes are classified as either surgical or medical. Causes may be also classified according to their site, whether as extrahepatic (BA, choledochal malformation (CM), cholelithiasis, inspissated bile or mucous plug, or spontaneous bile duct perforation) or an intra-hepatic.
Surgical causes of cholestasis include BA and non-biliary atresia or CM. BA represents the most common recognizable cause of NIC. Its incidence ranges from 0.55 to 1.3/10,000 live births. BA accounts for approximately 20–35 % of infantile cholestasis. Besides, it is frequently the leading cause of pediatric liver transplantation.
Pathologically BA is a rapid progressive inflammation and fibrosis of the bile ducts (both extrahepatic, and intrahepatic) leading to their partial or complete obliteration. The etiology of BA is likely multifactorial, including infections, malformations, vascular, autoimmune, and toxic factors, as well as genetic, and epigenetic causes.
BA is classified into types 1, 2 (a/b), and 3 according to the extent of extrahepatic bile duct involvement. Type 1 and 2 represent about 20% of BA cases, with their pathology limited to the distal extrahepatic bile ducts. Type 1 affects the CBD, and type 2 the common hepatic duct. These types carry a good prognosis after timely surgical correction. Type 3 is more common. It is characterized by gallbladder involvement, with fibrous tissue in most cases, mucocele in a few, and deep fibrous plates in the extrahepatic bile ducts. It involves atresia of the right and left hepatic ducts to the level of the porta hepatis. BA exists as an isolated pathology or in association with other malformations. Extrahepatic malformation is a common association, occurring in 20%. BA with splenic malformation (BASM) is a common extrahepatic malformation characterized by asplenia or polysplenia, cardiovascular malformations (dextrocardia, preduodenal portal vein, agenesis of retro-hepatic inferior vena cava, and anomalous superior vena cava), and laterality defects (intestinal malrotation and situs inversus). A score for diagnosis of BA based on clinical data, biochemical results, ultrasound and histological findings and having an accuracy of 98.83 has been proposed and validated.
Choledochal malformation (CM) is also termed congenital biliary dilatation. It is a malformation of the pancreaticobiliary system, characterized by a dilatated biliary tract associated commonly with pancreaticobiliary mal junction. It is classified into 5 types.
Timely surgical correction is mandatory to prevent progressive hepatic damage and biliary obstruction and their consequences, such as growth retardation, cholangitis, pancreatitis, hepatic fibrosis, and even cirrhosis and late malignant transformation in any biliary remnant. CM should be differentiated from cystic biliary atresia, liver cyst, and duodenal duplication.
Medical causes of NIC include genetic, endocrine, infectious, hematologic, and toxic conditions, with many of them described as monogenic liver disorders. These conditions are categorized as intrahepatic causes of cholestasis.
Genetic conditions include canalicular bile acid transport defects such as progressive familial intrahepatic cholestasis (PFIC), bile acid synthesis defects, lysosomal storage defects such as Niemann-Pick and Gaucher disease, amino acid metabolism defects such as tyrosinemia type 1, carbohydrate metabolism defects such as galactosemia and glycogen storage disease type IV, and urea cycle defect (Citrin deficiency), in addition to Alagille syndrome, cystic fibrosis, and alpha-1-antitrypsin deficiency.
Cholestasis may occur in association with endocrine disorders such as thyroid disorders, panhypopituitarism, and adrenal insufficiency or with infections such as cytomegalovirus, herpes simplex virus, parvovirus B19, human immunodeficiency virus (HIV), toxoplasma, syphilis, and congenital tuberculosis. It may be also a manifestation of hematologic conditions or an immune dysregulation such as hemophagocytic lymphohistiocytosis, congenital lupus, neonatal hemochromatosis (gestational alloimmune liver disease -GALD), and post-hemolytic conditions. In addition, cholestasis may occur secondary to certain conditions such as parenteral nutrition (parenteral nutrition associated cholestasis -PNALD), drugs, intestinal obstruction, cardiovascular disorders, neoplasia, and perinatal asphyxia.
Clinical manifestations (5-7)
History and physical examination are the initial steps in the evaluation and usually guide the diagnostic process. The report of acholic stools is a strong indicator of cholestasis. Dark urine is another nonspecific indicator of increased serum conjugated bilirubin levels.
Diagnosis of BA is time-critical and dependent on the identification of prolonged neonatal jaundice and pale or clay-colored stool. The identification of stool color may suffer from subjective reporting; an objective method of assessment, using a stool color card (SCC), may prove beneficial.
In Taiwan, screening infants with a stool color card was introduced in 2004. This markedly improved the sensitivity of detecting BA before 60 days, from 72.5% in 2004 to 97.5% in 2005. A recent study recommended using the stool color card as a mass neonatal screening tool, and including it in child health cards in low/middle-income countries such as Bangladesh for early pick-up of cholestatic cases.
Family history may suggest a genetic disorder such as cystic fibrosis, a-1-antitrypsin deficiency, or PFIC, or a disorder with an autosomal dominant inheritance pattern such as Alagille Syndrome.
Looking for bleeding or bruising due to a coagulopathy from vitamin K malabsorption or liver failure is essential in evaluating cholestatic infants.
Parameters of clinical interest in the history of the cholestatic infant
Family history
· Consanguinity increases the risk of autosomal recessive disorders.
· Neonatal cholestasis in parents or siblings may point to cystic fibrosis, a -1-antitrypsin deficiency, progressive familial intrahepatic cholestasis, and Alagille syndrome.
· History of repeated fetal loss or early demise as GALD.
· Spherocytosis and other hemolytic diseases which are known to aggravate conjugated hyperbilirubinemia
Prenatal history
· Prenatal ultrasonographic findings may denote the presence of a CM or cholelithiasis.
· Cholestasis of pregnancy may be seen in heterozygotes for PFIC gene mutations.
· Maternal infections such as TORCH infections.
➡️Infant history
· Gestational age: Prematurity is a risk factor for neonatal hepatitis.
· Small for gestational age increases the risk of neonatal cholestasis and congenital infections.
· Neonatal infections as in cases of urinary tract infection and sepsis-related cholestasis.
· Sources of nutrition such as breast milk, formula, or parenteral nutrition, are important to suspect galactosemia, and PNALD.
· Genetic and metabolic diseases may result in growth failure.
· Persistent vomiting may point to metabolic diseases.
· Delayed stooling may point to cystic fibrosis.
· The presence of acholic stools suggests biliary obstruction.
· Irritability, and lethargy suggest metabolic diseases, sepsis, or panhypopituitarism.
· Hypoglycemia may suggest metabolic or endocrinal conditions.
➡️Physical findings in children with neonatal cholestasis
· Assessment of general health: the finding of an ill-appearing infant may indicate that infections or metabolic diseases are the cause of cholestasis. Infants with biliary atresia typically appear generally thriving and well, apart from jaundice.
· Dysmorphic features. Neonates with Alagille syndrome may exhibit characteristic facial appearance with a broad nasal bridge, triangular facies, and deep-set eyes.
· Vision and slit lamp examination: may show cataracts (e.g., galactosemia- congenital rubella), posterior embryotoxon (e.g., Alagille syndrome), or septo-optic dysplasia.
· Hearing assessment: is helpful in congenital infections, and storage disease.
· Cardiac examination: murmur, signs of heart failure, and congenital heart disease are common in Alagille syndrome, BA, and BASM.
· Abdominal examination: looking for the presence of ascites, abdominal wall veins, liver size and consistency, spleen size and consistency, and umbilical hernia.
· A stool examination is crucial, and the primary physician should make every effort to view by himself the stool color does not depend on caregivers’ description. Acholic or hypopigmented stool suggest cholestasis or biliary obstruction.
· Neurologic examination: Note overall vigor, tone and neonatal reflexes.
➡️Extended Stepwise Diagnostic Approach for Cholestasis (2,8,9):
Step 1: Recognize cholestasis
- Timeframe: Evaluate any infant jaundiced beyond 2 weeks of age or 3 weeks for breastfed infants, with fractionated serum bilirubin to detect conjugated hyperbilirubinemia. The early recognition of cholestasis is crucial to prevent progressive liver damage.
- Initial Test:
- Measure total and direct serum bilirubin.
- Direct bilirubin (conjugated bilirubin) of more than 1 mg/dl, if the total bilirubin is less than 5 mg/dl, or a conjugated component of more than 20% of the total, when total bilirubin exceeds 5 mg/dl.
Step 2: Identify Red Flags for Cholestasis
- Acholic stools (pale or white stools).
- Dark urine.
- Failure to thrive, poor feeding, or irritability.
- Family history of liver disease, consanguinity, or genetic disorders.
- Dysmorphic features as in Alagille syndrome, and Down syndrome.
- Noticeable bleeding, e.g., that observed during circumcision.
- Extrahepatic features, e.g., heart murmurs, splenomegaly, or kidney abnormalities.
- The history of meconium ileus, and failure to pass stool early in life suggests cystic fibrosis.
Step 3: Comprehensive Physical Examination
- Abdominal examination for hepatomegaly, splenomegaly, and ascites.
- Assessment of dysmorphic features associated with genetic syndromes, e.g., Alagille syndrome.
- Panhypopituitarism: hypoplastic genitalia, poor growth, and hypoglycemia.
- Cardiac examination for murmurs or signs of heart failure.
- Neurological examination for poor muscle tone or developmental delay as in septo-optic dysplasia or Niemann-Pick disease type C.
Step 4: Initial Laboratory Workup
- Fractionated bilirubin: total and direct serum bilirubin.
- Liver function tests: alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma-glutamyl transferase (GGT), albumin, and coagulation profile (prothrombin time and INR). Severe coagulopathy unresponsive to vitamin K may indicate a genetic or metabolic disorder. Low GGT suggests PFIC types 1,2,4,5,6 or bile acid synthesis defects.
- Complete blood count
- Metabolic screen:
- Glucose check for hypoglycemia, which may indicate panhypopituitarism, galactosemia, or tyrosinemia.
- Thyroid functions: Thyroid stimulating hormone (TSH) and free T4 for hypothyroidism.
- Urine for non-glucose reducing substance.
Step 5: Diagnostic Imaging
- Abdominal ultrasound is first-line imaging. It evaluates liver anatomy and bile duct structure. It can assess:
- Gallbladder abnormalities such as absent gallbladder in biliary atresia.
- CM or biliary tree dilation.
- Signs of advanced liver disease such as coarse liver texture, splenomegaly or ascites.
Step 6: Second tier tests for genetic/ metabolic condition
If the initial workup is inconclusive or if the infant presents with syndromic features, move to a more detailed genetic and metabolic investigation.
· Genetic Disorders:
- Alagille Syndrome:
o GGT: Often disproportionately elevated.
o An echocardiogram to evaluate associated cardiac anomalies.
o Genetic testing for JAG1 or NOTCH2 mutations.
- Cystic Fibrosis:
o Sweat chloride test or CFTR genetic testing.
- PFIC:
o Check for low GGT in PFIC types 1, 2, 4, 5, and 6.
o Genetic testing: ATP8B1 (PFIC1), ABCB11 (PFIC2), ABCB4 (PFIC3), tight junction protein-2 (PFIC4), NR1H4 gene (PFIC5), and MYO5B (PFIC6).
- Alpha-1 Antitrypsin Deficiency:
o Alpha-1 antitrypsin phenotype and serum levels.
o Liver biopsy may show hepatocellular injury, cholestasis, or PAS-positive, diastase-resistant granules.
- Bile Acid Synthesis Defects:
o Serum bile acids are typically low with no pruritus despite severe conjugated jaundice.
o Urinary bile acid profiling via fast atom bombardment mass spectrometry.
o Genetic testing for enzyme deficiencies involved in bile acid synthesis.
· Metabolic Disorders:
- Galactosemia: enzyme assay for GALT deficiency, and genetic testing for GALT gene.
- Tyrosinemia: Check serum or urinary succinyl acetone, and AFP is usually >50.000.
- Urea cycle disorders: elevated serum citrulline in neonatal citrin deficiency.
- Biomarkers, enzyme assay or genetic mutations for diagnosis for Niemann-Pick disease type C and Gaucher disease.
Step 7: Infectious Causes
- Cytomegalovirus (CMV): polymerase chain reaction testing saliva, urine, or blood for CMV DNA. CMV IgM antibodies, though less sensitive.
- TORCH infections: Test for Toxoplasma, Rubella, Syphilis, and herpes simplex virus type 1 and 2 if there is maternal history or early onset jaundice with systemic illness.
- Urinary Tract Infections: Perform urine culture to rule out sepsis-related cholestasis.
Step 8: Endocrine Disorders
- Panhypopituitarism:
o Measure early morning cortisol, TSH, free T4, and perform MRI-brain imaging.
o Look for associated anomalies like septo-optic dysplasia.
Step 9: Advanced Diagnostic Techniques
- Liver biopsy:
o Consider if diagnostic uncertainty remains.
o Bile duct proliferation, and fibrosis are typical features in biliary atresia, while giant cell transformation is characteristic for neonatal hepatitis syndromes.
- Laparoscopic or intraoperative cholangiography is the gold standard for confirming BA by direct visualizing of the bile ducts.
- Magnetic Resonance Cholangio-Pancreatography (MRCP) is not reliable as the normal biliary tract is not consistently visualized in children under 3 months of age; false negative results are thus common.
- Hepatic iminodiacetic acid (HIDA) radionucloide scan is not readily available, time consuming and is not recommended anymore.
Step 10: Management Plan Based on Diagnosis
· BA: Early referral for Kasai portoenterostomy within the first 60 days is crucial.
· Genetic/metabolic disorders: Initiate targeted therapies, e.g., cholic acid for some bile acid synthesis defects, dietary management for galactosemia, fructosemia, and specific treatment for tyrosinemia.
· Infectious causes: Treat with appropriate antivirals for herpes simplex and CMV or antibiotics (bacterial infections).
· Endocrine causes: Treat with hormone replacement (e.g., cortisol, thyroxine) if panhypopituitarism or hypothyroidism is diagnosed.
Step 11: Multidisciplinary Management and Follow-Up
Consult specialists: Pediatric surgeons, gastroenterologist, geneticist, infectious disease expert, endocrinologist, metabolic diseases specialist, nutritionist or transplantation experts based on the underlying cause.
Regular monitoring of liver functions, growth, and response to treatment.
➡️Purpose and Scope
· This guideline has been developed to standardize the diagnosis of neonatal and infantile cholestasis (NIC). It provides an evidence-based guidance for healthcare personnel, such as general practitioners, general pediatricians, family medicine physicians, pediatric hepatologists, pediatric surgeons, neonatologists, nurse practitioners, physician’s assistants and pediatric gastroenterologists to help in early diagnosis of neonates and infants with cholestasis in Egypt.
· The guideline aims to provide recommendations to support: a) pediatricians working in primary and secondary care for the initial assessments of NIC; b) neonatologists and pediatric gastroenterologists working in a tertiary referral center for the extensive diagnostic work-up.
· The guideline focuses on timely detection of cholestasis (as its detection is often late in our health care settings), and identification of its causes (particularly treatable ones), which is crucial for the best patient outcome.
- Methods
➡️Methods of search:
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for search are: neonatal, infantile, cholestasis
Inclusion / exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated in the last 10 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO):
GIN: Number of results 1
SIGN: Number of results 1
WHO: Number of results 0
DynaMed and DynaMed Plus X: Number of results 0
NGC: Number of results 0
NHMRC: Number of results 0
BIGG- REC database: Number of results: 0
PEPARE platform database: Number of results: 0
ECRI Guidelines Trust database: Number of results: 0
GRADE ETD’S: Number of results: 0
2. Bibliographic databases (e.g., PubMed, Google Scholar):
Google scholar: Number of results 8
PubMed: Number of results 6
Cochrane Library: Number of results 0
3. Specialized professional societies (related to the pediatric subspecialty):
ESPGHAN and NASPGHAN: number of results 1
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 60% in the AGREE II Domain 3 (rigor of development) was retained).
Three guidelines were considered eligible for the AGREE II appraisal instrument which were:
1. Diagnostic approach to neonatal and infantile cholestasis: A position paper by the SIGENP liver disease working group (2022).
2. Guideline for the Evaluation of Cholestatic Jaundice in Infants: Joint Recommendations of the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition and the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition (2017).
3. Italian guidelines for the management and treatment of neonatal cholestasis (2015).
After reviewing all the previous criteria and the AGREE II appraisal results the GDG/ GAG recommended using 1 guideline: Diagnostic approach to neonatal and infantile cholestasis: A position paper by the SIGENP liver disease working group (2022). (Ranucci G, Della Corte C, Alberti D, Bondioni MP, Boroni G, Calvo PL, Cananzi M, Candusso M, Clemente MG, D'Antiga L, Degrassi I, De Ville De Goyet J, Di Dato F, Di Giorgio A, Vici CD, Ferrari F, Francalanci P, Fuoti M, Fusaro F, Gaio P, Grimaldi C, Iascone M, Indolfi G, Iorio R, Maggiore G, Mandato C, Matarazzo L, Monti L, Mosca F, Nebbia G, Nuti F, Paolella G, Pinon M, Roggero P, Sciveres M, Serranti D, Spada M, Vajro P, Nicastro E. Diagnostic approach to neonatal and infantile cholestasis: A position paper by the SIGENP liver disease working group. Dig Liver Dis. 2022 Jan;54(1):40-53. doi: 10.1016/j.dld.2021.09.011. Epub 2021 Oct 20. PMID: 34688573).
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Adaptation for 2 recommendation according to GRADE criteria to be suitable to our Economic implications (Evidence to Decision (EtD) table was done). In this guideline no EtD table was done.
- Development of Good Practice Statement
Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups; the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of hepatology
The main functions of the clinical panel were adolopment of Diagnostic approach to neonatal and infantile cholestasis: A position paper by the SIGENP liver disease working group (2022) guideline, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 7 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinicians subgroup, generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 3 clinical national experts who have interest and expertise in as well as eminent international reviewers in the diagnosis of neonatal and infantile cholestasis.
They were identified by Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism on the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of the guideline, EPG was responsible for overseeing the adolopment process. the timetable and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interests form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions [13]. Informed by the evidence required for the GRADE Evidence to Decision (EtD) framework(s) was(were) done while considering changing strength of recommendations according to availability of some resources in the recommendations (both ETD and changing strength of recommendation were not done in this guideline).
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
GRADE EtD’s contextual factors, criteria and considerations that link to the strength of recommendations:
Criteria and Considerations:
1. Benefits and harms: When a new recommendation is developed, desirable effects (benefits) need to be weighed against undesirable effects (risks/harms), considering any previous recommendation or another alternative. The larger the gap or gradient in favor of the desirable effects over the undesirable effects, the more likely that a strong recommendation will be made.
2. Certainty of the evidence about the effects: The higher the certainty of the scientific evidence base, the more likely that a strong will be made.
3. Values and preferences: If there is no important uncertainty or variability in how much people value the main outcomes, it is likely that a strong recommendation will be made. Uncertainty or variability around these values that could likely lead to different decisions, is more likely to lead to a conditional recommendation.
4. Economic implications: Lower costs (monetary, infrastructure, equipment or human resources) or greater cost-effectiveness are more likely to support a strong recommendation.
5. Equity and human rights: If an intervention will reduce inequities, improve equity or contribute to the realization of human rights, the greater the likelihood of a strong recommendation.
6. Feasibility: The greater the feasibility of an intervention to all stakeholders, the greater the likelihood of a strong recommendation.
7. Acceptability: If a recommendation is widely supported by health workers and program managers and there is widespread acceptance for implementation within the health service, the likelihood of a strong recommendation is greater.
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are clearly needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary with regard to actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include a well-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.
- Evidence to recommendations: Considerations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendations however, there was a need to change the strength of 2 recommendations (B2 and B3) as they lack feasibility. Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing the diagnosis, and emergency treatment of neonatal and infantile cholestasis into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
· Select the target populations and evaluate the outcome.
· Identify the local resources to support the implementation.
· Set timelines.
· Distribute the tasks to the members.
· Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
· Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
· Educational materials: printed or electronic information (software).
· Web-based education: computer-based educational activities.
· A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
· Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
· Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
· Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
· Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
· Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
· Mass media campaigns.
3. For Nurses
· Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
· Educational materials: printed.
· A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
· Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
· Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
· Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
· Administrative policies and procedures.
· Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
· International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
· Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
Guideline Implementation Tools
I. Stool card
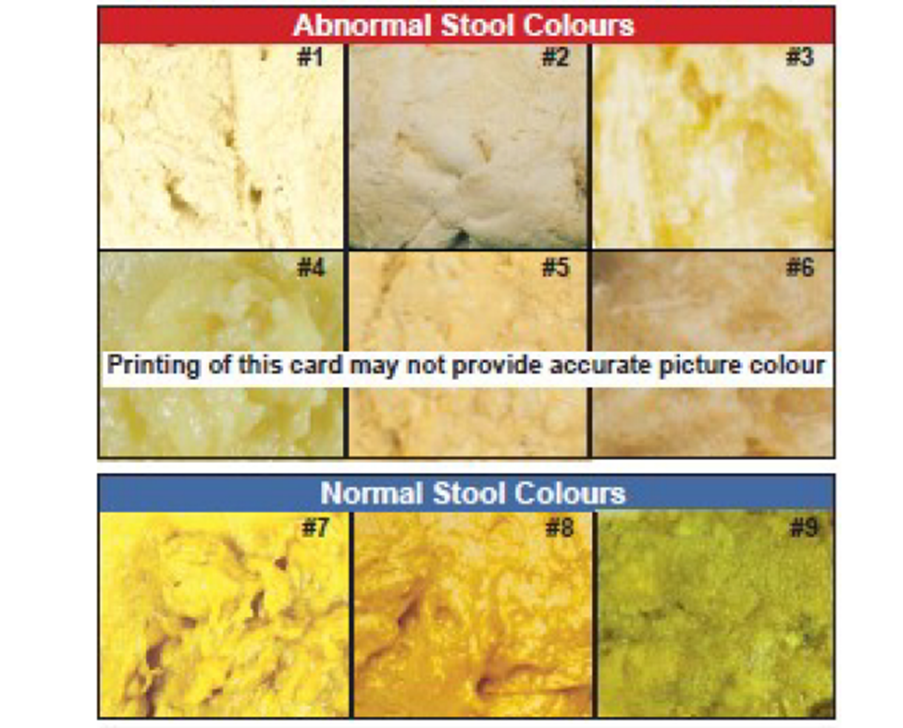
Figure 1: The stool color card (SCC), English version, developed by the Perinatal Services BC (British Columbia; in Vancouver, Canada (12).
II. Cholestasis Flow Chart
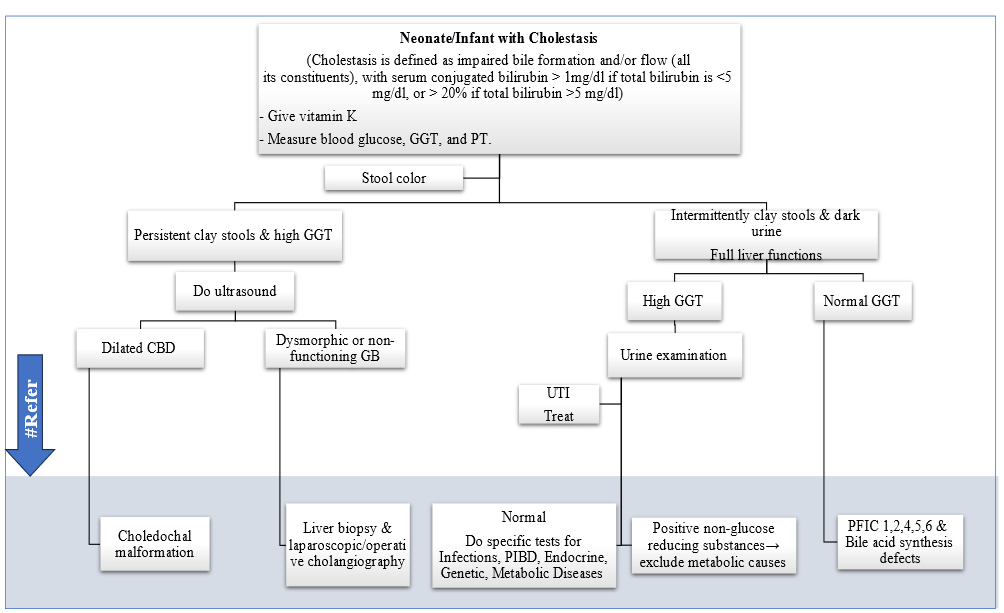
Figure 2: Neonatal and infantile cholestasis flow chart
# Referral to pediatric hepatologist is required. CBD: common bile duct, GB: gall bladder, GGT: Gamma-glutamyl transferase, PFIC: Progressive familial intrahepatic cholestasis, PIBD: Paucity of Interlobular Bile Ducts, PT: Prothrombin time, UTI: urinary tract infection. Adopted from: Manual of some Pediatric Liver Diseases (13).
- Limitations and suggestions for further research needs
The limitations we found are:
➡️Implementation limitations:
· Lack of availability of some diagnostic tests such as point of care ultrasound and Gamma-glutamyl transferase measurement.
· Lack of young physicians’ knowledge regarding neonatal cholestasis.
· Lack of public awareness regarding pathological causes of neonatal jaundice.
➡️Guideline limitations:
1. There was a very limited number of evidence-based guidelines dealing with NIC. After appraising that limited number using Agree II only one position paper was appropriate. So, we had to depend in our guideline on that position paper.
2. We had to resort to (good practice statement) more than once as there were no available appraised guideline statements to adapt.
3. Detailed management of NIC is not included in this guideline as after appraisal there were no good quality guidelines dealing with the entity of NIC management at the time of writing it. We hope to allegate another CPG for this topic.
➡️Monitoring and evaluating the impact of the guideline.
The following is a measure or indicators for implementing this adapted CPG for NIC:
· Adherence to Evidence-Based Clinical Practice Guideline for Diagnosis of Neonatal and Infantile Cholestasis:
‒ Numerator: Number of children with cholestasis, who were diagnosed as per the guideline recommendations.
‒ Denominator: Total number of children diagnosed with jaundice
‒ Data Source: Hospital or clinic patient records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
- Updating of the guideline
The EPG for Diagnosis of Neonatal and Infantile Cholestasis GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence- based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. Ranucci G, Della Corte C, Alberti D, Bondioni MP, Boroni G, Calvo PL, Cananzi M, Candusso M, Clemente MG, D'Antiga L, Degrassi I, De Ville De Goyet J, Di Dato F, Di Giorgio A, Vici CD, Ferrari F, Francalanci P, Fuoti M, Fusaro F, Gaio P, Grimaldi C, Iascone M, Indolfi G, Iorio R, Maggiore G, Mandato C, Matarazzo L, Monti L, Mosca F, Nebbia G, Nuti F, Paolella G, Pinon M, Roggero P, Sciveres M, Serranti D, Spada M, Vajro P, Nicastro E. Diagnostic approach to neonatal and infantile cholestasis: A position paper by the SIGENP liver disease working group. Dig Liver Dis. 2022 Jan;54(1):40-53. doi: 10.1016/j.dld.2021.09.011. Epub 2021 Oct 20. PMID: 34688573.
2. Fawaz R, Baumann U, Ekong U, Fischler B, Hadzic N, Mack CL, McLin VA, Molleston JP, Neimark E, Ng VL, Karpen SJ. Guideline for the Evaluation of Cholestatic Jaundice in Infants: Joint Recommendations of the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition and the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition. J Pediatr Gastroenterol Nutr. 2017 Jan;64(1):154-168. doi: 10.1097/MPG.0000000000001334. PMID: 27429428.
3. Banks JS, Saigal G, D'Alonzo JM, Bastos MD, Nguyen NV. Choledochal Malformations: Surgical Implications of Radiologic Findings. AJR Am J Roentgenol. 2018 Apr;210(4):748-760. doi: 10.2214/AJR.17.18402. PMID: 29446672.
4. El-Guindi MA, Sira MM, Sira AM, Salem TA, El-Abd OL, Konsowa HA, El-Azab DS, Allam AA. Design and validation of a diagnostic score for biliary atresia. J Hepatol. 2014 Jul;61(1):116-23. doi: 10.1016/j.jhep.2014.03.016. Erratum in: J Hepatol. 2015 Jul;63(1):289. PMID: 24657403.
5. Chen SM, Chang MH, Du JC, Lin CC, Chen AC, Lee HC, et al. Screening for biliary atresia by infant stool color card in Taiwan. Pediatrics 2006;117(4):1147–54.
6. Young S and Azzam RA. Infantile Cholestasis: Approach and Diagnostic Algorithm. In Textbook of Pediatric Gastroenterology, Hepatology and Nutrition A Comprehensive Guide to Practice, 2016. Editors; Guandalini S, Dhawan A, and Branski D. Chapter 55 page 625. Springer International Publishing.
7. El-Shabrawi MH, Baroudy SR, Hassanin FS, Farag AE. A pilot study of the value of a stool color card as a diagnostic tool for extrahepatic biliary atresia at a single tertiary referral center in a low/middle income country. Arab J Gastroenterol. 2021 Mar;22(1):61-65. doi: 10.1016/j.ajg.2020.12.004. Epub 2021 Mar 2. PMID: 33664005.
8. Wyllie R, Hyams LS and Kay M. PEDIATRIC GASTROINTESTINAL AND LIVER DISEASE, SIXTH EDITION ISBN: 978-0-323-67293-1 Copyright © 2021 by Elsevier, Inc.
9. Kelly D, Sharif K, Hartley J. Atlas of Pediatric Hepatolog. ISBN 978-3-319-69528-0 ISBN 978-3-319-69529-7 (eBook) https://doi.org/10.1007/978-3-319-69529-7. © Springer International Publishing AG, part of Springer Nature 2018.
10. Jagadisan B, Dhawan A. Emergencies in paediatric hepatology. J Hepatol. 2022 May;76(5):1199-1214. doi: 10.1016/j.jhep.2021.12.027. PMID: 34990749.
11. Feldman AG, Sokol RJ. Neonatal Cholestasis: Updates on Diagnostics, Therapeutics, and Prevention. Neoreviews. 2021 Dec 1;22(12):e819-e836. doi: 10.1542/neo.22-12-e819. PMID: 34850148; PMCID: PMC10103174.
12. The stool color card (SCC), English version, developed by the Perinatal Services BC (British Columbia; in Vancouver, Canada). Copyright 2019 Provincial Health Services Authority (used with permission). (http://www.perinatalservicesbc.ca/Documents/Screening/BiliaryAtresia/StoolColourCard_English.pdf).
13. Manual of some Pediatric Liver Diseases. The 2nd edition, 2016. Ain Shams University and Society of friends of liver patients in the Arab world.
14. The stool color card (SCC), Arabic version, developed by the Perinatal Services BC (British Columbia; Vancouver, Canada). Copyright 2019 Provincial Health Services Authority (used with permission).(http://www.perinatalservicesbc.ca/Documents/Screening/BiliaryAtresia/StoolColourCard_Arabic.pdf).
15. Abdel Baky A, Omar TEI, Amer YS; Egyptian Pediatric Clinical Practice Guidelines Committee (EPG). Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bull Natl Res Cent. 2023;47(1):88. https://doi.org/10.1186%2Fs42269-023-01059-0
16. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. Eastern Mediterranean Health Journal. 2023 Jul 1;29(7). https://www.emro.who.int/emhj-volume-29-2023/volume-29-issue-7/methodological-frameworks-for-adapting-global-practice-guidelines-to-national-context-in-the-eastern-mediterranean-region.html
17. Schünemann H, Brozek J, Guyatt G, Oxman A (editors). GRADE handbook: handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group; 2013 (Online updated version: https://gdt.gradepro.org/app/handbook/handbook.html Accessed 16/8/2024)
18. Klugar M, Lotfi T, Darzi AJ, et al. GRADE Guidance 39: Using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. Journal of Clinical Epidemiology. 2024 Aug 6:111494. https://doi.org/10.1016/j.jclinepi.2024.111494 (in press)
19. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The ‘Adapted ADAPTE’: an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the A lexandria C enter for E vidence‐B ased C linical P ractice G uidelines. Journal of evaluation in clinical practice. 2015 Dec;21(6):1095-106. https://doi.org/10.1111/jep.12479
20. Brouwers MC, Kho ME, Browman GP, Burgers JS, Cluzeau F, Feder G, Fervers B, Graham ID, Grimshaw J, Hanna SE, Littlejohns P, Makarski J, Zitzelsberger L; AGREE Next Steps Consortium. AGREE II: advancing guideline development, reporting and evaluation in health care. CMAJ. 2010 Dec 14;182(18):E839-42. https://doi.org/10.1503%2Fcmaj.090449
21. Agree II (2022) AGREE Enterprise website. Available at: https://www.agreetrust.org/resource-centre/agree-ii/ (Accessed: 16/8/2024).
22. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist[J]. Annals of Internal Medicine, 2022, 175(5):710-719. https://doi.org/10.7326/M21-4352 (Official RIGHT Statement Website: http://www.right-statement.org/extensions/13 Accessed 16/8/2024)
- Annexes
➡️Web annexes
The following annexes can be added as a package of standalone supplementary documents.
➡️Keywords: The MeSH terms for "Guideline for diagnosis of …neonatal and infantile cholestasis’. " on PubMed are: neonate, infantile, cholestasis, guideline
➡️Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Nanis Sulieman |
Associate Professor of Pediatrics Ain Shams University, Egypt |
None |
Not Applicable |
|
Dr. Ranin Soliman
|
1. Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. 2. Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. 3. Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
None |
Not applicable |
|
Dr. Lamis Mohsen Elsholkamy |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Ahmad Yousef |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
External Reviewer for methodology |
|||
|
|
|
|
|
|
International Peer Reviewers |
|
|
|
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
Annex Table 2. Guideline documentation; List of Retrieved Guidelines
|
N |
Title |
Database |
English |
Date |
Organization |
Type |
Evidence Based (method used) |
|
1 |
Diagnostic approach to neonatal and infantile cholestasis: A position paper by the SIGENP liver disease working groups |
Google scholar |
Yes |
2022 |
SIGENP liver disease working group; Italy |
Position paper |
Yes, Method: GRADE |
|
2 |
Italian guidelines for the management and treatment of neonatal cholestasis |
Google scholar |
Yes |
2015 |
Italian Society of Neonatology |
Guideline “review” |
Yes Method: Center for Evidence-Based-Medicine |
|
3 |
Efficacy and safety of ursodeoxycholic acid in children with cholestasis |
Google scholar |
Yes |
2023 |
Science and Technology Department of Sichuan Province, China |
A systematic review and meta-analysis |
-Quality of evidence (GRADE)
-Used Cochrane Collaboration risk of bias tool (Rob 2.0) |
|
4 |
Early differential diagnosis methods of biliary atresia |
Google scholar |
Yes |
2018 |
Authors, China |
A meta-analysis |
. Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2) |
|
5 |
Diagnostic Performance of Sonographic Features in Patients with Biliary Atresia |
Google scholar |
Yes |
2016 |
Authors, Korea |
A systematic review and meta-analysis |
. Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2) |
|
6 |
Systematic review of progressive familial intrahepatic cholestasis |
Google scholar |
Yes |
2019 |
Authors, multinational |
A systematic review and meta-analysis |
Used (PRISMA) guidelines protocol |
|
7 |
Systematic Review: The Epidemiology, Natural History, and Burden of Alagille Syndrome |
Google scholar |
Yes |
2018 |
Authors, multinational |
A systematic review |
Used (PRISMA) guidelines protocol |
|
8 |
Diagnostic Efficacy of Advanced Ultrasonography Imaging Techniques in Infants with Biliary Atresia (BA) |
Google scholar |
Yes |
2022 |
Authors, China |
A systematic review and meta-analysis |
Used (PRISMA) guidelines protocol Quality Assessment of Diagnostic Accuracy Studies 2 (QUADAS-2) |
|
9 |
Guideline for the Evaluation of Cholestatic Jaundice in Infants: Joint Recommendations of the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition and the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition.2017 |
PubMed |
yes |
2017 |
ESPGHAN; NSPGHAN |
CPG |
Yes Method: GRADE |
|
10 |
Outcomes of Alagille syndrome following the Kasai operation: a systematic review and meta-analysis |
PubMed |
yes |
2018 |
Fujishiro et al. |
systematic review |
Used: PRISMA guidelines Protocol |
|
11 |
Etiologies of conjugated hyperbilirubinemia in infancy: a systematic review of 1692 Subjects |
PubMed |
yes |
2015 |
Gottesman et al. |
systematic review |
Used: PRISMA guidelines Protocol |
|
12 |
The value of preoperative liver biopsy in the diagnosis of extrahepatic biliary atresia: A systematic review and meta-analysis |
PubMed |
yes |
2016 |
Lee et al. |
Systematic review |
|
➡️Annex Table 3. Results of the AGREE II assessment of the three source guidelines for
|
Guidelines |
Domain 1 Scope& purpose |
Domain 2
Stake hold involve |
Domain 3 Rigor of develop. |
Domain 4 Clarity of present. |
Domain 5 Applicability |
Domain 6
Editorial independence |
Overall |
|
1. Guideline for the Evaluation of Cholestatic Jaundice in Infants: Joint Recommendations of the North American Society for Pediatric Gastroenterology, Hepatology, and Nutrition and the European Society for Pediatric Gastroenterology, Hepatology, and Nutrition |
71% |
47% |
44% |
88% |
44% |
30% |
OA1:77% yes 3, yes with modifications 2, No 0 |
|
2. Diagnostic approach to neonatal and infantile cholestasis: A position paper by the SIGENP liver disease working group |
87% |
61% |
66% |
94% |
65% |
25%
|
OA 1 :60% OA 2: Yes - 4, Yes with modifications - 0, No - 0 |
|
3. Italian guidelines for the management and treatment of neonatal cholestasis |
76% |
39% |
30% |
80% |
28% |
65% |
OA1: 53% OA 2 : Yes - 2, Yes with modifications - 2, No - 1 |
Annex 4. Nurses and Parents Educational Guide in Arabic

بطاقة لون براز الرضع فى بريتش كولومبيا
برنامج كشف رتق القناة الصفراوية (14)
Appendix Table 5. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|
Gantt chart