the Management and Prevention of Peritonitis
| Site: | EHC | Egyptian Health Council |
| Course: | General surgery Guidelines |
| Book: | the Management and Prevention of Peritonitis |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 10 March 2026" Download Guideline
- Executive summary
These guidelines offer evidence-based recommendations on the targeted levels of clinical practice guidelines and provide healthcare professionals with practice guidance on the management of IAIs.
· Diagnostic paracentesis should be carried out without delay to confirm SBP in all cirrhotic patients with ascites on hospital admission, (Good practice statement).
· Ascitic neutrophil count >250/mm³ remains the gold standard for SBP diagnosis, (Good practice statement).
· We recommend for the diagnosis of IAIs should be based primarily on clinical assessment, basic laboratory tests, plain X-ray of the abdomen and ultrasound (Strong recommendation).
· We advise against performing imaging in case of suspected peritonitis due to organ perforation in a critically ill patient, if it delays the surgical procedure, (Conditional recommendation).
· When peritonitis due to perforated gastroduodenal ulcer is suspected, the indication for surgery can be based on clinical history and the presence of pneumoperitoneum on plain abdominal X-ray, (Good practice statement).
· In adult patients not undergoing immediate laparotomy, computed tomography (CT) scan is recommended to determine the presence of an intra-abdominal infection and its source, (Strong recommendation).
· Consider the diagnosis of ongoing IAI in the case of development or deterioration of organ dysfunction during the days following abdominal surgery for secondary peritonitis, (Conditional recommendation).
· If peritonitis is suspected, we recommend testing of the PD effluent for cell count, differential gram stain, and culture, (Strong recommendation).
· Peritonitis should be diagnosed when at least two of the following criteria are met:
1) Clinical features consistent with peritonitis; 2) Dialysis effluent WBC count > 100/µL (after ≥2h dwell) with >50% PMN; 3) Positive dialysis effluent culture, (Strong recommendation).
· Empirical broad spectrum antibiotic regimens are recommended, (Strong recommendation).
· We recommend that, empirical antibiotic therapy protocols for community- acquired IAI to be established on the basis of regular analysis of national and regional microbiological data in order to quantify and monitor the course of microbial resistance in the community, (Strong recommendation).
· In severe IAI, empirical antibiotic therapy should be adapted to the suspected organisms, (Strong recommendation).
· We recommend that, empirical broad spectrum antibiotic therapy to be initiated as soon as possible, using systemic intravenous (IV) route, after appropriate microbiological specimens have been obtained, (Strong recommendation).
· We recommend against empirical therapy against Candida in community-acquired IAI in the absence of signs of severity, (Strong recommendation).
· We advise administration of antifungal therapy in severe peritonitis (community-acquired or postoperative), in the presence of at least 3 of the following criteria: hemodynamic failure, female gender, upper gastrointestinal surgery, antibiotic therapy for more than 48 hours, (Conditional recommendation).
· When adequate source control has been achieved, we recommend the antibiotic treatment duration to be shortened, (Strong recommendation).
· Where feasible, we advise performing percutaneous drainage, as it is preferable to surgical drainage, (Conditional recommendation).
· We recommend that, a patient with suspected peritonitis due to organ perforation to be operated upon as rapidly as possible with proper resuscitation, especially in the presence of septic shock, (Strong recommendation).
· In the absence of hemodynamic instability (defined as the need for more than 0.1 mg/kg/min of epinephrine or norepinephrine), we advise that the decision to perform first-line image-guided percutaneous drainage for the management of intra-abdominal abscess in the absence of clinical or radiological signs of perforation to be based on a multidisciplinary discussion. This also allows for microbiological examination of peritoneal fluid samples, (Conditional recommendation).
· We advise obtaining peritoneal fluid samples in community-acquired IAI in order to identify the microorganism and determine their susceptibility to anti-infective agents, (Conditional recommendation).
· Routine source control procedures are recommended to remove infected fluid and tissue to prevent ongoing contamination in patients with IAI, except for those with clinical problems for which there is clear evidence that a non-interventional approach is associated with a good clinical outcome, (Strong recommendation).
· We recommend that source control should be undertaken within 24 hours of the diagnosis of IAI, except for those infections for which clinical evidence indicates a non-interventional or delayed approach is appropriate. Source control should be undertaken in a more urgent manner in patients with sepsis or septic shock, (Strong recommendation).
· We recommend that, surgical source control procedures should involve the resection or suture of a diseased or perforated viscus (e.g., diverticular perforation, gastroduodenal perforation), removal of the infected organ (e.g., appendix, gallbladder), debridement of necrotic tissue, resection of ischemic bowel, and repair or resection of traumatic perforations, (Strong recommendation).
· We advise performing laparoscopy, as a less invasive approach for both the diagnosis and treatment for intra-abdominal infections, particularly when performed by experienced surgeons, (Conditional recommendation).
· We recommend against laparoscopy for the treatment of peritonitis due to perforated peptic ulcer in a patient presenting more than one of the following risk factors: state of shock on admission, ASA score III- IV, and presence of symptoms for more than 24 hours, (Strong recommendation).
· We recommend against laparoscopy in the case of purulent or fecal peritonitis due to diverticulosis, (Strong recommendation).
· For intra-abdominal abscesses, we recommend percutaneous drainage as the preferred first-line therapy over open surgical intervention, where feasible, due to its association with lower complication rates and shorter hospital stays, (Strong recommendation).
· Drainage should be checked by CT scan in the presence of signs of deterioration, (Strong recommendation).
· We recommend against primary closure for controlling persistent infection, preventing abdominal compartment syndrome, or deferring definitive intervention and anastomosis. It is recommended as a potentially life-saving strategy in a carefully selected group of surgical patients with severe abdominal sepsis, particularly as part of a damage control strategy, (Strong recommendation).
· We recommend performing re-laparotomy on the fourth or fifth day after the index operation in the absence of any signs of clinical or laboratory improvement, (Strong recommendation).
· We recommend that, in the case of postoperative abscess, the benefit-risk balance of image-guided percutaneous drainage versus re-laparotomy should be assessed by a multidisciplinary team. (Strong recommendation).
· We recommend performing double contrast-enhanced computed tomography of the abdomen and pelvis. In case of renal impairment, only oral contrast is done. (Strong recommendation).
· We recommend removal of the PD catheter in refractory peritonitis episodes, defined as failure of the PD effluent to clear after 5 days of appropriate antibiotics, (Strong recommendation).
· We recommend immediate catheter removal when fungi are identified in PD effluent, (Strong recommendation).
· We advise that treatment with an appropriate anti- fungal agent be continued for at least 2 weeks after catheter removal, (Conditional recommendation).
· We advise administration of anti-tuberculous therapy, instead of PD catheter removal, as the primary treatment of peritonitis caused by Mycobacterium tuberculosis, (Conditional recommendation).
· We recommend adjustment of the antibiotic therapy once results and sensitivities are known, (Strong recommendation).
· In patients suspected to be infected by resistant Enterobacteriaceae, ampicillin- and/or vancomycin-resistant enterococci or methicillin-resistant Staphylococcus aureus (MRSA), we recommend that, these strains should probably be taken into account in the empirical antibiotic therapy for healthcare-associated peritonitis, (Strong recommendation).
· We recommend that empirical antifungal therapy to be initiated in healthcare-associated IAI, when yeast is detected on direct examination. Antifungal therapy (echinocandins in the case of serious infection or fluconazole-resistant strains) should probably be initiated in all cases of healthcare-associated IAI in which peritoneal fluid culture (apart from closed suction drains and drainage systems, etc.) is positive for yeasts, (Strong recommendation).
· We advise primary prophylaxis to patients considered at high risk, as defined by an ascitic protein count <1.5 g/dL. However, it is important that the potential risks and benefits and existing uncertainties are communicated to patients, (Conditional recommendation).
· For patients who have recovered from an episode of SBP, we advise for treatment with norfloxacin, ciprofloxacin, or co-trimoxazole to prevent further episodes of SBP, (Conditional recommendation).
· For patients presenting with gastrointestinal bleeding and underlying ascites due to cirrhosis, we recommend receiving prophylactic antibiotic treatment (cefotaxime has been widely studied but the antibiotic should be chosen based on local data) to prevent the development of SBP, (Strong recommendation).
· We recommend that systemic prophylactic antibiotics be administered immediately prior to catheter placement, (Strong recommendation).
· We advise prophylactic antibiotics after wet contamination of the PD system to prevent peritonitis, (Conditional recommendation).
· We advise antibiotic prophylaxis prior to colonoscopy and invasive gynecological procedure, (Conditional recommendation).
· We advise performing drainage of PD fluid to keep the abdomen empty before endoscopic gastrointestinal and invasive or instrumental gynecological procedures, (Conditional recommendation).
· To prevent fungal peritonitis, we recommend that anti-fungal prophylaxis be co-prescribed whenever PD patients receive an antibiotic course, regardless of the indication, (Strong recommendation).
- Recommendations
Section 1: Diagnosis of peritonitis:
(A): Primary peritonitis (Spontaneous bacterial peritonitis, SBP):
· Diagnostic paracentesis should be carried out without delay to confirm SBP in all cirrhotic patients with ascites on hospital admission, (Good practice statement).
· Ascitic neutrophil count >250/mm³ remains the gold standard for SBP diagnosis, (Good practice statement).
(B): Secondary peritonitis:
· We recommend for the diagnosis of IAIs should be based primarily on clinical assessment, basic laboratory tests, plain X-ray of the abdomen and ultrasound (Strong recommendation, moderate certainty evidence, (3 & 4)).
· We advise against performing imaging in case of suspected peritonitis due to organ perforation in a critically ill patient, if it delays the surgical procedure, (Conditional recommendation, low certainty evidence, (4)).
· When peritonitis due to perforated gastroduodenal ulcer is suspected, the indication for surgery can be based on clinical history and the presence of pneumoperitoneum on plain abdominal X-ray, (Good practice statement).
· In adult patients not undergoing immediate laparotomy, computed tomography (CT) scan is recommended to determine the presence of an intra-abdominal infection and its source, (Strong recommendation, moderate certainty evidence, (4)).
(C): Tertiary peritonitis:
· Consider the diagnosis of ongoing IAI in the case of development or deterioration of organ dysfunction during the days following abdominal surgery for secondary peritonitis, (Conditional recommendation, low certainty evidence, (3)).
(D): Peritoneal dialysis (PD) associated peritonitis:
· If peritonitis is suspected, we recommend testing of the PD effluent for cell count, differential gram stain, and culture, (Strong recommendation, moderate certainty evidence, (5)).
· Peritonitis should be diagnosed when at least two of the following criteria are met:
1) Clinical features consistent with peritonitis; 2) Dialysis effluent WBC count > 100/µL (after ≥2h dwell) with >50% PMN; 3) Positive dialysis effluent culture, (Strong recommendation, low certainty evidence, (5).
Section 2: Medical management:
· Empirical broad spectrum antibiotic regimens are recommended, (Strong recommendation, moderate certainty evidence, (6)).
· We recommend that, empirical antibiotic therapy protocols for community- acquired IAI to be established on the basis of regular analysis of national and regional microbiological data in order to quantify and monitor the course of microbial resistance in the community, (Strong recommendation, high certainty evidence, (3)).
· In severe IAI, empirical antibiotic therapy should be adapted to the suspected organisms, (Strong recommendation, moderate certainty evidence, (3)).
· We recommend that, empirical broad spectrum antibiotic therapy to be initiated as soon as possible, using systemic intravenous (IV) route, after appropriate microbiological specimens have been obtained, (Strong recommendation, moderate certainty evidence, (3 & 6)).
· We recommend against empirical therapy against Candida in community-acquired IAI in the absence of signs of severity, (Strong recommendation, moderate certainty evidence, (3)).
· We advise administration of antifungal therapy in severe peritonitis (community-acquired or postoperative), in the presence of at least 3 of the following criteria: hemodynamic failure, female gender, upper gastrointestinal surgery, antibiotic therapy for more than 48 hours, (Conditional recommendation, low certainty evidence, (3)).
· When adequate source control has been achieved, we recommend the antibiotic treatment duration to be shortened, (Strong recommendation, moderate certainty evidence, (6)).
Section 3: Surgical management:
(A): Primary peritonitis (Spontaneous bacterial peritonitis, SBP):
· Where feasible, we advise performing percutaneous drainage, as it is preferable to surgical drainage, (Conditional recommendation, moderate certainty evidence, (4)).
(B): Secondary peritonitis:
· We recommend that, a patient with suspected peritonitis due to organ perforation to be operated upon as rapidly as possible with proper resuscitation, especially in the presence of septic shock, (Strong recommendation, high certainty evidence, (3)).
· In the absence of hemodynamic instability (defined as the need for more than 0.1 mg/kg/min of epinephrine or norepinephrine), we advise that the decision to perform first-line image-guided percutaneous drainage for the management of intra-abdominal abscess in the absence of clinical or radiological signs of perforation to be based on a multidisciplinary discussion. This also allows for microbiological examination of peritoneal fluid samples, (Conditional recommendation, low certainty evidence, (3)).
· We advise obtaining peritoneal fluid samples in community-acquired IAI in order to identify the microorganism and determine their susceptibility to anti-infective agents, (Conditional recommendation, low certainty evidence, (3)).
· Routine source control procedures are recommended to remove infected fluid and tissue to prevent ongoing contamination in patients with IAI, except for those with clinical problems for which there is clear evidence that a non-interventional approach is associated with a good clinical outcome, (Strong recommendation, moderate certainty evidence, (3, 4 & 6)).
· We recommend that source control should be undertaken within 24 hours of the diagnosis of IAI, except for those infections for which clinical evidence indicates a non-interventional or delayed approach is appropriate. Source control should be undertaken in a more urgent manner in patients with sepsis or septic shock, (Strong recommendation, moderate certainty evidence, (3, 4 & 6)).
· We recommend that, surgical source control procedures should involve the resection or suture of a diseased or perforated viscus (e.g., diverticular perforation, gastroduodenal perforation), removal of the infected organ (e.g., appendix, gallbladder), debridement of necrotic tissue, resection of ischemic bowel, and repair or resection of traumatic perforations, (Strong recommendation, high certainty evidence, (6)).
· We advise performing laparoscopy, as a less invasive approach for both the diagnosis and treatment for intra-abdominal infections, particularly when performed by experienced surgeons, (Conditional recommendation, high certainty evidence, (6)).
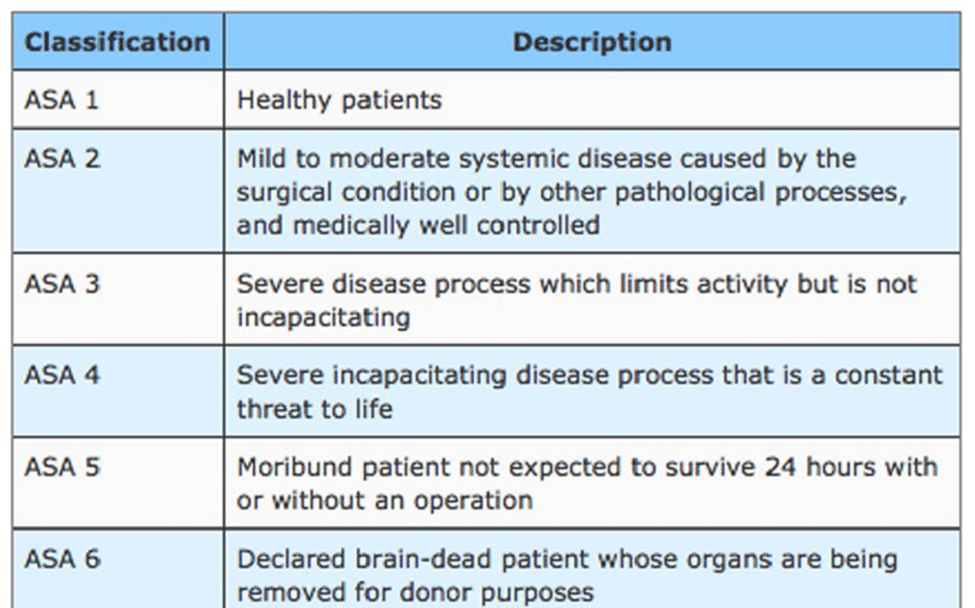
· We recommend against laparoscopy for the treatment of peritonitis due to perforated peptic ulcer in a patient presenting more than one of the following risk factors: state of shock on admission, ASA score III- IV, and presence of symptoms for more than 24 hours, (Strong recommendation, moderate certainty evidence, (3)), (table 3).
· We recommend against laparoscopy in the case of purulent or fecal peritonitis due to diverticulosis, (Strong recommendation, high certainty evidence, (3)).
· For intra-abdominal abscesses, we recommend percutaneous drainage as the preferred first-line therapy over open surgical intervention, where feasible, due to its association with lower complication rates and shorter hospital stays, (Strong recommendation, moderate certainty evidence, (4)).
· Drainage should be checked by CT scan in the presence of signs of deterioration, (Strong recommendation, high certainty evidence, (3)).
· We recommend against primary closure for controlling persistent infection, preventing abdominal compartment syndrome, or deferring definitive intervention and anastomosis. It is recommended as a potentially life-saving strategy in a carefully selected group of surgical patients with severe abdominal sepsis, particularly as part of a damage control strategy, (Strong recommendation, moderate certainty evidence, (3)).
(C): Tertiary peritonitis:
· We recommend performing re-laparotomy on the fourth or fifth day after the index operation in the absence of any signs of clinical or laboratory improvement, (Strong recommendation, moderate certainty evidence, (3)).
· We recommend that, in the case of postoperative abscess, the benefit-risk balance of image-guided percutaneous drainage versus re-laparotomy should be assessed by a multidisciplinary team. (Strong recommendation, moderate certainty evidence, (3)).
· We recommend performing double contrast-enhanced computed tomography of the abdomen and pelvis. In case of renal impairment, only oral contrast is done. (Strong recommendation, moderate certainty evidence, (3)).
(D): Peritoneal Dialysis (PD) associated peritonitis:
· We recommend removal of the PD catheter in refractory peritonitis episodes, defined as failure of the PD effluent to clear after 5 days of appropriate antibiotics, (Strong recommendation, moderate certainty evidence, (5)).
· We recommend immediate catheter removal when fungi are identified in PD effluent, (Strong recommendation, low certainty evidence, (5)).
· We advise that treatment with an appropriate anti- fungal agent be continued for at least 2 weeks after catheter removal, (Conditional recommendation, low certainty evidence, (5).
· We advise administration of anti-tuberculous therapy, instead of PD catheter removal, as the primary treatment of peritonitis caused by Mycobacterium tuberculosis, (Conditional recommendation, low certainty evidence, (5)).
Section 4: Post-operative care and complications management:
· We recommend adjustment of the antibiotic therapy once results and sensitivities are known, (Strong recommendation, moderate certainty evidence, (4)).
· In patients suspected to be infected by resistant Enterobacteriaceae, ampicillin- and/or vancomycin-resistant enterococci or methicillin-resistant Staphylococcus aureus (MRSA), we recommend that, these strains should probably be taken into account in the empirical antibiotic therapy for healthcare-associated peritonitis, (Strong recommendation, moderate certainty evidence, (3)).
· We recommend that empirical antifungal therapy to be initiated in healthcare-associated IAI, when yeast is detected on direct examination. Antifungal therapy (echinocandins in the case of serious infection or fluconazole-resistant strains) should probably be initiated in all cases of healthcare-associated IAI in which peritoneal fluid culture (apart from closed suction drains and drainage systems, etc.) is positive for yeasts, (Strong recommendation, moderate certainty evidence, (3)).
Section 5: Peritonitis prevention:
(A) Spontaneous Bacterial Peritonitis (SBP) Prophylaxis:
· We advise primary prophylaxis to patients considered at high risk, as defined by an ascitic protein count <1.5 g/dL. However, it is important that the potential risks and benefits and existing uncertainties are communicated to patients, (Conditional recommendation, low certainty evidence, (4)).
· For patients who have recovered from an episode of SBP, we advise for treatment with norfloxacin, ciprofloxacin, or co-trimoxazole to prevent further episodes of SBP, (Conditional recommendation, low certainty evidence, (4)).
· For patients presenting with gastrointestinal bleeding and underlying ascites due to cirrhosis, we recommend receiving prophylactic antibiotic treatment (cefotaxime has been widely studied but the antibiotic should be chosen based on local data) to prevent the development of SBP, (Strong recommendation, high certainty evidence, (4)).
(B) Peritoneal Dialysis (PD)-Associated Peritonitis Prevention:
· We recommend that systemic prophylactic antibiotics be administered immediately prior to catheter placement, (Strong recommendation, high certainty evidence, (5)).
· We advise prophylactic antibiotics after wet contamination of the PD system to prevent peritonitis, (Conditional recommendation, very low certainty evidence, (5)).
· We advise antibiotic prophylaxis prior to colonoscopy and invasive gynecological procedure, (Conditional recommendation, low certainty evidence, (5)).
· We advise performing drainage of PD fluid to keep the abdomen empty before endoscopic gastrointestinal and invasive or instrumental gynecological procedures, (Conditional recommendation, very low certainty evidence, (5)).
· To prevent fungal peritonitis, we recommend that anti-fungal prophylaxis be co-prescribed whenever PD patients receive an antibiotic course, regardless of the indication, (Strong recommendation, moderate certainty evidence, (5)).
- Abbreviations
AGREE II |
Appraisal of Guidelines for Research and Evaluation II. |
ASA |
American society of anesthesiologists |
C & S |
Culture and Sensitivity. |
CT |
Computed Tomography. |
GDG |
Guideline Development Group. |
GI |
Gastrointestinal. |
GRADE |
Grading of Recommendations Assessment, Development, and Evaluation |
IAIs |
Intra-Abdominal Infections. |
I&D |
Incision and Drainage. |
IDSA |
Infectious Diseases Society of America. |
IV |
Intravenous. |
MRI |
Magnetic Resonance Imaging. |
MRSA |
Methicillin-Resistant Staphylococcus Aureus. |
SIRS |
Systemic Inflammatory Response Syndrome. |
SSTIs |
Skin and Soft Tissue Infections. |
SBP |
Spontaneous Bacterial Peritonitis |
PD |
Peritoneal Dialysis |
PMN |
Polymorphonuclear |
TMP/SMX |
Trimethoprim- |
- Acknowledgement
We would like to acknowledge the Guideline Development Group, (GDG) committee for developing this guideline.
Chair of GDG: Mostafa Abdel Hamed Soliman; professor of surgery, Cairo University.
Moderator of GDG: Mohamed Ali Mohamed Nada, professor of surgery, Ain Shams University.
Members of GDG (Alphabetically):
Abdelwahab Mohamed Ezzat; professor of surgery, Ain Shams University.
Ahmed Abdel Raouf Elgeidie; professor of digestive surgery, Mansoura University.
Alaa Abdallah; professor of surgery, Ain Shams University.
Atef Abdelghani Salem; professor of surgery, Benha University.
Hesham Abdel Raouf Elakad; professor of surgery, Ain Shams University.
Khaled Abdallah Elfiky; professor of surgery, Ain Shams University.
Khaled Amer; professor of surgery, Military medical academy.
Khaled Safwat; professor of surgical oncology and endoscopy, Zagazig University.
Ibrahim Elzayat; head of surgery department, Aswan University.
Mohamed Ibrahim Abdelhamid Elsayed, professor of surgery, Zagazig University.
Tarek Ibrahim; professor of surgery, National liver institute Menofia University.
- Glossary
Definition:
Peritonitis is defined as an inflammation of the peritoneal cavity, which can be localized or diffuse, and its origin can be infectious or aseptic, (1).
Types:
1. Primary peritonitis, (Spontaneous Bacterial Peritonitis, SBP):
This form occurs in the absence of an identifiable intra-abdominal source of infection, (2). It is most frequently observed in patients with ascites, a condition characterized by excess fluid accumulation in the peritoneum, often associated with underlying diseases such as liver cirrhosis, kidney failure, heart failure, or cancer, (2).
2. Secondary peritonitis:
Representing the most common type, secondary peritonitis arises from direct contamination of the peritoneal cavity, typically due to a breach in the integrity of the gastrointestinal or urogenital tracts or associated organs, (3). Common instigating factors include a ruptured appendix, perforated stomach ulcer, diverticulitis, or trauma, (4).
3. Tertiary peritonitis:
This is a less common but highly severe form, defined as a persistent or recurrent intra-abdominal infection that manifests at least 48 hours after a seemingly successful initial intervention for secondary peritonitis, (3). It is frequently linked to nosocomial pathogens and the emergence of multi-drug resistant organisms (4).
4. Peritoneal Dialysis (PD) associated peritonitis:
This specific type of primary infection is introduced into the peritoneal cavity via medical instruments, most notably peritoneal dialysis catheters, which are used for long-term waste removal in patients with kidney failure, (5).
- Introduction
Peritonitis is a critical inflammatory condition affecting the peritoneum, the thin membrane that lines the inner abdominal wall and covers the abdominal organs. This condition is predominantly caused by infection, although it can also result from exposure to irritating bodily fluids, (1). Intra-abdominal infections (IAIs) are common surgical emergencies and represent an important intra-hospital cause of morbidity and mortality, especially if poorly treated. IAIs represent a notable factor contributing to the loss of both human lives and resources across global hospital settings. The estimated overall mortality rate was 9.2% among patients affected by complicated intra-abdominal infections, (6). Given its potential for rapid progression to severe systemic complications, including septicemia, sepsis, multi-organ failure and death, peritonitis is consistently recognized and treated as a medical emergency, (3). A comprehensive understanding of its classification and the imperative for prompt, evidence-based management is crucial for improving patient outcomes.
- Scope and purpose
The scope of these guidelines is to establish a national guideline for peritonitis as regard the diagnosis, need for surgery, timing, and type of surgery to achieve a best outcome as much as we can.
The main purposes of these guidelines are to minimize malpractice and poor surgical decision, to improve the quality of medical care and surgical service, to provide the best clinical practice to our patients, and finally to reduce the cost of medical service.
- Target audience
The principle targeted audiences are the practicing surgeons, however, gastroenterologists, nephrologists, ICU specialists and nursing staff may be included as well.
- Methods
A comprehensive search for guidelines was undertaken to identify the relevant guidelines to consider for adaptation.
Inclusion/ exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting only national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated in 2010 and later)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author, not on behalf of an organization to be valid and comprehensive, a guideline ideally requires multidisciplinary input
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in:
• Developing organization/authors
• Date of publication, posting, and release
• Country/language of publication
• Date of posting and/or release
• Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least three members. The panel decided on a cut-off point (any guideline scoring above 50% on the rigor dimension was retained). The GDG decided to adapt the Guidelines for management of intra-abdominal infections, 2015, Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America, 2010, Peritonitis guideline recommendations: 2022 update on prevention and treatment, 2022 and Management of intra-abdominal infections: recommendations by the Italian council for the optimization of antimicrobial use, 2024
Evidence assessment
According to WHO Handbook for Guidelines, we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed GRADE information is available on the following sites:
• GRADE working group: https://www.gradeworkinggroup.org/
• GRADE online training modules: http://cebgrade.mcmaster.ca/
Specifically, the quality of evidence was graded as ‘High’, ‘Moderate’, ‘Low’ or ‘Very low’, (table 1& 2).
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
these are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Research needs
1. Randomized controlled trials are needed to compare the efficacy and safety of different anti- biotic regimens.
2. The recognition of risk factors of peritonitis from observational data.
- Clinical indicators of monitoring
1. CBC, CRP.
2. Plain X-Ray abdomen and chest.
3. CT abdomen and pelvis with contrast
4. Documentation of the management of peritonitis.
5. Documentation of the indication of surgery in peritonitis.
6. Documentation of the type of operation.
- Updating of the guideline
The GDG committee for guidelines development is responsible for the continuous evaluation of evidence available about peritonitis. The present guideline will be updated in case of significant changes based on new evidence.
- Annexes
Table 1. Quality and Significance of the four levels of evidence in GRADE

Table 2. Factors that determine How to upgrade or downgrade the quality of evidence
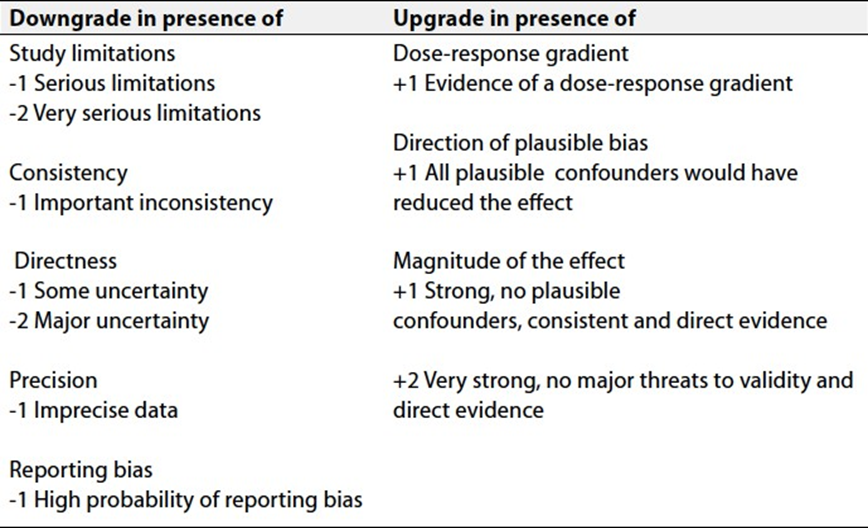
Table 3. ASA score, (7)
- References
1. Singh K, Singh R, Singh S, et al. Secondary Bacterial Peritonitis: A Review of Pathophysiology, Diagnosis, and Management. J Clin Diagn Res. 2015 Nov;9(11):OE01-5.
2. Bajaj JS, O'Leary JG, Tandon P, et al. Potential preventability of spontaneous bacterial peritonitis. Clin Gastroenterol Hepatol. 2012 Oct;10(10):1150-5.
3. Philippe M, Herve´ D, Marc L etal. Guidelines for management of intra-abdominal infections. Anaesth Crit Care Pain Med 34 (2015) 117–130.
4. Solomkin JS, Mazuski JE, Bradley JS, et al. Diagnosis and management of complicated intra-abdominal infection in adults and children: guidelines by the Surgical Infection Society and the Infectious Diseases Society of America. Clin Infect Dis. 2010 Jan 15;50(2):133-64.
5. Li PKT, Chow KM, Cho Y, et al. ISPD peritonitis guideline recommendations: 2022 update on prevention and treatment. Perit Dial Int. 2022 Mar;42(2):110-153.
6. Sartelli M, Catena F, Abu-Zidan FM, et al. Management of intra-abdominal infections: recommendations by the Italian council for the optimization of antimicrobial use. World J Emerg Surg. 2024 Jan 18;19(1):10.
7. Horvath B, Kloesel B, Todd MM, Cole DJ, and Prielipp RC: The evolution, current value, and future of the American Society of Anesthesiologists Physical Status Classification System. Anesthesiology 2021; 135:904–19.