NIPPLE DISCHARGE
| Site: | EHC | Egyptian Health Council |
| Course: | Radiology Guidelines |
| Book: | NIPPLE DISCHARGE |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:52 AM |
Description
"last update: 2 April 2026" Download Guideline
- Executive Summary
The scope of this guideline is concerned with imaging of female patients in the adult age group >18 years presenting with nipple discharge either physiologic or pathologic.
The recommendations according to the type of nipple discharge as well as the age group of the patient are as follows:
ADULT FEMALE, PHYSIOLOGIC NIPPLE DISCHARGE:
1. Digital Mammography:
If clinical findings are consistent with physiologic nipple discharge, routine screening mammography for patients 40 years of age or older is recommended. No further radiologic investigation is required and diagnostic breast imaging is unnecessary (3).
Strong recommendation
2. Image-guided Core Biopsy/ Fine Needle Aspiration:
We recommend against the use of image guided core biopsy or fine needle aspiration in these cases.
Strong recommendation
ADULT FEMALE, 40 YEARS OF AGE OR OLDER, PATHOLOGIC NIPPLE DISCHARGE:
1. Digital Mammography:
Mammography is the initial imaging modality in this age group. Both breasts should be imaged in two views; craniocaudal and mediolateral oblique views enabling the entire breast to be completely screened for any additional lesions. Additional mammographic views with spot compression and magnification are required for better evaluation of the retroareolar region when necessary (6).
Strong recommendation
2. Digital Breast Tomosynthesis:
It is recommended as an initial diagnostic tool if available. Many studies have proposed the use of DBT in assessing nipple discharge (7). It has also been proven that the sensitivity of DBT is higher than that of mammography with better detection rate and diagnosis of breast cancer.
Strong recommendation
3. Breast ultrasound:
Breast US is recommended as a complementary tool for mammography in initial evaluation and assessing extent of involvement.
US is also an important tool to guide for biopsy and localization for surgical excision.
Strong recommendation
4. MRI Breast:
MRI is not recommended as initial imaging for patients with pathologic nipple discharge. in patients with pathologic nipple discharge and negative mammography/ ultrasound, MRI is recommended to detect lesions of concern preoperatively. Imaging follow-up can be safely recommended for patients with normal MRI findings, thus avoiding unnecessary surgical duct excision (8).
Strong recommendation
5. Contrast enhanced mammography:
With CEM, lesions could be identified according to their density, morphologic as well as enhancement characteristics (9). It is currently being used for diagnostic purposes especially in dense breasts, preoperative evaluation, assessment of extent of disease and MRI contraindication. Ongoing studies also propose future directions in breast cancer screening (10). It may be considered in patients with pathologic nipple discharge and negative mammography/ ultrasound according to its availability.
Conditional recommendation
6. Image-Guided Core Biopsy:
Image-guided core needle biopsy is not recommended for initial evaluation of patients with pathologic nipple discharge.
Biopsy procedures can be guided by stereotactic mammography, contrast enhanced mammography, or US, depending on the imaging modality which best depicts the abnormality detected.
Vacuum-assisted core biopsy (VAB) is useful in complete sampling/ removing of small intraductal papillary lesions which may be therapeutic and can lead to cessation of nipple discharge in 90-97.2% of cases (11). Surgical major duct excision remains the gold standard for exclusion of malignancy in patients with unremarkable imaging.
Strong recommendation
7. Image-Guided Fine Needle Aspiration:
It is not recommended as the initial examination for evaluation of pathologic nipple discharge. Studies have shown that core needle biopsy is superior to fine needle aspiration (FNA) regarding accuracy and precise histological grading of breast cancer (12).
Conditional recommendation
ADULT FEMALE, 30 to 39 YEARS OF AGE, PATHOLOGIC NIPPLE DISCHARGE:
Same guidelines as adult female 40 years or older with the following consideration;
US is recommended as the initial imaging modality in this age group with the addition of mammography when necessary.
Strong recommendation
ADULT FEMALE, YOUNGER THAN 30 YEARS OF AGE, PATHOLOGIC NIPPLE DISCHARGE:
1. Breast ultrasound:
US is the initial imaging modality of choice in this age group.
Strong recommendation
2. Digital Mammography or Digital Breast Tomosynthesis:
Diagnostic mammography or DBT is beneficial if US shows a suspicious finding.
Strong recommendation
3. Contrast studies (contrast enhanced mammography/MRI):
Not recommended as initial imaging for patients with pathologic nipple discharge.
In women with dense breasts, CEM or contrast MRI should be recommended for proper staging and to exclude multicentricity/bilaterality.
Conditional recommendation
4. Image-guided Core Biopsy
If an imaging abnormality is seen, US can be used to guide biopsy.
Good practice statement
- RECOMMENDATIONS
ADULT FEMALE, PHYSIOLOGIC NIPPLE DISCHARGE:
1. Digital Mammography:
2. If clinical findings are consistent with physiologic nipple discharge, routine screening mammography for patients 40 years of age or older is recommended. No further radiologic investigation is required and diagnostic breast imaging is unnecessary (3).
Remarks: Physiologic nipple discharge tends to occur with compression. Studies have proposed being mostly related to benign causes, with no association with insitu or invasive carcinoma (19-21).
Strong recommendation
High-Quality Evidence (studies well-designed and account for most common biases) (19-21).
3. Image-guided Core Biopsy/ Fine Needle Aspiration:
We recommend against the use of image guided core biopsy or fine needle aspiration in these cases.
Strong recommendation
High-Quality Evidence (studies well-designed and account for most common biases) (20, 21).
ADULT FEMALE, 40 YEARS OF AGE OR OLDER, PATHOLOGIC NIPPLE DISCHARGE:
1. Digital Mammography:
Mammography is the initial imaging modality in this age group. Both breasts should be imaged in two views; craniocaudal and mediolateral oblique views enabling the entire breast to be completely screened for any additional lesions. Additional mammographic views with spot compression and magnification are required for better evaluation of the retroareolar region when necessary (6).
Remarks: Despite its low sensitivity (15-68%) and positive predictive value (PPV) 42% for detection of malignancy as the cause of nipple discharge, mammography still plays an important role in breast imaging because of its high specificity (38-98%) and high negative predictive value (NPV) 90% (19-22).
Strong recommendation
High-Quality Evidence (studies well-designed and account for most common biases) (19-22).
2. Digital Breast Tomosynthesis:
It is recommended as an initial diagnostic tool if available. Many studies have proposed the use of DBT in assessing nipple discharge (7). It has also been proven that the sensitivity of DBT is higher than that of mammography with better detection rate and diagnosis of breast cancer.
Remarks: This is possibly due to the reduced risk of obscuration of pathology by the overlapping breast tissues as the reconstructed DBT slices are demonstrated similarly to computed tomography reconstructed slices (5). DBT has the potential to increase workflow efficiency in a symptomatic setting by reducing benign biopsies (23).
Strong recommendation
High-Quality Evidence (studies well-designed and account for common biases) (7,23).
3. Breast ultrasound:
Breast US is recommended as a complementary tool for mammography in initial evaluation and assessing extent of involvement.
US is also an important tool to guide for biopsy and localization for surgical excision.
Remarks: Studies propose a sensitivity of 56%–80%, specificity of 61%–75%, PPV of 29%–39%, and NPV of 90%–91% for US alone in detecting underlying malignancy in patients with pathologic nipple discharge (5,19,23). False-positive results may be attributed to ductal wall volume averaging in a tortuous duct, intraductal/periductal fibrosis, blood clots, or inspissated secretions (24).
Strong recommendation
High-Quality Evidence (review and observational studies well-designed and account for common biases) (19-21, 24-26).
4. MRI Breast:
MRI is not recommended as initial imaging for patients with pathologic nipple discharge. in patients with pathologic nipple discharge and negative mammography/ ultrasound, MRI is recommended to detect lesions of concern preoperatively. Imaging follow-up can be safely recommended for patients with normal MRI findings, thus avoiding unnecessary surgical duct excision (8).
Remarks: MRI should only be reserved for cases that are occult in mammography and US with failure to identify an underlying cause of pathologic nipple discharge (27,28).
Results shown by many studies with regards to breast MRI in the detection of the cause of pathologic nipple discharge revealed a sensitivity of 86-100% for invasive cancer and 40% to 100% for noninvasive disease (29-32).
Strong recommendation
High-Quality Evidence (systematic review and observational studies well-designed and account for common biases) (27)
5. Contrast enhanced mammography:
With CEM, lesions could be identified according to their density, morphologic as well as enhancement characteristics (9). It is currently being used for diagnostic purposes especially in dense breasts, preoperative evaluation, assessment of extent of disease and MRI contraindication. Ongoing studies also propose future directions in breast cancer screening (10). It may be considered in patients with pathologic nipple discharge and negative mammography/ ultrasound according to its availability.
Remarks: Recent studies have proposed that the level of radiation exposure from CEM is similar to and within the range of radiation doses that patients are exposed to from other mammography examinations (33).
A study by Fakhry et al., 2022 in role of CEM for evaluation of pathological nipple discharge revealed a higher diagnostic accuracy of 79.3% compared to mammography and ultrasound 76.3% (34).
Conditional recommendation
Moderate-Quality Evidence (observational study) (34).
6. Image-Guided Core Biopsy:
Image-guided core needle biopsy is not recommended for initial evaluation of patients with pathologic nipple discharge.
Biopsy procedures can be guided by stereotactic mammography, contrast enhanced mammography, or US, depending on the imaging modality which best depicts the abnormality detected.
Vacuum-assisted core biopsy (VAB) is useful in complete sampling/ removing of small intraductal papillary lesions which may be therapeutic and can lead to cessation of nipple discharge in 90-97.2% of cases (11). Surgical major duct excision remains the gold standard for exclusion of malignancy in patients with unremarkable imaging.
Remarks: Core biopsy is preferred over FNA because the larger gauge needle improves sampling (12, 35). Studies have shown reported rates of papillomas upgrade to malignancy between 3% and 14% (36).
Strong recommendation
High-Quality Evidence (randomized controlled clinical trial and observational studies well-designed and account for common biases) (11,26,32,35-37)
7. Image-Guided Fine Needle Aspiration:
It is not recommended as the initial examination for evaluation of pathologic nipple discharge. Studies have shown that core needle biopsy is superior to FNA regarding accuracy and precise histological grading of breast cancer (12).
Remarks: Breast FNA can be done to assess lesions difficult to access by core biopsy or lymph nodes. Hypocellular cystic and fibrotic lesions, degenerative changes, necrosis and epithelial hyperplasia make evaluating the smear difficult. False negative results are common in special types of carcinomas, like lobular and tubular, and false positive results may be seen with lactation and therapy (38).
Breast FNA is safe, fast, cost effective and almost complication free. However, it mostly doesn't give a definite diagnosis, can't differentiate between atypical ductal hyperplasia and low grade DCIS or between high grade DCIS and invasive cancer. Moreover, its accuracy is influenced by the expertise of the aspirator as well as the reader giving equivocal, false positive and false negative results. This is often due to poor sampling technique, poor tumor localization, small tumor size and nonpalpable breast lesions (39).
Conditional recommendation
Moderate-Quality Evidence (randomized controlled clinical trial, review and observational studies) (12,35,38,39).
ADULT FEMALE, 30 to 39 YEARS OF AGE, PATHOLOGIC NIPPLE DISCHARGE:
Same guidelines as adult female 40 years or older with the following consideration;
US is recommended as the initial imaging modality in this age group with the addition of mammography when necessary.
Remarks: It has been proven that US has a higher sensitivity of 95.7% than that of mammography 60.9% for the detection of breast cancer in females 30 to 39 years of age (40,41).
However, mammography remains the gold standard owing to its ability to detect microcalcifications, taking into consideration that patients with DCIS commonly present with nipple discharge.
Strong recommendation
High-Quality Evidence (studies well-designed and account for common biases) (40,41).
ADULT FEMALE, YOUNGER THAN 30 YEARS OF AGE, PATHOLOGIC NIPPLE DISCHARGE:
1. Breast ultrasound:
US is the initial imaging modality of choice in this age group.
Remarks: A study by Yue et al where US was done for 692 of 955 symptomatic females aged <25 years, showed that 21 patients only had indeterminate findings. No cancer was detected in this group after biopsy or clinical follow-up (42).
Strong recommendation
High-Quality Evidence (study well-designed and account for common biases) (42).
2. Digital Mammography or Digital Breast Tomosynthesis:
Diagnostic mammography or DBT are beneficial if US shows a suspicious finding.
Remarks: DBT demonstrates the true extent of the lesion and exclude contralateral abnormalities particularly in young women with dense breasts (43).
Strong recommendation
High-Quality Evidence (study well-designed and account for common biases) (7,23,43).
3. Contrast studies (contrast enhanced mammography/MRI):
Not recommended as initial imaging for patients with pathologic nipple discharge.
In women with dense breasts, CEM or contrast MRI should be recommended for proper staging and to exclude multicentricity/bilaterality.
Remarks: CEM provides superior imaging performance compared to standard mammography and is considered a relatively affordable accessible imaging tool with a sensitivity approaching MRI.
Conditional recommendation
Moderate-Quality Evidence (systematic review and observational studies) (44,45).
4. Image-guided Core Biopsy
If an imaging abnormality is seen, US can be used to guide biopsy.
Remarks: To our knowledge, no relevant evidence in the literature exists for evaluation of pathologic nipple discharge in this age group.
Good practice statement
- Acknowledgement
We would like to acknowledge the Egyptian Health Council, Committee of National Egyptian Guidelines, and Radiology Scientific Committee for adapting this Guidelines.
Chair of the Panel:
Tarek El-Diasty
Scientific Group Members:
Anan Atef, Dalia Ahmed, Ekram Hamed, Heba El-Kassas, Ihab Reda, Khaled Matrawy, Maha Hussein Helal, Mohamed Fawzi, Mohamed Shaker, Ragab Hani, Rasha Kamal, Sahar Mansour, Samar El-Maadawy, Rana Hussein, Wael Elshawaf, Wahid Tantawy.
- Abbreviations
CEM: Contrast enhanced mammography
DM: Digital mammography
DBT: Digital breast tomosynthesis
DCIS: Ductal carcinoma insitu
FNA: Fine needle aspiration
MRI: Magnetic Resonance Imaging
NPV: Negative predictive value
PPV: Positive predictive value
US: Ultrasound
VAB: Vacuum-assisted core biopsy
WHO: World Health Organization
- Glossary
Nipple discharge: Liquid leak from the nipples spontaneously or when they are compressed. It can be clear, white, yellow, green or bloody. Nipple discharge may be physiologic, but sometimes it can be pathologic representing a sign of an underlying benign or malignant condition, including a breast cyst or breast cancer (3).
Contrast-enhanced mammography: Contrast-enhanced mammography (CEM) is an emerging imaging diagnostic tool that uses iodinated contrast media for the visualization of breast neovascularity in a fashion similar to magnetic resonance imaging (MRI) (4).
Core biopsy: a biopsy in which a cylindrical sample of tissue is obtained (as from a kidney or breast) by a hollow needle.
Digital breast tomosynthesis: Tomosynthesis or “3D” mammography is a new type of digital x-ray mammogram which creates 2D and 3D-like pictures of the breasts. This tool improves the ability of mammography to detect early breast cancers and decreases the number of women “called back” for additional tests for findings that are not cancers (5).
Fine needle aspiration: the process of obtaining a sample of cells and bits of tissue for examination by applying suction through a fine needle attached to a syringe.
Magnetic resonance imaging (MRI): a technique that produces computerized images of internal body tissues using a powerful magnetic field and pulses of radio waves.
Mammography: X-ray examination of the breasts.
Ultrasound: the diagnostic or therapeutic use of ultrasound and especially a noninvasive technique involving the formation of a two-dimensional image used for the examination and measurement of internal body structures and the detection of bodily abnormalities.
- Introduction
In Egypt, based on the updated statistics according to the World Health Organization (WHO) 2022 for breast cancer, there is 26 845 new case each year among females with a crude incidence rate of 51.1/100 000 normal population and an age standardized incidence rate of 55.4/100 000 normal population. Thus, according to these data, breast cancer is the most common malignancy among females representing 34.9 % of female cancers (13,14). Nipple discharge is the third most common breast symptom after pain and palpable lump (15). It is categorized as physiologic or pathologic.
Physiologic nipple discharge may be white, green, or yellow in color. It is typically non-spontaneous, bilateral, and occurs in multiple ducts (16). Several studies have revealed that it is mostly benign with no evidence of insitu or invasive carcinoma (17). Examples include premenopausal women experiencing milky nipple discharge unrelated to normal breastfeeding known as galactorrhea, as well as duct ectasia as a manifestation of normal involution of the breast with stagnant secretions that may accumulate (3).
Pathologic nipple discharge may be associated with benign or malignant causes. It is typically unilateral, spontaneous, originating from a single duct orifice, and is often clear or blood-stained (16). Patient demographics such as age, help determine the degree of suspicion of the pathologic nipple discharge. If associated with palpable abnormality, nipple eczema or retraction, diagnostic imaging should be considered (18). The most common cause of pathologic nipple discharge is papillomas followed by duct ectasia, postoperative fluid collection, and retroareolar needle biopsy. Breast cancer accounts for 5–15% of cases, the most common type being ductal carcinoma in situ (19).
Following a comprehensive clinical breast examination, often conducted by the referring physician or a board-certified breast clinician, the radiologist must demonstrate concordance between the imaging findings and the clinical data (1,2).
- Scope and purpose
The scope of this guideline is concerned with imaging of female patients in the adult age group >18 years presenting with nipple discharge either physiologic or pathologic.
The purpose is to guide healthcare providers in accurately evaluating female patients presenting with nipple discharge, by a multimodality imaging approach in a timely manner and to differentiate between benign and suspicious cases that need to be further evaluated.
- The target audience
· Radiologists
· General surgeons
· Breast surgeons
· Oncology surgeons
· Medical oncologists
· Obstetricians and gynecologists
- Methods
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation.
inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting only national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated 2015 and later)
• Selecting peer reviewed publications only
• Selecting guidelines written in English language
• Excluding guidelines written by a single author not on behalf of an organization in order to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
• Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
The following characteristics of the retrieved guidelines were summarized in a table:
• Developing organisation/authors
• Date of publication, posting, and release
• Country/language of publication
• Date of posting and/or release
• Dates of the search used by the source guideline developers
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided a cut-off point or rank the guidelines (any guideline scoring above 50% on the rigour dimension was retained). These guidelines were adapted mainly from American College of Radiology ACR Appropriateness Criteria®, Evaluation of Nipple Discharge update in 2022 with partial incorporation of insights from reputable sources to enhance its comprehensiveness and applicability (1,2).
- Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available on the following sites:
■ GRADE working group: https://www.gradeworkinggroup.org
■ GRADE online training modules: http://cebgrade.mcmaster.ca/
Table 1 Quality of evidence in GRADE
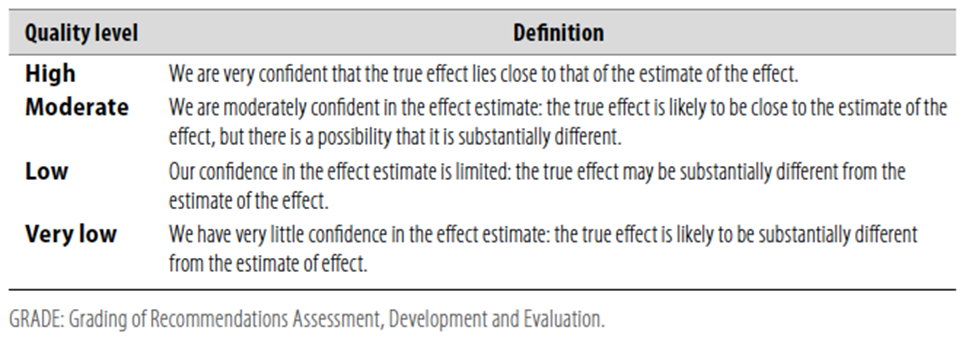
Table 2 Significance of the four levels of evidence
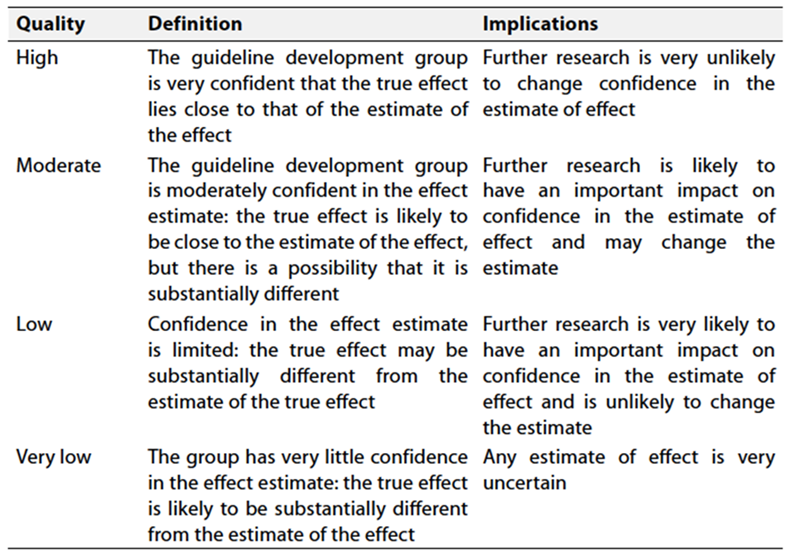
Table 3 Factors that determine How to upgrade or downgrade the quality of evidence.
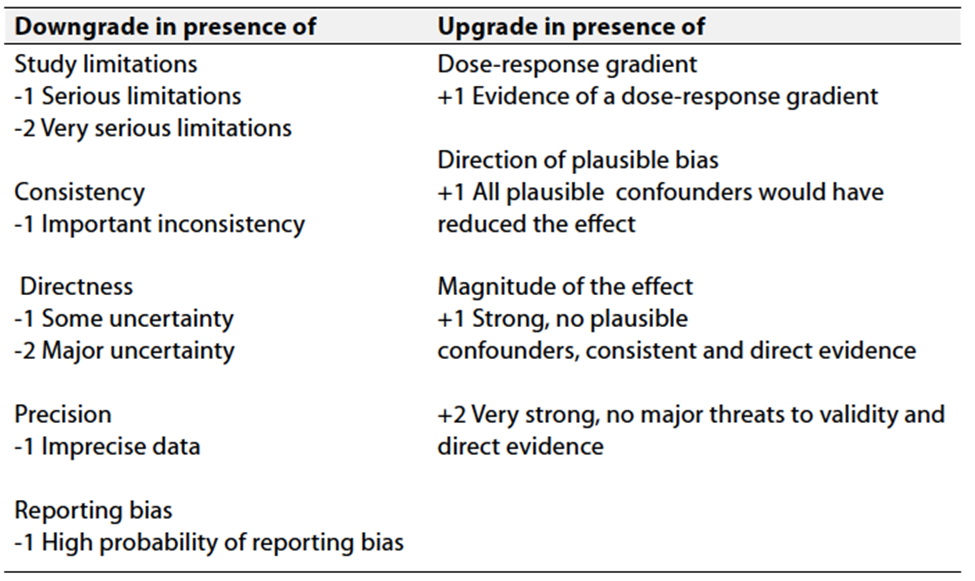
The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation.
Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Clinical indicators for monitoring
Patient file review for the following:
· Ultrasound imaging in all patients presenting with nipple discharge.
· Mammography imaging in all patients 40 years of age or older and in young patients if US shows a suspicious finding.
· Image guided core biopsy procedure in case of suspicious imaging.
· The physician should document follow-up of patients with BIRADS 3 lesions and educate the patient for the importance and the need for follow-up.
These indicators cover aspects such as documentation, diagnostic procedures, and patient education, providing a comprehensive approach to monitoring physician adherence to the clinical guidelines.
- Updating the guideline
To keep these recommendations up to date and ensure its validity it will be periodically updated. This will be done whenever strong new evidence is available and necessitates updating.
- Research Gaps
1. Prevalence of nipple discharge among patients with breast cancer
2. Role of contrast enhanced mammography in evaluation of nipple discharge
3. Contrast enhanced mammography in patients with pathologic nipple discharge: a comparison with breast MRI
- REFERENCES
1. Sanford MF, Slanetz PJ, Lewin AA, Baskies AM, Bozzuto L, Branton SA, et al. ACR appropriateness criteria® Evaluation of Nipple Discharge: 2022 update. Journal of the American College of Radiology. 2022 Nov;19(11). doi:10.1016/j.jacr.2022.09.020
2. [NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Breast Cancer Screening and Diagnosis Version 2.2024]. [cited 2024 Jul 12]. Available from: https://www.nccn.org/professionals/physician_gls/pdf/breast-screening.pdf
3. Gupta D, Mendelson EB, Karst I. Nipple discharge: Current clinical and imaging evaluation. American Journal of Roentgenology. 2021 Feb;216(2):330–9. doi:10.2214/ajr.19.22025
4. Patel BK, Lobbes MBI, Lewin J (2018) Contrast enhanced spectral mammography: A review. Seminars in ultrasound, CT, and MRI 39:70-79. Doi:10.1053/j.sult.2017.08.005
5. Skaane P, Bandos AI, Gullien R, Eben EB, Ekseth U, Haakenaasen U, et al. Comparison of Digital Mammography alone and Digital Mammography Plus Tomosynthesis in a population-based screening program. Radiology. 2013 Apr;267(1):47–56. doi:10.1148/radiol.12121373
6. Alcock C, Layer GT. Predicting occult malignancy in nipple discharge. ANZ Journal of Surgery. 2010 Sept;80(9):646–9. doi:10.1111/j.1445-2197.2010.05270.x
7. Whelehan P, Ali K, Vinnicombe S, Ball G, Cox J, Farry P, et al. Digital Breast Tomosynthesis: Sensitivity for cancer in younger symptomatic women. The British Journal of Radiology. 2021 Mar 1;94(1119):20201105. doi:10.1259/bjr.20201105
8. Boisserie-Lacroix M, Doutriaux-Dumoulin I, Chopier J, Boyer B, Depetiteville M-P, Hoppe S, et al. Diagnostic accuracy of breast MRI for patients with suspicious nipple discharge and negative mammography and ultrasound: A prospective study. European Radiology. 2021 Apr 13;31(10):7783–91. doi:10.1007/s00330-021-07790-4
9. Lewin JM, Patel BK, Tanna A. Contrast-Enhanced Mammography: A scientific review. Journal of Breast Imaging. 2019 Dec 12;2(1):7–15. doi:10.1093/jbi/wbz074
10. Jochelson MS, Lobbes MB. Contrast-Enhanced Mammography: State of the art. Radiology. 2021 Apr;299(1):36–48. doi:10.1148/radiol.2021201948
11. Dennis MA, Parker S, Kaske TI, Stavros AT, Camp J. Incidental treatment of nipple discharge caused by benign intraductal papilloma through diagnostic mammotome biopsy. American Journal of Roentgenology. 2000 May;174(5):1263–8. doi:10.2214/ajr.174.5.1741263
12. Garg S, Mohan H, Bal A, Attri AK, Kochhar S. A comparative analysis of core needle biopsy and fine‐needle aspiration cytology in the evaluation of palpable and mammographically detected suspicious breast lesions. Diagnostic Cytopathology. 2007 Oct 8;35(11):681–9. doi:10.1002/dc.20721
13. Ibrahim AS, Khaled HM, Mikhail NN, Baraka H, Kamel H. Cancer incidence in Egypt: Results of the National Population-Based Cancer Registry Program. Journal of Cancer Epidemiology. 2014;2014:1–18. doi:10.1155/2014/437971
14. Egypt - International Agency for Research on Cancer [Internet]. [cited 2024Jun27]. Available from: https://gco.iarc.fr/today/data/factsheets/populations/818-egypt-fact-sheets.pdf
18. Li GZ, Wong SM, Lester S, Nakhlis F. Evaluating the risk of underlying malignancy in patients with pathologic nipple discharge. The Breast Journal. 2018 Mar 8;24(4):624–7. doi:10.1111/tbj.13018
19. Bahl M, Baker JA, Greenup RA, Ghate SV. Diagnostic value of ultrasound in female patients with nipple discharge. American Journal of Roentgenology. 2015 Jul;205(1):203–8. doi:10.2214/ajr.14.13354
20. Ashfaq A, Senior D, Pockaj BA, Wasif N, Pizzitola VJ, Giurescu ME, et al. Validation study of a modern treatment algorithm for nipple discharge. The American Journal of Surgery. 2014 Aug;208(2):222–7. doi:10.1016/j.amjsurg.2013.12.035
21. Sabel MS, Helvie MA, Breslin T, Curry A, Diehl KM, Cimmino VM, et al. Is duct excision still necessary for all cases of suspicious nipple discharge? The Breast Journal. 2011 Dec 30;18(2):157–62. doi:10.1111/j.1524-4741.2011.01207.x
22. Cabioglu N, Hunt KK, Singletary ES, Stephens TW, Marcy S, Meric F, et al. Surgical decision making and factors determining a diagnosis of breast carcinoma in women presenting with nipple discharge. Journal of the American College of Surgeons. 2003 Mar;196(3):354–64. doi:10.1016/s1072-7515(02)01606-x
23. Bansal GJ, Young P. Digital Breast Tomosynthesis within a symptomatic “one-stop breast clinic” for characterization of subtle findings. The British Journal of Radiology. 2015 Sept;88(1053):20140855. doi:10.1259/bjr.20140855
24. Labib PL, Gallegos N, Hegarty D. Nipple discharge. BMJ. 2015 Jul 21; doi:10.1136/bmj.h3123
25. Sarica O, Zeybek E, Ozturk E. Evaluation of nipple-areola complex with ultrasonography and magnetic resonance imaging. Journal of Computer Assisted Tomography. 2010 Jul;34(4):575–86. doi:10.1097/rct.0b013e3181d74a88
26. Morrogh M, Park A, Elkin EB, King TA. Lessons learned from 416 cases of nipple discharge of the breast. The American Journal of Surgery. 2010 Jul;200(1):73–80. doi:10.1016/j.amjsurg.2009.06.021
27. Lubina N, Schedelbeck U, Roth A, Weng AM, Geissinger E, Hönig A, et al. 3.0 Tesla Breast Magnetic Resonance Imaging in patients with nipple discharge when mammography and Ultrasound Fail. European Radiology. 2014 Nov 30;25(5):1285–93. doi:10.1007/s00330-014-3521-2
28. Avdan Aslan A, Gültekin S. What is the role of Breast MRI in the management of women with pathologic nipple discharge and normal conventional imaging? Irish Journal of Medical Science (1971 -). 2022 Nov 21;192(5):2331–5. doi:10.1007/s11845-022-03230-4
29. Lorenzon M, Zuiani C, Linda A, Londero V, Girometti R, Bazzocchi M. Magnetic resonance imaging in patients with nipple discharge: Should we recommend it? European Radiology. 2010 Nov 30;21(5):899–907. doi:10.1007/s00330-010-2009-y
30. Nakahara H, Namba K, Watanabe R, Furusawa H, Matsu T, Akiyama F, et al. A comparison of Mr Imaging, galactography and ultrasonography in patients with nipple discharge. Breast Cancer. 2003 Oct;10(4):320–9. doi:10.1007/bf02967652
31. van Gelder L, Bisschops RH, Menke‐Pluymers MB, Westenend PJ, Plaisier PW. Magnetic resonance imaging in patients with unilateral bloody nipple discharge; useful when conventional diagnostics are negative? World Journal of Surgery. 2014 Aug 15;39(1):184–6. doi:10.1007/s00268-014-2701-1
32. Bahl M, Baker JA, Greenup RA, Ghate SV. Evaluation of pathologic nipple discharge: What is the added diagnostic value of MRI? Annals of Surgical Oncology. 2015 Aug 7;22(S3):435–41. doi:10.1245/s10434-015-4792-9
33. Phillips J, Mihai G, Hassonjee SE, Raj SD, Palmer MR, Brook A, et al. Comparative dose of contrast-enhanced spectral mammography (CESM), Digital Mammography, and Digital Breast Tomosynthesis. American Journal of Roentgenology. 2018 Oct;211(4):839–46. doi:10.2214/ajr.17.19036
34. Fakhry S, Abdel Rahman RW, Shaalan HS, Hassan MH, Tealab SH, Sayed SB. The added role of contrast-enhanced spectral mammography in the evaluation of pathological nipple discharge. Egyptian Journal of Radiology and Nuclear Medicine. 2022 Apr 12;53(1). doi:10.1186/s43055-022-00766-4
35. Homesh NA, Issa MA, El-Sofiani HA. The diagnostic accuracy of fine needle aspiration cytology versus core needle biopsy for palpable breast lump(s). Saudi Med J 2005;26:42-6.
36. Liberman L, Tornos C, Huzjan R, Bartella L, Morris EA, Dershaw DD. Is surgical excision warranted after benign, concordant diagnosis of papilloma at percutaneous breast biopsy? AJR Am J Roentgenol. 2006;186(5):1328-1334
37. Rissanen T, Reinikainen H, Apaja-Sarkkinen M. Breast sonography in localizing the cause of nipple discharge. Journal of Ultrasound in Medicine. 2007 Aug;26(8):1031–9. doi:10.7863/jum.2007.26.8.1031
38. Mendoza P, Lacambra M, Tan P-H, Tse GM. Fine needle aspiration cytology of the breast: The nonmalignant categories. Pathology Research International. 2011 May 19;2011:1–8. doi:10.4061/2011/547580
39. Cytology [Internet]. [cited 2024 Jul 15]. Available from: https://www.pathologyoutlines.com/topic/breastcytology.html
40. Lehman CD, Lee CI, Loving VA, Portillo MS, Peacock S, DeMartini WB. Accuracy and value of breast ultrasound for primary imaging evaluation of symptomatic women 30-39 years of age. American Journal of Roentgenology. 2012 Nov;199(5):1169–77. doi:10.2214/ajr.12.8842
41. Osako T, Iwase T, Takahashi K, lijima K, Miyagi Y, Nishimura S, et al. Diagnostic mammography and ultrasonography for palpable and nonpalpable breast cancer in women aged 30 to 39 years. Breast Cancer. 2007 Jul;14(3):255–9. doi:10.2325/jbcs.14.255
42. Yue D, Swinson C, Ravichandran D. Triple assessment is not necessary in most young women referred with breast symptoms. The Annals of The Royal College of Surgeons of England. 2015 Sept 1;97(6):466–8. doi:10.1308/rcsann.2015.0019
43. Lee WK, Chung J, Cha E, Lee JE, Kim JH (2016) Digital breast tomosynthesis and breast ultrasound: Additional roles in dense breasts with category 0 at conventional digital mammography. European journal of radiology 85:291-296. Doi:10.1016/j.ejrad.2015.09.026
44. Pötsch N, Vatteroni G, Clauser P, Helbich TH, Baltzer PAT (2022) Contrast-enhanced mammography versus contrast-enhanced breast MRI: A systematic review and meta-analysis. Radiology 305:94. Doi:10.1148/radiol.212530
45. Coffey K, Jochelson MS (2022) Contrast-enhanced mammography in breast cancer screening. European journal of radiology 156:110513. Doi:10.1016/j.ejrad.2022.110513