the Management of Neutropenia in Pediatric Age Groups
| Site: | EHC | Egyptian Health Council |
| Course: | Pediatrics Guidelines |
| Book: | the Management of Neutropenia in Pediatric Age Groups |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:52 AM |
Description
"last update: 6 April 2026" Download Guideline
- Executive Summary
Neutropenia is a reduction in the ANC below the lower limit of the normal range for the age and ethnic origin of the affected subject. It can be classified as congenital or acquired. Neutropenic individuals are at increased risk of infection. The greatest susceptibility of infection is seen with ANC below 500 /mm3. Real world data have shown that the work-up of patients with neutropenia is mostly based on the physicians’ experience and local practices rather than on the guided clinical and laboratory evidence. Thus, the diagnosis and monitoring of neutropenic patients remain varied and challenging.
This guideline focuses on the prevention and management of neutropenia in pediatric age groups.
After reviewing all the inclusion and exclusion criteria and quality appraisal results, the GDG/ GAG recommended using the following source original clinical practice guidelines (CPGs):
1. Guideline for Managing Fever and Neutropenia in Pediatric Patients With Cancer and Hematopoietic Cell Transplantation Recipients: 2023 Update. ASCO (2023)
2. The European Guidelines on Diagnosis and Management of Neutropenia in Adults and Children: A Consensus Between the European Hematology Association and the EuNet-INNOCHRON COST Action. EHA-EuNet-INNOCHRON (2023)
3. Guideline for Antibacterial Prophylaxis Administration in Pediatric Cancer and Hematopoietic Stem Cell Transplantation. IDSA (2020)
4. Guideline for the Management of Clostridium Difficile Infection in Children and Adolescents With Cancer and Pediatric Hematopoietic Stem-Cell Transplantation Recipients. ASCO (2018)
We conducted Adolopment for these guidelines: (Adoption, Adaptation, and Development)
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statements
Recommendations and Good Practice Statements (GPS)
This version of the CPG includes recommendations and good practice statements on the following three sub-sections:
A. Diagnosis of Neutropenia in children
The guideline covers pediatric patients suffering from primary or secondary neutropenia.
This guideline emphasizes definitions of neutropenia in the different age groups with different etiopathogenesis as well as the criteria for the definition of febrile neutropenia.
B. Management of Neutropenia.
This section includes recommendations and good practice statements on first- and second-line investigations in cases of febrile and afebrile neutropenia. This section also highlights recommendations on the different pharmacological and non-pharmacological therapies for children with neutropenia.
C. Prevention of infections in children with neutropenia.
This section outlines the main lines and recommendations in preventing infections in children with neutropenia.
We can summarize the guidelines’ recommendations for the management of pediatric neutropenia in the following:
We recommend the use of the following definition of neutropenia
- The definition of neutropenia varies according to the patient’s ethnic origin and age. SoR: GPS
- At term neonates: Neutropenia is defined as an absolute neutrophil count (ANC) level of 2.5 × 10⁹/L for term/near-term neonates 72–240 hours following delivery. SoR: GPS
- In preterm newborns: Neutropenia is defined as an ANC level of 1.0 × 10⁹/L for preterm neonates. SoR: GPS
- Infants and Children: The widely accepted cutoff level of ANC for the definition of neutropenia in Caucasian newborns and infants up to the age of 1 year is 1.0 × 10⁹/L. SoR: GPS
- From the age of 1 year to adulthood the cutoff level for neutropenia is 1.5 × 10⁹/L. SoR: GPS
We recommend the use of the following classification of neutropenia (severity)
- Neutropenia is classified as mild when ANC is between 1.0 and 1.5 × 10⁹/L. SoR: GPS
- Moderate when ANC is 0.5 to 1.0 × 10⁹/L. SoR: GPS
- Severe when ANC is <0.5 × 10⁹/L. SoR: GPS
- The term agranulocytosis is used for severe neutropenia with ANC <0.2 × 10⁹/L. SoR: GPS
We recommend the use of the following classification of neutropenia (duration)
- Neutropenia is characterized as acute when duration is <3 months. SoR: GPS
- Chronic when duration is >3 months. SoR: GPS
We recommend the use of the following classification of neutropenia (etiology)
- An extended, pathogenesis-based classification categorizes neutropenia as congenital versus acquired and likely acquired. SoR: GPS
We recommend the use of the following congenital neutropenia and its classification
- Congenital neutropenia (CN) comprises a group of genetic diseases characterized by impaired production, differentiation, and survival of neutrophils in the bone marrow (BM), susceptibility to infections, and increased propensity to MDS/AML transformation. SoR: GPS
- CN can be further subclassified into disorders where neutropenia is the only abnormality and those where neutropenia is associated with extra-hematological manifestations, immunodeficiency/immune dysregulation, metabolic disorders and nutritional deficiencies, or as part of more complex BM failure syndromes. SoR: GPS
- The classification also takes into consideration the genes that have been identified as responsible for each CN subtype. SoR: GPS
- Individuals of African and Middle Eastern descent display normal ANCs in the range from 0.5 to 1.5 × 10⁹/L and less frequently even lower. SoR: GPS
- This variation, previously termed ethnic neutropenia, is usually inherited as an autosomal recessive trait associated with a polymorphism (rs2814778, −46T>C) in the GATA box in the promoter region of the ACKR1 gene (DARC). SoR: GPS
- In homozygosity (C/C), the polymorphism results in the absence of Duffy antigen expression specifically on red blood cells, a phenotype known as Duffy-null. SoR: GPS
- The guideline working group suggests the introduction of the term ACKR1/DARC-associated neutropenia (ADAN), instead of ethnic neutropenia, to emphasize the genetic rather than the ethnic basis of this entity. SoR: GPS
We recommend the use of the following causes of neutropenia in the neonatal period
- Females have ANC counts on average 2.0 × 10⁹/L higher than males. SoR: GPS
- ANC in capillary blood is on average 1.5–2.0 × 10⁹/L higher than in cord blood. SoR: GPS
- ANC is on average higher at altitude than at sea level. SoR: GPS
- Severe necrotizing enterocolitis in the newborn, especially if preterm, is frequently associated with transiently low ANCs. SoR: GPS
- Maternal tobacco smoking is associated with lower ANC. SoR: GPS
- Maternal chemotherapy results in neutropenia. SoR: GPS
- Maternal antiretroviral therapy results in neutropenia. SoR: GPS
- Maternal hypertension during pregnancy results in neutropenia. SoR: GPS
- Prenatal growth retardation is an independent risk factor for neutropenia. SoR: GPS
- Rh-hemolytic disease of the newborn is associated with neutropenia in about 50% of newborns. SoR: GPS
- Twin-twin transfusion syndrome: neutropenia always present in the donor twin. SoR: GPS
- Neutropenia is present in 67% of infants with asphyxia. SoR: GPS
Types of immune neutropenia specific to neonates
- AIN, which is unusual, but not impossible, at <1 month of age. SoR: GPS
- Neonatal alloimmune neutropenia (NAN): genetic mismatch for HNA polymorphism → maternal immunization → alloantibodies cross placenta → neutropenia in baby. SoR: GPS
- NAN secondary to maternal AIN (rarest form). SoR: GPS
Causes of acquired neutropenia
- Classified as primary/idiopathic (antineutrophil antibodies or unknown mechanisms). SoR: GPS
- Secondary due to infections, autoimmune diseases, drugs, nutritional deficiencies, hypersplenism, or hematologic diseases. SoR: GPS
Required points in history taking
- Patient history should include occurrence of infections and their frequency, type, severity, and need for hospitalization. SoR: GPS
- History of omphalitis, gingivitis, periodontitis, skin infections, abscesses, pneumonias, duration and response to antibiotics. SoR: GPS
- Presence of congenital malformations in the patient or family. SoR: GPS
- For adult patients: drug history, autoimmune disorders. SoR: GPS
- Detailed family history: ethnic origin, consanguinity, recurrent infections, neutropenia in family members, unexplained infant death or miscarriages. SoR: GPS
- Symptoms denoting autoimmune or other diseases. SoR: GPS
- History of chronic viral infections (hepatitis, HIV). SoR: GPS
- Careful inquiry about drug administration (including OTC, supplements, recreational drugs). SoR: GPS
➡️Required detailed clinical examination
- Careful clinical examination of skin, mucous membranes, respiratory tract, abdomen to exclude infection, lymphadenopathy, hepatosplenomegaly. SoR: GPS
- Examination should focus on growth, cognitive impairment, developmental delay, dysmorphism, nail/hair/skin abnormalities, bronchiectasis, hepatomegaly/splenomegaly, organ malformation, superficial veins, photophobia, nystagmus, albinism, neuropathy. SoR: GPS
- Cardiac function, enlarged lymph nodes, joint symptoms, autoimmune/metabolic/gastrointestinal/nutritional diseases should be considered. SoR: GPS
- Febrile neutropenia (FN) is defined as an oral temperature of >38.3°C or two consecutive readings of >38.0°C for 2 h and an ANC of <0.5 × 10⁹/L or expected to fall below 0.5 × 10⁹/L. SoR: GPS
- A.5.a. Approach to investigate children with neutropenia
- Patients with acute neutropenia, particularly in the presence of symptoms/signs of infection, may require immediate investigation and even hospitalization depending on the severity of neutropenia and symptoms. SoR: GPS
- For patients with chronic, isolated neutropenia without a phenotype suggestive of any underlying CN syndrome, a flowchart of basic investigation is recommended. SoR: GPS
- If the initial evaluation does not suggest ADAN, nor postinfectious or drug-induced neutropenia, the first level of investigation, possibly adjusted to the availability of the recommended tests. SoR: GPS
- A.5.b. First and second lines of investigations for children with neutropenia
- First-line investigations: Complete blood count (CBC)s, peripheral blood (PB) smear. SoR: GPS
- Biochemistry tests including liver and kidney function, c-reactive protein (CRP). SoR: GPS
- Vitamin B12 and folate. SoR: GPS
- Virology antibody screening (i.e., HepB, HepC, HIV, EBV, and CMV). SoR: GPS
- Thyroid hormones (FT3, FT4, TSH), antithyroid antibodies (anti-TG and anti-TPO). SoR: GPS
- Antineutrophil antibodies by granulocyte immunofluorescence test (GIFT) and/or granulocyte agglutination test (GAT). SoR: GPS
- Immunoglobulin levels and flow cytometric analysis of PB lymphocyte subsets. SoR: GPS
- Additional investigation in children: flow cytometric analysis of CD3+TCRα/β+CD4-CD8- (double negative) PB T lymphocytes. SoR: GPS
- Second-line investigations: CBCs in family members, serial blood counts twice a week over a period of 6 weeks to exclude CyN, copper; ceruloplasmin, anti-tissue transglutaminase IgA, deamidated gliadin peptide antibodies IgA/IgG and pancreatic isoamylase. SoR: GPS
- Additional investigation in children: rheumatoid factor (RF), antinuclear antibody (ANA), extractable nuclear antigen (ENA), and double stranded DNA (ds-DNA). SoR: GPS
- In children, young adults, and considered for adults: genetic investigations. SoR: GPS
- A.5.c. Confirming the diagnosis of congenital neutropenia
- Following negative results of first-level investigation, all patients with SCN and recurrent infections and/or family history of severe neutropenia and typical anomalies should undergo genetic work-up using multigene next generation sequencing (NGS) methods. SoR: GPS
- Patients with congenital neutropenia may have positive anti-neutrophil antibodies. In case of severe infections patients should in spite positive antibodies undergo genetic testing. SoR: GPS
- Family history or clinical findings may suggest another specific neutropenia-associated gene to be sequenced (e.g., TAZ, G6PC3, SBDS). SoR: GPS
- A targeted NGS panel including all genes known to be mutated in CN (>30) is a reasonable first step that provides uniform sequencing coverage for all genes of interest and requires simpler bioinformatics analysis. SoR: GPS
- The choice of genes within the panel should include not only all those that strictly cause neutropenia when mutated but also genes resulting in diseases in which neutropenia is a secondary feature; whole exome sequencing (WES) can also be used in cases where no mutations were detected in a panel. SoR: GPS
- A.5.d. Diagnosing cyclic neutropenia
- Complete blood counts (CBC) in family members, serial blood counts twice or thrice a week over a period of 6 weeks to exclude CyN. SoR: GPS
- A.5.e. Diagnosing autoimmune neutropenia
- Antineutrophil antibody testing should be performed as first-line investigation in both children and adults. SoR: GPS
- Indirect granulocyte immunofluorescence test (GIFT) is recommended as a first-line assay in reference laboratories. SoR: GPS
- A positive GIFT in combination with laboratory tests and clinical picture can support diagnosis of autoimmune neutropenia (AIN) but does not exclude the diagnosis of other types of neutropenia. SoR: GPS
- With a negative indirect GIFT, if the clinical suspicion of AIN remains high, GIFT should be repeated several times. SoR: GPS
- A.5.f. Family screening in genetically proven congenital neutropenia
- Following identification of the responsible gene(s), Sanger sequencing is also recommended for mutation screening of the members of affected families. SoR: GPS
- Multigene NGS or WES ideally should include patient and parental DNA (trio analysis). SoR: GPS
- A.5.g. Role of bone marrow examination
- Diagnostic BM with morphology, cytogenetics, and NGS of genes related to myeloid malignancies should be performed: In pediatric patients with severe and moderate chronic neutropenia with the exception of patients with primary AIN with positive anti-granulocyte antibodies and drug-induced neutropenia. SoR: GPS
- Patients with congenital neutropenia may have positive antibody test. SoR: GPS
- In patients with suggested AIN but negative granulocyte antibody test, if patients suffer from recurrent infections. SoR: GPS
- In any patients before G-CSF treatment. SoR: GPS
- Repeated BM follow-up should be performed in patients with decreasing ANC or additional changes in other blood cell counts (e.g., anemia and thrombocytopenia) or erythrocyte indices. SoR: GPS
- A.5.h. Role of flow cytometry
- FC is an important tool in the diagnosis of neutropenia associated with PID syndromes such as ALPS, CVID, and HIGM syndrome. SoR: GPS
- Assessment of a PNH clone by FC testing is also recommended. SoR: GPS
- Flow FISH is recommended when a telomere biology disorder is suspected. SoR: GPS
- A.5.i. Role of genetic testing
- Genetic diagnosis is important to confirm the diagnosis of CN, estimate the risk for MDS/AML, support stem cell donor selection for patients, and family counseling. SoR: GPS
- When the clinical picture, inheritance, or bone marrow features (i.e., block at the promyelocyte stage) are indicative of a specific gene mutation, single-gene analysis by Sanger sequencing technique could be applied. SoR: GPS
- For CN where the clinical picture does not suggest a specific genetic cause, we recommend the use of NGS techniques such as multigene panels or targeted WES. SoR: GPS
- For patients for whom a genetic cause is not identified by the above methods, WGS and RNA-sequencing may be powerful diagnostic tools. SoR: GPS
- NGS analysis of bone marrow or peripheral blood for acquired somatic variants is recommended for patients with chronic neutropenia. SoR: GPS
- Screening for known mutations is recommended in family members. SoR: GPS
- It is important to validate germline mutations mainly in fibroblasts or hair follicles keratinocytes (cells from buccal swab are less indicated for possible blood contamination), in the presence of leukemic blasts in PB. SoR: GPS
- A.5.j. Initial evaluation for children presenting with fever and neutropenia (ASCO 2023)
- Adopt a validated risk stratification strategy and incorporate it into routine clinical management. SoR: Strong
- Obtain blood cultures at the onset of FN from all lumens of central venous catheters. SoR: Strong
- Consider obtaining peripheral blood cultures concurrent with central venous catheter cultures. SoR: Conditional
- Consider urinalysis and urine culture in patients where a clean-catch, mid-stream specimen is readily available. SoR: Conditional
- Obtain chest radiography only in patients with respiratory signs or symptoms. SoR: Strong
- Treatment of Neutropenia
- B.1. Treatment of infection in patients with fever and non-severe neutropenia
- Individuals with an ANC >1.0 × 10⁹/L can be managed as per those with normal ANC. SoR: GPS
- B.2. Treatment of fever and neutropenia (FN)
- Infants with primary AIN should be excluded from these recommendations, since ANC is not indicative of infectious risk in these patients. SoR: GPS
- Low-risk FN
- Consider initial or step-down outpatient management if the infrastructure is in place to ensure careful monitoring and follow-up. SoR: Conditional
- Consider oral antibacterial therapy administration if the patient is able to tolerate this route of administration reliably. SoR: Conditional
- High-risk FN
- Use monotherapy with an antipseudomonal β-lactam, a fourth-generation cephalosporin or a carbapenem as empiric antibacterial therapy in pediatric high-risk FN. SoR: Strong
- Reserve addition of a second anti-Gram-negative agent or a glycopeptide for patients who are clinically unstable, when a resistant infection is suspected, or for centers with a high rate of resistant pathogens. SoR: Strong
- Ongoing management – Modification of treatment
- In patients who are responding to initial empiric antibacterial therapy, discontinue double coverage for Gram-negative infection or empiric glycopeptide (if initiated) after 24–72 hours if there is no specific microbiologic indication to continue combination therapy. SoR: Strong
- Do not broaden the initial empiric antibacterial regimen based solely on persistent fever in patients who are clinically stable. SoR: Strong
- In patients with persistent fever who become clinically unstable, escalate the initial empiric antibacterial regimen to include coverage for resistant Gram-negative, Gram-positive and anaerobic bacteria. SoR: Strong
- Ongoing management – Cessation of treatment
- In both high-risk and low-risk FN patients who have been clinically well and afebrile for at least 24 hours, discontinue empiric antibacterial therapy if blood cultures remain negative at 48 hours if there is evidence of marrow recovery. SoR: Strong
- In patients with low-risk FN who have been clinically well and afebrile for at least 24 hours, consider discontinuation of empiric antibacterial therapy if blood cultures remain negative at 48 hours despite no evidence of marrow recovery. SoR: Conditional
- B.3. Treatment of severe chronic neutropenia
- Use of G-CSF
- Patients usually need life-time treatment. SoR: GPS
- The therapeutic target (ANC ≥1.0 × 10⁹/L and ≤5.0 × 10⁹/L) is considered achievable with a G-CSF starting dose of 5 μg/kg/d, but the individual variability of the response may require modifications of the subsequent doses. SoR: GPS
- It is suggested to increase G-CSF over 2 weeks up to doubling initial dose if ANC remain <1.0 × 10⁹/L and to reduce G-CSF if ANC reach >5.0 × 10⁹/L. SoR: GPS
- Maintain the initial dose if the target ANC (≥1.0 × 10⁹/L and ≤5.0 × 10⁹/L) is achieved and increase by 2.5 μg/kg/day every 5–7 days if ANC remains <1.0 × 10⁹/L. N.B: Depending on clinical situation and known underlying genotype doubling of G-CSF may be indicated up to 50 µg/kg/day to evaluate G-CSF response. SoR: GPS
- Experts agreed on the non-superiority of lenograstim vs. filgrastim as the number of infections was similar in patients treated with the two preparations. SoR: GPS
- The use of pegylated G-CSF in neutropenia has been rarely reported in pediatric age. SoR: GPS
- Hematopoietic stem cell transplantation (HSCT)
- Strong indications for HSCT include: (1) Established transformation to MDS/Acute Leukemia or bone marrow dysplastic features with high-risk acquired cytogenetic abnormalities (monosomy 7, trisomy 8, trisomy 21) or with a combination of acquired leukemia-associated somatic mutations (e.g., RUNX1, ASXL1, SETBP1). CSF3R mutations alone are not an indication of HSCT. (2) CN due to mutations carrying an intrinsic high risk of leukemic transformation per se, i.e., GATA2 mutations, high-risk ELANE mutations, or clones with biallelic TP53 mutations in SDS. (3) No response to G-CSF (doses >20 mcg/kg/d to reach ANC of 1.0 × 10⁹/L), poor response to G-CSF (doses between 10 and 20 mcg/kg/d failing to reach ANC of 1.0 × 10⁹/L) or poor control of infection irrespective of the G-CSF dose. Potential indications: adequate management of infections with G-CSF at “intermediate doses” (10–15 mcg/kg/d) with availability of a healthy HLA-identical sibling or HLA identical matched donor. Weak indication: G-CSF response at doses up to 10 μg/kg/d, good tolerability and compliance to daily subcutaneous injections, infections control, and unavailability of HLA matched donors. SoR: GPS
- B.4. Treatment of cyclic neutropenia (CyN)
- In cyclic neutropenia (CyN) G-CSF treatment has been shown to shorten the degree and the duration of neutropenia conferring the patients a better quality of life. For these reasons, the long-term pattern of treatment was considered adequate to CyN. SoR: GPS
- This has to be intended as a life-time treatment not to be performed daily but only during the lowest levels of neutrophils in cycling periods of neutropenia. SoR: GPS
- Usually, patients affected by CyN need lower G-CSF doses (median 2.4 μg/kg/day) than SCN patients. A starting dose of 1–3 μg/kg/d should maintain ANC around 1.5 × 10⁹/L; for lower counts the dosage may be increased up to two-fold within 2–4 weeks. Given the ANC fluctuations it is suggested to calculate the final G-CSF dose after a number of observations. SoR: GPS
- Daily administration during neutropenia is more appropriate than the intermittent schedule. Initial suggested dose during neutropenic periods is 2 μg/kg/day. If ANC target is not achieved, increases of 2 μg/kg/d every neutropenic phase are recommended. SoR: GPS
- The panel recommends the use of G-CSF in severe CN and in CyN, particularly in those patients with recurrent or severe infections. The panel considers ANC over 1.0 × 10⁹/L as the protective threshold against infections. For CyN, a lower nadir may be accepted. In CyN, G-CSF doses may be lower than in severe CN. The standard dose should be ≤3 mcg/kg/d continuously. G-CSF may be also given every other day. Dosage may be adjusted to avoid nadir <0.5 × 10⁹/L and clinical conditions such as mouth ulcers, fevers, or infections. SoR: GPS
- B.5. Treatment of idiopathic neutropenia (IN)
- In the majority of cases, IN requires an on-demand treatment only during infections or surgery. In these patients an initial dose of 1–2 μg/kg/d is considered sufficient to achieve goal ANC. SoR: GPS
- Some patients with a diagnosis of IN may have severe and/or recurrent infections and may require long-term treatment. A starting dose (1–2 μg/kg/die) of G-CSF can be sufficient to achieve the goal ANC and a rhythm of 2–3 times/week may ensure protective values of neutrophils. SoR: GPS
- B.6. Treatment of autoimmune neutropenia (AIN)
- G-CSF
- The use of G-CSF in AIN is generally necessary only in case of severe infections and therefore the on-demand treatment is considered appropriate. Some forms of AIN may have severe and/or recurrent infections requiring a long-term treatment. SoR: GPS
- An initial low dose of G-CSF (1–2 mg/kg/die) to be subsequently adjusted. SoR: GPS
- Other treatment modalities
- Various immune-regulating drugs (e.g., cyclosporine, methotrexate, low-dose cyclophosphamide) have been used in refractory cases, but overall efficacy has not been determined. SoR GPS
- G-CSF
- The use of G-CSF in AIN is generally necessary only in case of severe infections and therefore the on-demand treatment is considered appropriate. Some forms of AIN may have severe and/or recurrent infections requiring a long-term treatment. SoR: GPS
- An initial low dose of G-CSF (1–2 mg/kg/die) to be subsequently adjusted. SoR: GPS
- Other treatment modalities
- Various immune-regulating drugs (e.g., cyclosporine, methotrexate, low-dose cyclophosphamide) have been used in refractory cases, but overall efficacy has not been determined. SoR: GPS
- In case of an underlying immune dysregulation/deficiency, particularly in childhood, more targeted treatments (e.g., mycophenolate mofetil and rapamycin) may be used. SoR: GPS
- The role of rituximab (and other antibodies to CD20) is well established as effective treatment for many autoimmune disorders. However, its role as treatment for AIN is unclear. SoR: GPS
- Pediatric patients actively receiving cytotoxic chemotherapy. Down Syndrome. Children with immune deficiency. Patient with Central venous catheter. Compromise of mucosal barriers (e.g. mucositis, typhlitis). Myelosuppression. Children with hematologic malignancies have an increased risk relative to children with solid tumors (SoR: GPS)
- Personal protective equipment (gown, gloves. . .) in patients with contagious infection or colonization by multidrug resistant (MDR) microorganisms (SoR: GPS) Aseptic technique in central venous catheter (CVC) handling (SoR: GPS) Hand, surface and food preparation hygiene measures (SoR: GPS) Chlorhexidine gluconate (CHG) baths (CHB) involve cleansing the patient’s body each day with CHG‑impregnated wipes. Literature from pediatric patients with temporary CVCs inserted during critical care admissions suggests that CHB may have a role in reducing the rate of CLABSIs in these patients (SoR: GPS)
- Avoid contact with pets associated with a high risk: turtles, cat’s litter, stables and new pets. This does not apply to other pets that are correctly vaccinated, in regard to which the sole precaution is to avoid contact with their faeces (SoR: GPS) Prevention of IFI (high risk patients/situations) Avoid flowers and plants in hospital rooms and the bedroom of the patient. Hospital rooms with HEPA filters and positive pressure (>12 room air changes/h). Use of FPP2 masks in closed spaces without HEPA filters (except the usual place of residence, where ventilation, avoiding plants, moisture and construction work). Recommendation not supported by evidence: Avoid pools, especially public or indoor ones. Low bacteria diet not proven to reduce the incidence or severity of infection. Personal protective equipment for patients in absence of microbial isolation that justifies it (SoR: GPS)
- G‑CSF in patients with solid tumors receiving intensive chemotherapy, who need to maintain cytotoxic dose/chemotherapy intensity and/or with previous history of fever neutropenia (FN) (SoR: GPS) Annual vaccination against seasonal flu of patients and their household contacts and health care workers (SoR: GPS)
- AML and relapsed ALL: Consider systemic antibacterial prophylaxis administration in children receiving intensive chemotherapy expected to result in severe neutropenia (absolute neutrophil count <500/µl for at least 7 days) (SoR: Conditional) Newly diagnosed ALL: Do not use systemic antibacterial prophylaxis routinely for children receiving induction chemotherapy (SoR: Conditional)
- Therapy not expected to result in severe neutropenia (ANC <500/µl for at least 7 days): Do not use systemic antibacterial prophylaxis (SoR: Strong) Autologous HSCT: Do not use systemic antibacterial prophylaxis routinely (SoR: Conditional) Allogeneic HSCT: Do not use systemic antibacterial prophylaxis routinely (SoR: Conditional)
- Levofloxacin is the preferred agent if systemic antibacterial prophylaxis is planned (SoR: Strong) If systemic antibacterial prophylaxis is planned; we suggest that administration be restricted to the expected period of severe neutropenia (absolute neutrophil count <500/μL) (SoR: Conditional)
- AML: Administer systemic antifungal prophylaxis to children and adolescents receiving treatment of AML that is expected to result in profound and prolonged neutropenia (SoR: Strong)
- ALL (newly diagnosed and relapsed): Consider administering systemic antifungal prophylaxis to children and adolescents at high risk for invasive fungal disease (IFD) (SoR: Conditional)
- ALL (low risk): Do not routinely administer systemic antifungal prophylaxis (SoR: Strong) Other malignancies (lymphomas and solid tumors): Do not routinely administer systemic antifungal prophylaxis to children and adolescents with cancer at low risk for IFD, such as most pediatric patients with lymphomas and solid tumors (SoR: Strong)
- Allogeneic HSCT: Administer systemic antifungal prophylaxis pre‑engraftment and to those receiving systemic immunosuppression for GVHD (SoR: Strong)
- Autologous HSCT: We suggest that systemic antifungal prophylaxis not be used routinely (SoR: Strong) Mold‑active agent: In choosing a mold‑active agent, administer an echinocandin or a mold‑active azole (SoR: Strong) Amphotericin: Do not use amphotericin routinely as systemic antifungal prophylaxis (SoR: Strong) If systemic antifungal prophylaxis is warranted, consider administration during periods of observed or expected severe neutropenia. For allogeneic HSCT recipients, consider administration during systemic immunosuppression for GVHD treatment (SoR: Conditional)
- Do not use probiotics routinely for the prevention of CDI in children and adolescents with cancer and pediatric HSCT patients (SoR: Conditional) Use either oral metronidazole or oral vancomycin for the treatment of non‑severe CDI in children and adolescents with cancer and pediatric HSCT patients (SoR: Strong) Use oral vancomycin for the treatment of severe CDI in children and adolescents with cancer and pediatric HSCT patients (SoR: Strong) Do not use monoclonal antibodies routinely for the treatment of CDI in children and adolescents with cancer and pediatric HSCT patients (SoR: Strong)
- Risk for PJP pneumonia is highest in patients with leukemia and lymphoma, prophylaxis is also recommended for children with solid tumors undergoing chemotherapy that is likely to cause lymphopenia. Data in patients with HIV shows that the risk of PJP is highest in those with CD4 counts under 200 x106/L, and one study from the solid organ transplantation population found that odds of PJP infection were significantly increased when absolute lymphocyte count (ALC) was < 500 x106/L. Sulfamethoxazole (TMP‑SMX) is highly effective. For patients who do not tolerate TMP‑SMX, pentamidine, dapsone, and atovaquone are alternative options, Optimal duration of PJP prophylaxis is unknown. One guideline suggests continuing until 3 months after the end of therapy and normalization of lymphocyte count (SoR: GPS)
- Use cryotherapy for older, cooperative pediatric patients receiving treatment for cancer or undergoing HSCT who will receive short infusions of melphalan or 5‑fluorouracil (SoR: GPS) Consider using cryotherapy for older, cooperative pediatric patients receiving treatment for cancer or undergoing HSCT who will receive short infusions of chemotherapy associated with mucositis other than melphalan or 5‑fluorouracil (SoR: GPS) Do not administer palifermin routinely to pediatric patients with cancer receiving treatment for cancer or undergoing HSCT (SoR: GPS) Use intraoral photo‑biomodulation therapy in the red‑light spectrum (620–750 nm) for pediatric patients undergoing autologous or allogeneic HSCT and for pediatric patients who will receive radiotherapy for head and neck carcinoma (SoR: GPS) Consider using intraoral photo‑biomodulation therapy in the red‑light spectrum (620–750 nm) for pediatric patients who will receive radiotherapy for head and neck cancers other than carcinoma (SoR: GPS) Do not administer GCSFs to pediatric patients receiving treatment for cancer or undergoing HSCT for the purpose of mucositis prevention (SoR: GPS)
- The use of G‑CSF in in patients with solid tumors receiving intensive chemotherapy, who need to maintain cytotoxic dose/chemotherapy intensity and/or with previous history of FN reduces the duration of neutropenia (SoR: GPS)
- Annual vaccination against seasonal flu of patients and their household contacts and health care workers. Despite potential lack of efficacy in certain populations, the American Academy of Pediatrics and Center for Disease Control still recommend yearly influenza vacations in pediatric oncology patients based on potential benefit with low risk of reaction (SoR: GPS)
- Careful clinical examination of skin and mucous membranes, upper and lower respiratory tract and abdomen to exclude underlying infection, lymphadenopathy, and/or hepatosplenomegaly (SoR: GPS) Vital signs: temperature, heart rate, respiratory rate, blood pressure and oxygen saturation. Pediatric assessment triangle (early detection of sepsis). Review of systems, including neurologic assessment, perfusion, skin and mucosae (oral and perianal, avoid rectal palpation), any area with pain, scars, devices (SoR: GPS)
- In chronic neutropenia patients, we recommend performing CBC with differential WBC counts and morphological evaluation every 3–4 months (SoR: GPS) When approaching adulthood, CN patients should be transferred to a dedicated hematology specialist (SoR: GPS)
- Annual BM and cytogenetics follow‑up should be performed in patients with congenital BM failure syndromes independent of ANC and treatment with G‑CSF. undefined SCN (after extensive investigation) with G‑CSF treatment, may be considered (SoR: GPS) Repeated BM follow‑up should be performed in patients with decreasing ANC or additional changes in other blood cell counts (e.g., anemia and thrombocytopenia) or erythrocyte indices (SoR: GPS)
- Pediatric patients actively receiving cytotoxic chemotherapy. Down Syndrome. Children with immune deficiency. Patient with Central venous catheter. Compromise of mucosal barriers (e.g. mucositis, typhlitis). Myelosuppression. Children with hematologic malignancies have an increased risk relative to children with solid tumors (SoR: GPS)
- Personal protective equipment (gown, gloves. . .) in patients with contagious infection or colonization by multidrug resistant (MDR) microorganisms (SoR: GPS) Aseptic technique in central venous catheter (CVC) handling (SoR: GPS) Hand, surface and food preparation hygiene measures (SoR: GPS) Chlorhexidine gluconate (CHG) baths (CHB) involve cleansing the patient’s body each day with CHG‑impregnated wipes. Literature from pediatric patients with temporary CVCs inserted during critical care admissions suggests that CHB may have a role in reducing the rate of CLABSIs in these patients (SoR: GPS) Avoid contact with pets associated with a high risk: turtles, cat’s litter, stables and new pets. This does not apply to other pets that are correctly vaccinated, in regard to which the sole precaution is to avoid contact with their faeces (SoR: GPS) Prevention of IFI (high risk patients/situations) Avoid flowers and plants in hospital rooms and the bedroom of the patient. Hospital rooms with HEPA filters and positive pressure (>12 room air changes/h). Use of FPP2 masks in closed spaces without HEPA filters (except the usual place of residence, where ventilation, avoiding plants, moisture and construction work). Recommendation not supported by evidence: Avoid pools, especially public or indoor ones. Low bacteria diet not proven to reduce the incidence or severity of infection. Personal protective equipment for patients in absence of microbial isolation that justifies it (SoR: GPS)
- G‑CSF in patients with solid tumors receiving intensive chemotherapy, who need to maintain cytotoxic dose/chemotherapy intensity and/or with previous history of fever neutropenia (FN) (SoR: GPS) Annual vaccination against seasonal flu of patients and their household contacts and health care workers (SoR: GPS)
- AML and relapsed ALL: Consider systemic antibacterial prophylaxis administration in children receiving intensive chemotherapy expected to result in severe neutropenia (absolute neutrophil count <500/µl for at least 7 days) (SoR: Conditional) Newly diagnosed ALL: Do not use systemic antibacterial prophylaxis routinely for children receiving induction chemotherapy (SoR: Conditional) Therapy not expected to result in severe neutropenia (ANC <500/µl for at least 7 days): Do not use systemic antibacterial prophylaxis (SoR: Strong) Autologous HSCT: Do not use systemic antibacterial prophylaxis routinely (SoR: Conditional) Allogeneic HSCT: Do not use systemic antibacterial prophylaxis routinely (SoR: Conditional)
- Levofloxacin is the preferred agent if systemic antibacterial prophylaxis is planned (SoR: Strong)
- If systemic antibacterial prophylaxis is planned; we suggest that administration be restricted to the expected period of severe neutropenia (absolute neutrophil count <500/μL) (SoR: Conditional)
- AML: Administer systemic antifungal prophylaxis to children and adolescents receiving treatment of AML that is expected to result in profound and prolonged neutropenia (SoR: Strong) ALL (newly diagnosed and relapsed): Consider administering systemic antifungal prophylaxis to children and adolescents at high risk for invasive fungal disease (IFD) (SoR: Conditional) ALL (low risk): Do not routinely administer systemic antifungal prophylaxis (SoR: Strong) Other malignancies (lymphomas and solid tumors): Do not routinely administer systemic antifungal prophylaxis to children and adolescents with cancer at low risk for IFD, such as most pediatric patients with lymphomas and solid tumors (SoR: Strong) Allogeneic HSCT: Administer systemic antifungal prophylaxis pre‑engraftment and to those receiving systemic immunosuppression for GVHD (SoR: Strong)
- Autologous HSCT: We suggest that systemic antifungal prophylaxis not be used routinely (SoR: Strong)
- Mold‑active agent: In choosing a mold‑active agent, administer an echinocandin or a mold‑active azole (SoR: Strong)
- Amphotericin: Do not use amphotericin routinely as systemic antifungal prophylaxis (SoR: Strong) If systemic antifungal prophylaxis is warranted, consider administration during periods of observed or expected severe neutropenia. For allogeneic HSCT recipients, consider administration during systemic immunosuppression for GVHD treatment (SoR: Conditional)
- Do not use probiotics routinely for the prevention of CDI in children and adolescents with cancer and pediatric HSCT patients (SoR: Conditional)
- Use either oral metronidazole or oral vancomycin for the treatment of non‑severe CDI in children and adolescents with cancer and pediatric HSCT patients (SoR: Strong)
- Use oral vancomycin for the treatment of severe CDI in children and adolescents with cancer and pediatric HSCT patients (SoR: Strong) Do not use monoclonal antibodies routinely for the treatment of CDI in children and adolescents with cancer and pediatric HSCT patients (SoR: Strong)
- Risk for PJP pneumonia is highest in patients with leukemia and lymphoma, prophylaxis is also recommended for children with solid tumors undergoing chemotherapy that is likely to cause lymphopenia. Data in patients with HIV shows that the risk of PJP is highest in those with CD4 counts under 200 x106/L, and one study from the solid organ transplantation population found that odds of PJP infection were significantly increased when absolute lymphocyte count (ALC) was < 500 x106/L. Sulfamethoxazole (TMP‑SMX) is highly effective. For patients who do not tolerate TMP‑SMX, pentamidine, dapsone, and atovaquone are alternative options, Optimal duration of PJP prophylaxis is unknown. One guideline suggests continuing until 3 months after the end of therapy and normalization of lymphocyte count (SoR: GPS)
- Use cryotherapy for older, cooperative pediatric patients receiving treatment for cancer or undergoing HSCT who will receive short infusions of melphalan or 5‑fluorouracil (SoR: GPS)
- Consider using cryotherapy for older, cooperative pediatric patients receiving treatment for cancer or undergoing HSCT who will receive short infusions of chemotherapy associated with mucositis other than melphalan or 5‑fluorouracil (SoR: GPS)
- Do not administer palifermin routinely to pediatric patients with cancer receiving treatment for cancer or undergoing HSCT (SoR: GPS)
- Use intraoral photo‑biomodulation therapy in the red‑light spectrum (620–750 nm) for pediatric patients undergoing autologous or allogeneic HSCT and for pediatric patients who will receive radiotherapy for head and neck carcinoma (SoR: GPS)
- Consider using intraoral photo‑biomodulation therapy in the red‑light spectrum (620–750 nm) for pediatric patients who will receive radiotherapy for head and neck cancers other than carcinoma (SoR: GPS)
- Do not administer GCSFs to pediatric patients receiving treatment for cancer or undergoing HSCT for the purpose of mucositis prevention (SoR: GPS)
- The use of G‑CSF in in patients with solid tumors receiving intensive chemotherapy, who need to maintain cytotoxic dose/chemotherapy intensity and/or with previous history of FN reduces the duration of neutropenia (SoR: GPS)
- Annual vaccination against seasonal flu of patients and their household contacts and health care workers. Despite potential lack of efficacy in certain populations, the American Academy of Pediatrics and Center for Disease Control still recommend yearly influenza vacations in pediatric oncology patients based on potential benefit with low risk of reaction (SoR: GPS)
- Careful clinical examination of skin and mucous membranes, upper and lower respiratory tract and abdomen to exclude underlying infection, lymphadenopathy, and/or hepatosplenomegaly (SoR: GPS)
- Vital signs: temperature, heart rate, respiratory rate, blood pressure and oxygen saturation. Pediatric assessment triangle (early detection of sepsis). Review of systems, including neurologic assessment, perfusion, skin and mucosae (oral and perianal, avoid rectal palpation), any area with pain, scars, devices (SoR: GPS)
- In chronic neutropenia patients, we recommend performing CBC with differential WBC counts and morphological evaluation every 3–4 months (SoR: GPS) When approaching adulthood, CN patients should be transferred to a dedicated hematology specialist (SoR: GPS)
- Annual BM and cytogenetics follow‑up should be performed in patients with congenital BM failure syndromes independent of ANC and treatment with G‑CSF. undefined SCN (after extensive investigation) with G‑CSF treatment, may be considered (SoR: GPS) Repeated BM follow‑up should be performed in patients with decreasing ANC or additional changes in other blood cell counts (e.g., anemia and thrombocytopenia) or erythrocyte indices (SoR: GPS)
- The key markers of malignant transformation to MDS or leukemia in CN patients are the following: typical dysplastic features in PB (pseudo Pelger‑Huet anomaly, hypogranulation, hyper segmentation, reticulated nucleus, and ringed‑shaped nuclei) and BM (defective granulation, maturation arrest at myelocyte stage, and increase in monocytoid forms); cytogenetic abnormalities (e.g., CSF3R, RUNX1, and ASXL1); and high frequency of somatic mutations in leukemia‑associated driver genes. The most common chromosomal defects in patients with CN at the MDS stage are trisomy 21 and monosomy 7 (SoR: GPS)
➡️Guideline Registration
PREPARE (Practice guideline REgistration for transPAREncy), WHO Collaborating Center for Guideline Implementation and Knowledge Translation, EBM Center, University of Lanzhou, Lanzhou, China. Registration Number: PREPARE-2024CN145. Link: http://www.guidelines-registry.org/
- Recommendations
|
Table 3. Recommendations |
|
|
|
|
A. Diagnosis of neutropenia |
|
|
|
|
Source Guideline |
Recommendations |
QoE |
SoR |
|
Definition of neutropenia |
|||
|
A.1. We recommend the use of the following definition of neutropenia in neonates, infants and children |
|||
|
EHA-EuNet-INNOCHRON 2023 |
The definition of neutropenia varies according to the patient’s ethnic origin and age. Neonates: • At term neonates Neutropenia is defined as an absolute neutrophil count (ANC) level of 2.5 x10*9/L for term/near-term neonates 72–240 hours following delivery. • In preterm newborns Neutropenia is defined as an ANC level of 1.0 x10*9/L for preterm neonates. Infants and Children • The widely accepted cutoff level of ANC for the definition of neutropenia in Caucasian newborns and infants up to the age of 1 year is 1.0 x10*9/L. • From the age of 1 year to adulthood the cutoff level for neutropenia is 1.5 x 10*9/L. |
|
GPS |
|
Classification of neutropenia |
|||
|
A.2.a. We recommend the use of the following classification of neutropenia according to severity |
|||
|
EHA-EuNet-INNOCHRON 2023 |
• The term agranulocytosis is used for severe neutropenia with ANC <0.2 x 10*9/L. |
|
GPS |
|
A.2.b. We recommend the use of the following classification of neutropenia according to duration |
|||
|
EHA-EuNet-INNOCHRON 2023 |
|
GPS |
|
|
A.2.c. We recommend the use of the following classification of neutropenia according to etiology? |
|||
|
EHA-EuNet-INNOCHRON 2023 |
|
|
GPS |
|
A.2.d. We recommend the use of the following congenital neutropenia definition and its classification? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
• CN can be further subclassified into disorders where neutropenia is the only abnormality and those where neutropenia is associated with extra-hematological manifestations, immunodeficiency/immune dysregulation, metabolic disorders and nutritional deficiencies, or as part of more complex BM failure syndromes. • The classification also takes into consideration the genes that have been identified as responsible for each CN subtype. • Individuals of African and Middle Eastern descent display normal ANCs in the range from 0.5 to 1.5 x10*9/L and less frequently even lower. • This variation, previously termed ethnic neutropenia, is usually inherited as an autosomal recessive trait associated with a polymorphism (rs2814778, −46T>C) in the GATA box in the promoter region of the atypical chemokine receptor-1 (ACKR1) gene, also known as the duffy antigen receptor for chemokines (DARC). • In homozygosity (C/C), the polymorphism results in the absence of Duffy antigen expression specifically on red blood cells, a phenotype known as Duffy-null. • The guideline working group suggests the introduction of the term ACKR1/DARC-associated neutropenia (ADAN), instead of ethnic neutropenia, to emphasize the genetic rather than the ethnic basis of this entity. |
|
GPS |
|
Causes of neutropenia |
|||
|
A.3.a. We recommend the use of the following causes of neutropenia in the neonatal period? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
· females have ANC counts on average 2.0 x 10*9/L higher than males. · ANC in capillary blood is on average 1.5–2.0 x10*9/L higher than in cord blood. · ANC is on average higher at altitude than at sea level. · Severe necrotizing enterocolitis in the newborn, especially if preterm, is frequently associated with transiently low ANCs.
Examples of variables related to pregnancy or delivery include the following: · maternal tobacco smoking is associated with lower ANC. · maternal chemotherapy results in neutropenia · maternal antiretroviral therapy results in neutropenia. · maternal hypertension during pregnancy results in neutropenia. · prenatal growth retardation is an independent risk factor for neutropenia. · Rh-hemolytic disease of the newborn is associated with neutropenia in about 50% of newborns whether or not combined with severe anemia and thrombocytopenia. · twin-twin transfusion syndrome is a rare condition with neutropenia always present in the donor twin (the anemic one). · neutropenia is present in 67% of infants with asphyxia.
|
|
GPS |
|
A.3.b. What are the types of immune neutropenia specific to neonates? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
Neonatal immune neutropenia can be subclassified as the following: 1. AIN, which is unusual, but not impossible, at <1 month of Age. 2. Neonatal alloimmune neutropenia (NAN) in which a genetic mismatch for a polymorphism in one of the genes encoding human neutrophil antigens (HNA) between mother and fetus leads to immunization of the pregnant woman, passage of alloantibodies over the placenta and neutropenia in the baby. Indirect antineutrophil antibodies are positive in the mother and in the newborn. The diagnostic confirmation may be obtained through a positive crossmatch between maternal sera and paternal granulocytes (even if not routinely indicated). 3. NAN secondary to maternal AIN is the rarest immune neutropenia of early infancy. The duration of this neutropenia is on an average the same as the classic NAN.
|
|
GPS |
|
A.3.c. What are the following causes of acquired neutropenia to be considered? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
|
GPS |
|
|
Clinical diagnosis of neutropenia |
|||
|
A.4.a. What are the required points in history taking for patients presenting with neutropenia? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
• Patient history should include inquiry about occurrence of infections and their frequency, type, severity, and need for hospitalization. Specifically, history of omphalitis, gingivitis, periodontitis, skin infections, abscesses, and pneumonias as well as duration and response to antibiotics should be also investigated. • Presence of congenital malformations in the patient or family is important. • For adult patients, drug history is important, as well as work-up for autoimmune and other disorders that may be associated with neutropenia. • Detailed family history should include ethnic origin, consanguinity, occurrence of recurrent infections, and neutropenia in other family members, as well as unexplained infant death or miscarriages. • A detailed history should be taken of any symptoms denoting underlying autoimmune or other diseases that may result in secondary neutropenia. • History of chronic viral infections such as viral hepatitis or HIV should also be obtained. • Careful inquiry should be made concerning drug administration, including over the counter drugs, substances often denominated as natural supplements, and recreational drugs; neutropenia can be linked not only to drugs that the patient has recently started but also to drugs that have recently been discontinued. |
|
GPS |
|
A.4.b. What is the required detailed clinical examination in patients presenting with neutropenia? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
• Careful clinical examination of skin and mucous membranes, upper and lower respiratory tract and abdomen to exclude underlying infection, lymphadenopathy, and/or hepatosplenomegaly. • Clinicians should be aware that neutropenic patients might have only subtle symptoms of infection due to reduced inflammatory response. • In children and adults, clinical examination is crucial to detect congenital disorders. It should focus on growth, evidence of cognitive impairment, developmental delay, dysmorphism (mainly skeletal), nail, hair or skin abnormalities, signs of bronchiectasis due to recurrent chest infections, hepatomegaly or splenomegaly, organ malformation, evidence of superficial veins, and finally signs of photophobia, nystagmus, oculocutaneous albinism, and neuropathy. The absence of obvious clinical signs does not exclude the presence of a congenital disorder. • Cardiac function, presence of enlarged lymph nodes, joint symptoms, and symptoms compatible with autoimmune, metabolic, gastrointestinal, or nutritional diseases should also be considered.
|
|
GPS |
|
A.4.Cc What is the diagnostic clinical criteria for children presenting with fever and neutropenia? |
|||
|
ASCO 2023 |
Febrile neutropenia (FN) is defined as an oral temperature of >38.3°C or two consecutive readings of >38.0°C for 2 h and an absolute neutrophil count (ANC) of <0.5 × 109/l or expected to fall below 0.5 × 109/l.. |
|
GPS |
|
Laboratory work up for diagnosis |
|||
|
A.5.a. What is the approach to investigate children with neutropenia?? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
• Patients with acute neutropenia, particularly in the presence of symptoms/signs of infection, may require immediate investigation and even hospitalization depending on the severity of neutropenia and symptoms. • For patients with chronic, isolated neutropenia without a phenotype suggestive of any underlying CN syndrome, a flowchart of basic investigation is recommended. • If the initial evaluation does not suggest ADAN, nor postinfectious or drug-induced neutropenia, the first level of investigation, possibly adjusted to the availability of the recommended tests |
|
GPS |
|
A.5.b. What are the first and second lines of investigations for children with neutropenia? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
First-line investigations • Complete blood count (CBC)s, peripheral blood (PB) smear. • Biochemistry tests including liver and kidney function, c-reactive protein (CRP). • Vitamin B12 and folate. • Virology antibody screening (i.e., HepB, HepC, HIV, EBV, and CMV). • Thyroid hormones (FT3, FT4, TSH), antithyroid antibodies (anti-TG and anti-TPO). • Antineutrophil antibodies by granulocyte immunofluorescence test (GIFT) and/or granulocyte agglutination test (GAT). • Immunoglobulin levels and flow cytometric analysis of PB lymphocyte subsets, • Additional investigation in children: flow cytometric analysis of CD3+TCRα/β+CD4-CD8- (double negative) PB T lymphocytes. Second-line investigations • CBCs in family members, serial blood counts twice a week over a period of 6 weeks to exclude CyN, copper; ceruloplasmin, anti-tissue transglutaminase IgA, deamidated gliadin peptide antibodies IgA/IgG and pancreatic isoamylase. • Additional investigation in children: rheumatoid factor (RF), antinuclear antibody (ANA), extractable nuclear antigen (ENA), and double stranded DNA (ds-DNA). • In children, young adults, and considered for adults: genetic investigations. |
|
GPS |
|
A.5.c How to confirm the diagnosis of congenital neutropenia? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
• Following negative results of first-level investigation, all patients with SCN and recurrent infections and/ or family history of severe neutropenia and typical anomalies should undergo genetic work-up using multigene next generation sequencing (NGS) methods. • Patients with congenital neutropenia may have positive anti-neutrophil antibodies. In case of severe infections patients should in spite positive antibodies undergo genetic testing |
|
GPS |
|
• However, family history or clinical findings may suggest another specific neutropenia-associated gene to be sequenced. For example, in the presence of cardiomyopathy, TAZ (Barth syndrome) sequencing may be diagnostic, while in the presence of cardiac and genitourinary malformations sequencing of G6PC3 may lead to diagnosis. Poor growth, malabsorption, fatty stool, and bone malformation suggest SBDS mutations that are found in most patients with Shwachman-Diamond syndrome (SDS). |
|||
|
• A targeted NGS panel including all genes known to be mutated in CN (>30) is a reasonable first step that provides uniform sequencing coverage for all genes of interest and requires simpler bioinformatics analysis. |
|||
|
• The choice of genes within the panel should include not only all those that strictly cause neutropenia when mutated but also genes resulting in diseases in which neutropenia is a secondary feature (immunodeficiency/immune dysregulation, metabolic and nutritional deficiency, and other BMF syndromes) whole exome sequencing (WES) can also be used in cases where no mutations were detected in a panel |
|||
|
A.5.d. How to diagnose cyclic neutropenia? |
|||
|
EHA-EuNet-INNOCHRON 2023 |
Complete blood counts (CBC) in family members, serial blood counts twice or thrice a week over a period of 6 weeks to exclude CyN.
|
|
GPS |
|
A.5.e. How to diagnose autoimmune neutropenia? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
• Antineutrophil antibody testing should be performed as first-line investigation in both children and adults. • Indirect granulocyte immunofluorescence test (GIFT) is recommended as a first-line assay in reference laboratories. • A positive GIFT in combination with laboratory tests and clinical picture can support diagnosis of autoimmune neutropenia (AIN) but does not exclude the diagnosis of other types of neutropenia. • With a negative indirect GIFT, if the clinical suspicion of AIN remains high, GIFT should be repeated several times. |
|
GPS |
|
A.5.f. Is family Screening indicated in patients with genetically proven congenital neutropenia? |
|||
|
EHA-EuNet-INNOCHRON 2023 |
• Following identification of the responsible gene(s), Sanger sequencing is also recommended for mutation screening of the members of affected families. • Multigene NGS or WES ideally should include patient and parental DNA (trio analysis). |
|
GPS |
|
A.5.g. What is the role of bone marrow examination in the diagnosis of children with neutropenia? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
Diagnostic BM with morphology, cytogenetics, and NGS of genes related to myeloid malignancies should be performed: 1. In pediatric patients with severe and moderate chronic neutropenia with the exception of patients with primary AIN with positive anti-granulocyte antibodies and drug-induced neutropenia. N.B: Patients with congenital neutropenia may have positive antibody test. 2. In patients with suggested AIN but negative granulocyte antibody test, if patients suffer from recurrent infections. 3. In any patients before G-CSF treatment. Repeated BM follow-up should be performed in patients: With decreasing ANC or additional changes in other blood cell counts (e.g., anemia and thrombocytopenia) or erythrocyte indices. |
|
GPS |
|
A.5.h. What is the role of flowcytometry for the diagnosis children presenting with chronic neutropenia? |
|||
|
EHA-EuNet-INNOCHRON 2023 |
FC is an important tool in the diagnosis of neutropenia associated with PID syndromes such as ALPS, CVID, and HIGM syndrome. Assessment of a PNH clone by FC testing is also recommended. Flow FISH is recommended when a telomere biology disorder is suspected. |
|
GPS |
|
A.5.i. What is the role of genetic testing in the diagnosis of children with neutropenia? |
|||
|
EHA-EuNet-INNOCHRON 2023
|
• Genetic diagnosis is important to confirm the diagnosis of CN, estimate the risk for MDS/AML, support stem cell donor selection for patients, and family counseling. |
|
GPS |
|
• When the clinical picture, inheritance, or bone marrow features (i.e., block at the promyelocyte stage) are indicative of a specific gene mutation, single-gene analysis by Sanger sequencing technique could be applied. |
|||
|
• For CN where the clinical picture does not suggest a specific genetic cause, we recommend the use of NGS techniques such as multigene panels or targeted WES. |
|||
|
• For patients for whom a genetic cause is not identified by the above methods, WGS and RNA-sequencing may be powerful diagnostic tools. |
|||
|
• NGS analysis of bone marrow or peripheral blood for acquired somatic variants is recommended for patients with chronic neutropenia. |
|||
|
• Screening for known mutations is recommended in family members. |
|||
|
• It is important to validate germline mutations mainly in fibroblasts or hair follicles keratinocytes (cells from buccal swab are less indicated for possible blood contamination), in the presence of leukemic blasts in PB. |
|||
|
A.5.j. What is the required initial evaluation for children presenting with fever and neutropenia? |
|||
|
ASCO 2023 |
• Adopt a validated risk stratification strategy and incorporate it into routine clinical management |
Low |
Strong |
|
ASCO 2023 |
• Obtain blood cultures at the onset of FN from all lumens of central venous catheters. |
Low |
Strong |
|
ASCO 2023 |
• Consider obtaining peripheral blood cultures concurrent with central venous catheter cultures. |
Moderate |
Conditional |
|
ASCO 2023 |
• Consider urinalysis and urine culture in patients where a clean-catch, mid-stream specimen is readily available |
Low |
Conditional |
|
ASCO 2023 |
• Obtain chest radiography only in patients with respiratory signs or symptoms. |
Moderate |
Strong |
|
Table 4. Recommendations on treatment of neutropenia |
|
|
|
|
B. Management of neutropenia in pediatric age groups |
|
|
|
|
Source Guideline |
Recommendations |
QoE |
SoR |
|
B.1. What is the treatment of infection in patients with fever and non-severe neutropenia? |
|||
|
Fioredda et al 2011* |
Individuals with an ANC >1.0 x 10*9 /L can be managed as per those with normal ANC.. |
|
GPS |
|
B.2. What is the treatment of fever and neutropenia (FN)? |
|||
|
|
Infants with primary AIN should be excluded from these recommendations, since ANC is not indicative of infectious risk in these patients. |
|
GPS |
|
|
I. Initial treatment |
|
|
|
|
In low-risk FN |
|
|
|
ASCO 2023 |
· Consider initial or step-down outpatient management if the infrastructure is in place to ensure careful monitoring and follow-up. |
Moderate |
Conditional |
|
ASCO 2023 |
· Consider oral antibacterial therapy administration if the patient is able to tolerate this route of administration reliably. |
Moderate |
Conditional |
|
|
In high-risk FN |
|
|
|
ASCO 2023 |
· Use monotherapy with an antipseudomonal b-lactam, a fourth-generation cephalosporin or a carbapenem as empiric antibacterial therapy in pediatric high-risk FN |
High |
Strong |
|
ASCO 2023 |
· Reserve addition of a second anti-Gram-negative agent or a glycopeptide for patients who are clinically unstable, when a resistant infection is suspected, or for centers with a high rate of resistant pathogens |
moderate |
Strong |
|
|
II. Ongoing management |
|
|
|
|
Modification of treatment |
|
|
|
ASCO 2023 |
· In patients who are responding to initial empiric antibacterial therapy, discontinue double coverage for Gram-negative infection or empiric glycopeptide (if initiated) after 24-72 hours if there is no specific microbiologic indication to continue combination therapy. |
Moderate |
Strong |
|
ASCO 2023 |
· Do not broaden the initial empiric antibacterial regimen based solely on persistent fever in patients who are clinically stable. |
Low |
Strong |
|
ASCO 2023 |
· In patients with persistent fever who become clinically unstable, escalate the initial empiric antibacterial regimen to include coverage for resistant Gram-negative, Gram-positive and anaerobic bacteria. |
Very low |
Strong |
|
|
Cessation of treatment |
|
|
|
ASCO 2023 |
· In both high-risk and low-risk FN patients who have been clinically well and afebrile for at least 24 hours, discontinue empiric antibacterial therapy if blood cultures remain negative at 48 hours if there is evidence of marrow recovery |
Low |
Strong |
|
ASCO 2023 |
· In patients with low-risk FN who have been clinically well and afebrile for at least 24 hours, consider discontinuation of empiric antibacterial therapy if blood cultures remain negative at 48 hours despite no evidence of marrow recovery. |
Moderate |
Conditional |
|
B.3. What is the treatment of severe chronic neutropenia |
|||
|
|
I. The use of G-CSF |
|
|
|
Fioredda et al 2011 |
· Patients usually need life-time treatment. |
|
GPS |
|
· The therapeutic target (ANC ≥1.0 × 10*9/L and ≤5.0 × 10*9/L) is considered achievable with a G-CSF starting dose of 5 μg/kg/d, but the individual variability of the response may require modifications of the subsequent doses. |
|
GPS |
|
|
· It is suggested to increase G-CSF over 2 weeks up to doubling initial dose if ANC remain <1.0 × 10*9/L and to reduce G-CSF if ANC reach >5.0 × 10*9/L. |
|
GPS |
|
|
· Maintain the initial dose if the target ANC (≥1.0 × 10*9/L and ≤5.0 × 10*9/L) is achieved and increase by 2.5 μg/kg/day every 5–7 days if ANC remains <1.0 × 10*9/L. N.B: Depending on clinical situation and known underlying genotype doubling of G-CSF may be indicated up to 50µg/kg/day to evaluate G-CSF response |
|
GPS |
|
|
· Experts agreed on the non-superiority of lenograstim vs. filgrastim as the number of infections was similar in patients treated with the two preparations. |
|
GPS |
|
|
· The use of pegylated G-CSF in neutropenia has been rarely reported in pediatric age. |
|
GPS |
|
|
|
II. Hematopoietic stem cell transplantation (HSCT) |
|
|
|
Fioredda et al 2025 |
Strong indications for HSCT include: (1) Established transformation to MDS/Acute Leukemia or bone marrow dysplastic features with high‐risk acquired cytogenetic abnormalities (monosomy 7, trisomy 8, and trisomy 21) or with a combination of acquired leukemia‐associated somatic mutations (e.g., RUNX1, ASXL1, and SETBP1). CSF3R mutations alone are not an indication of HSCT. (2) CN due to mutations carrying an intrinsic high risk of leukemic transformation per se, i.e., GATA2 mutations, high‐risk ELANE mutations, or clones with biallelic TP53 mutations in SDS. (3) No response to G‐CSF (doses > 20 mcg/kg/d to reach ANC of 1.0 × 109/L), poor response to G‐CSF (doses between 10 and 20 mcg/kg/d failing to reach ANC of 1.0 × 109/L) or poor control of infection irrespective of the G‐CSF dose.
Potential indications to HSCT adequate management of infections with G‐CSF at “intermediate doses” (10–15 mcg/kg/d) with availability of a healthy HLA‐identical sibling or HLA identical matched donor.
Weak indication of HSCT GCSF response at doses up to 10 μg/kg/d, good tolerability and compliance to daily subcutaneous injections, infections control, and unavailability of HLA matched donors. |
|
GPS
GPS |
|
B.4. What is the treatment of cyclic neutropenia (CyN)? |
|||
|
Fioredda et al 2011 |
· In cyclic neutropenia (CyN) G-CSF treatment has been shown to shorten the degree and the duration of neutropenia conferring the patients a better quality of life. For these reasons, the long-term pattern of treatment was considered adequate to CyN. |
|
GPS |
|
Fioredda et al 2011 |
· This has to be intended as a life-time treatment not to be performed daily but only during the lowest levels of neutrophils in cycling periods of neutropenia. |
|
GPS |
|
Fioredda et al 2011 |
· Usually, patients affected by CyN need lower G-CSF doses (median 2.4 μg/kg/day) than SCN patients. A starting dose of 1–3 μg/kg/d should maintain ANC around 1.5 × 10*9/L; for lower counts the dosage may be increased up to two-fold within 2–4 weeks. · Given the ANC fluctuations it is suggested to calculate the final G-CSF dose after a number of observations. |
|
GPS |
|
Fioredda et al 2011 |
· Daily administration during neutropenia is more appropriate than the intermittent schedule. Initial suggested dose during neutropenic periods is 2 μg/kg/day. If ANC target is not achieved, increases of 2 μg/kg/d every neutropenic phase are recommended |
|
GPS |
|
|
· the panel recommends the use of G‐CSF in severe CN and in CyN, particularly in those patients with recurrent or severe infections. · The panel considers ANC over 1.0 × 109/L as the protective threshold against infections. For CyN, a lower nadir may be accepted. · In CyN, G‐CSF doses may be lower than in severe CN. The standard dose should be ≤3 mcg/kg/d continuously. G‐CSF may be also given every other day. Dosage may be adjusted to avoid nadir <0.5 × 109/L and clinical conditions such as mouth ulcers, fevers, or infections. |
|
|
|
B.5. What is the treatment of idiopathic neutropenia (IN)? |
|||
|
Fioredda et al 2011 |
· In the majority of cases, IN requires an on-demand treatment only during infections or surgery. In these patients an initial does of 1–2 μg/kg/d is considered sufficient to achieve goal ANC. |
|
GPS |
|
Fioredda et al 2011 |
· Some patients with a diagnosis of IN may have severe and/or recurrent infections and may require long-term treatment. A starting dose (1–2μg/kg/die) of G-CSF can be sufficient to achieve the goal ANC and a rhythm of 2–3 times/week may ensure protective values of neutrophils. |
|
GPS |
|
B.6. What is the treatment of autoimmune neutropenia (AIN)? |
|||
|
|
I. G-CSF |
|
|
|
Fioredda et al 2011 |
· The use of G-CSF in AIN is generally necessary only in case of severe infections and therefore the on-demand treatment is considered appropriate. Some forms of AIN may have severe and/or recurrent infections requiring a long-term treatment. |
|
GPS |
|
· An initial low dose of G-CSF (1–2 mg/kg/die) to be subsequently adjusted. |
|
GPS |
|
|
|
II. Other treatment modalities: |
|
|
|
Fioredda et al 2011 |
· Various immune-regulating drugs (e.g., cyclosporine, methotrexate, low-dose cyclophosphamide) have been used in refractory cases, but overall efficacy has not been determined. |
|
GPS |
|
· In case of an underlying immune dysregulation/deficiency, particularly in childhood, more targeted treatments (e.g., mycophenolate mofetil and rapamycin) may be used. |
|
GPS |
|
|
· The role of rituximab (and other antibodies to CD20) is well established as effective treatment for many autoimmune disorders. However, its role as treatment for AIN is unclear. |
|
GPS |
|
Table 5. Prevention and monitoring of neutropenia |
|
|
|
|||
|
C. Prevention and monitoring of neutropenia in pediatric age groups |
|
|
|
|||
|
Source Guideline |
Recommendations |
QoE |
SoR |
|
||
|
I. Prevention of complications |
|
|||||
|
C.1. Identify groups at high-risk for infections)? |
|
|||||
|
IDSA 2020 |
• Pediatric patients actively receiving cytotoxic chemotherapy. • Down Syndrome. • Children with immune deficiency. • Patient with Central venous catheter. • Compromise of mucosal barriers (e.g. mucositis, typhlitis). • Myelosuppression. • Children with hematologic malignancies have an increased risk relative to children with solid tumors. |
|
GPS
|
|
||
|
C.2. How to prevent infection; pharmacological (chemoprophylaxis) and non-pharmacological in high-risk population? |
|
|||||
|
|
1. Non-pharmacological |
|
|
|
||
|
Martínez Campos et al 2023* |
• Personal protective equipment (gown, gloves. . .) in patients with contagious infection or colonization by multidrug resistant (MDR) microorganisms. |
|
GPS |
|
||
|
• Aseptic technique in central venous catheter (CVC) handling. |
|
GPS |
|
|||
|
• Hand, surface and food preparation hygiene measures. |
|
GPS |
|
|||
|
• Chlorhexidine gluconate (CHG) baths (CHB) involve cleansing the patient’s body each day with CHG-impregnated wipes. Literature from pediatric patients with temporary CVCs inserted during critical care admissions suggests that CHB may have a role in reducing the rate of CLABSIs in these patients. |
|
GPS |
|
|||
|
• Avoid contact with pets associated with a high risk: turtles, cat’s litter, stables and new pets. This does not apply to other pets that are correctly vaccinated, in regard to which the sole precaution is to avoid contact with their faeces. |
|
GPS |
|
|||
|
• Prevention of IFI (high risk patients/situations) - Avoid flowers and plants in hospital rooms and the bedroom of the patient. - Hospital rooms with HEPA filters and positive pressure (>12 room air changes/h). - Use of FPP2 masks in closed spaces without HEPA filters (except the usual place of residence, where ventilation, avoiding plants, moisture and construction work). - Recommendation not supported by evidence: § Avoid pools, especially public or indoor ones. § Low bacteria diet not proven to reduce the incidence or severity of infection. § Personal protective equipment for patients in absence of microbial isolation that justifies it.
|
|
GPS |
|
|||
|
|
2.Pharmacological: |
|
|
|
||
|
Martínez Campos et al 2023* |
• G-CSF in patients with solid tumors receiving intensive chemotherapy, who need to maintain cytotoxic dose/chemotherapy intensity and/or with previous history of fever neutropenia (FN). |
|
GPS |
|
||
|
• Annual vaccination against seasonal flu of patients and their household contacts and health care workers. |
|
GPS |
|
|||
|
C.3.a. What are the guidelines for Antibacterial Prophylaxis in different causes of neutropenia? |
|
|||||
|
IDSA 2020 |
• Consider systemic antibacterial prophylaxis administration in children with AML and relapsed ALL receiving intensive chemotherapy expected to result in severe neutropenia (absolute neutrophil count <500/µl for at least 7 days). |
High |
Conditional |
|
||
|
IDSA 2020 |
• Do not use systemic antibacterial prophylaxis routinely for children receiving induction chemotherapy for newly diagnosed ALL. |
Low |
Conditional |
|
||
|
IDSA 2020 |
• Do not use systemic antibacterial prophylaxis for children whose therapy is not expected to result in severe neutropenia (ANC <500/µl for at least 7 days). |
Moderate |
Strong |
|
||
|
IDSA 2020 |
• Do not use systemic antibacterial prophylaxis routinely for children undergoing autologous HSCT. |
Moderate |
Conditional |
|
||
|
IDSA 2020 |
• Do not use systemic antibacterial prophylaxis routinely for children undergoing allogeneic HSCT. |
Moderate |
Conditional |
|
||
|
C.3.b. What to use (Type, dose, route)? how long? |
|
|||||
|
IDSA 2020 |
• Levofloxacin is the preferred agent if systemic antibacterial prophylaxis is planned. |
Moderate |
Strong |
|
||
|
C.3.c. When should systemic antibacterial prophylaxis be stopped? |
|
|||||
|
IDSA 2020 |
• If systemic antibacterial prophylaxis is planned; we suggest that administration be restricted to the expected period of severe neutropenia (absolute neutrophil count <500/μL. |
Low |
Conditional |
|||
|
C.4.a. What are the guidelines for Antifungal Prophylaxis in different causes of neutropenia? When to start? & What to use (Type, dose, route)? & how long? How do we follow its side effects and effectiveness? |
|
|||||
|
IDSA 2020 |
Acute myeloid leukemia (AML) • Administer systemic antifungal prophylaxis to children and adolescents receiving treatment of AML that is expected to result in profound and prolonged neutropenia. |
High |
Strong |
|
||
|
|
Acute lymphoblastic leukemia (ALL) |
|
|
|
||
|
IDSA 2020 |
• Consider administering systemic antifungal prophylaxis to children and adolescents with newly diagnosed and relapsed ALL at high risk for invasive fungal disease (IFD) |
Low |
Conditional |
|
||
|
• Do not routinely administer systemic antifungal prophylaxis to children and adolescents with ALL at low risk for IFD. |
Low |
Strong |
|
|||
|
IDSA 2020 |
Other malignancies including most patients with lymphomas and solid tumors • Do not routinely administer systemic antifungal prophylaxis to children and adolescents with cancer at low risk for IFD, such as most pediatric patients with lymphomas and solid tumors. |
Moderate |
Strong |
|
||
|
|
Hematopoietic stem cell transplantation (HSCT) |
|
|
|
||
|
IDSA 2020 |
• Administer systemic antifungal prophylaxis to children and adolescents undergoing allogeneic HSCT pre-engraftment and to those receiving systemic immunosuppression for the treatment of graft-versus host disease (GVHD). |
Moderate |
Strong |
|
||
|
• We suggest that systemic antifungal prophylaxis not be used routinely in children and adolescents undergoing autologous HSCT. |
High |
Strong |
|
|||
|
• In choosing a mold-active agent, administer an echinocandin or a mold-active azole. |
Moderate |
Strong |
|
|||
|
• Do not use amphotericin routinely as systemic antifungal prophylaxis. |
Low |
Strong |
|
|||
|
C.4.b. When should systemic antifungal prophylaxis be started and stopped? |
|
|||||
|
IDSA 2020 |
If systemic antifungal prophylaxis is warranted, consider administration during periods of observed or expected severe neutropenia. For allogeneic HSCT recipients, consider administration during systemic immunosuppression for GVHD treatment
|
Low |
Conditional |
|
||
|
C.5. What are the guidelines for preventing Clostridium difficile infection (CDI) in children and adolescents with neutropenia? When to start? & What to use (Type, dose, route)? & what intervention? how long? How to follow the side effects and the effectiveness? |
|
|||||
|
ASCO 2018 |
Do not use probiotics routinely for the prevention of CDI in children and adolescents with cancer and pediatric HSCT patients |
Low |
Conditional |
|
||
|
ASCO 2018 |
Use either oral metronidazole or oral vancomycin for the treatment of non-severe CDI in children and adolescents with cancer and pediatric HSCT patients |
Low |
Strong |
|
||
|
ASCO 2018 |
Use oral vancomycin for the treatment of severe CDI in children and adolescents with cancer and pediatric HSCT patients |
Low |
Strong |
|
||
|
ASCO 2018
|
Do not use monoclonal antibodies routinely for the treatment of CDI in children and adolescents with cancer and pediatric HSCT patients |
Low |
Strong |
|
||
|
C.6. What are the guidelines for Prophylaxis against Pneumocystis jirovecii pneumonia (PJP)? |
|
|||||
|
ASCO 2020 |
· Risk for PJP pneumonia is highest in patients with leukemia and lymphoma, prophylaxis is also recommended for children with solid tumors undergoing chemotherapy that is likely to cause lymphopenia. · Data in patients with HIV shows that the risk of PJP is highest in those with CD4 counts under 200 x10*6/L, and one study from the solid organ transplantation population found that odds of PJP infection were significantly increased when absolute lymphocyte count (ALC) was < 500 x10*6/L. · Sulfamethoxazole (TMP-SMX) is highly effective. · For patients who do not tolerate TMP-SMX, pentamidine, dapsone, and atovaquone are alternative options, · Optimal duration of PJP prophylaxis is unknown. One guideline suggests continuing until 3 months after the end of therapy and normalization of lymphocyte count.
|
|
GPS |
|
||
|
C.7. What are the guidelines for the prevention of oral and oropharyngeal Mucositis? When to start? & What to use (Type, dose, route)? & what intervention? how long? How to follow the side effects and the effectiveness? |
|
|||||
|
Patel P et al, 2021 |
Use cryotherapy for older, cooperative pediatric patients receiving treatment for cancer or undergoing HSCT who will receive short infusions of melphalan or 5-fluorouracil. |
|
GPS |
|
||
|
Patel P et al, 2021 |
Consider using cryotherapy for older, cooperative pediatric patients receiving treatment for cancer or undergoing HSCT who will receive short infusions of chemotherapy associated with mucositis other than melphalan or 5-fluorouracil. |
|
GPS |
|
||
|
Patel P et al, 2021** |
Do not administer palifermin routinely to pediatric patients with cancer receiving treatment for cancer or undergoing HSCT. |
|
GPS |
|
||
|
Patel P et al, 2021** |
Use intraoral photo-biomodulation therapy in the red-light spectrum (620–750 nm) for pediatric patients undergoing autologous or allogeneic HSCT and for pediatric patients who will receive radiotherapy for head and neck carcinoma. |
|
GPS |
|
||
|
Patel P et al, 2021** |
Consider using intraoral photo-biomodulation therapy in the red-light spectrum (620–750 nm) for pediatric patients who will receive radiotherapy for head and neck cancers other than carcinoma. |
|
GPS |
|
||
|
Patel P et al, 2021**
|
Do not administer GCSFs to pediatric patients receiving treatment for cancer or undergoing HSCT for the purpose of mucositis prevention. |
|
GPS |
|
||
|
C.8. What are the guidelines for growth factor therapy (granulocyte stimulating factor (GSF)? |
|
|||||
|
Martínez Campos et al 2023 |
The use of G-CSF in in patients with solid tumors receiving intensive chemotherapy, who need to maintain cytotoxic dose/chemotherapy intensity and/or with previous history of FN reduces the duration of neutropenia. |
|
GPS |
|
||
|
C.9. How to provide vaccination? When to revaccinate? And how? |
|
|||||
|
Martínez Campos et al 2023 |
Annual vaccination against seasonal flu of patients and their household contacts and health care workers. Despite potential lack of efficacy in certain populations, the American Academy of Pediatrics and Center for Disease Control still recommend yearly influenza vacations in pediatric oncology patients based on potential benefit with low risk of reaction |
|
GPS |
|
||
|
II. Follow up and monitoring |
|
|
||||
|
C.10. What are the clinical indicators that should be assessed in each clinic visit? |
|
|||||
|
EHA-EuNet 2023
|
• Careful clinical examination of skin and mucous membranes, upper and lower respiratory tract and abdomen to exclude underlying infection, lymphadenopathy, and/or hepatosplenomegaly. |
|
GPS |
|
||
|
EHA-EuNet 2023
|
• Vital signs: temperature, heart rate, respiratory rate, blood pressure and oxygen saturation. • Pediatric assessment triangle (early detection of sepsis). • Review of systems, including neurologic assessment, perfusion, skin and mucosae (oral and perianal, avoid rectal palpation), any area with pain, scars, devices. |
|
GPS |
|
||
|
C.11. What are the laboratory tests that should be done during the follow-up of the neutropenic child and how frequent? |
|
|||||
|
EHA-EuNet 2023 |
In chronic neutropenia patients, we recommend performing CBC with differential WBC counts and morphological evaluation every 3–4 months. |
|
GPS |
|
||
|
EHA-EuNet 2023 |
When approaching adulthood, CN patients should be transferred to a dedicated hematology specialist. |
|
GPS |
|
||
|
C.12.a. How often the bone marrow study should be done? |
|
|||||
|
EHA-EuNet 2023
|
· Annual BM and cytogenetics follow-up should be performed in patients with • congenital BM failure syndromes independent of ANC and treatment with G-CSF. • undefined SCN (after extensive investigation) with G-CSF treatment, may be considered. |
|
GPS |
|
||
|
EHA-EuNet 2023 |
· Repeated BM follow-up should be performed in patients with decreasing ANC or additional changes in other blood cell counts (e.g., anemia and thrombocytopenia) or erythrocyte indices. |
|
GPS |
|
||
|
C.12.b. What are the surrogate markers of transformation into MDS/leukemia? What are the tests that should be done on the bone marrow sample? |
|
|||||
|
EHA-EuNet 2023
|
· The key markers of malignant transformation to MDS or leukemia in CN patients are the following: typical dysplastic features in PB (pseudo Pelger-Huet anomaly, hypogranulation, hyper segmentation, reticulated nucleus, and ringed-shaped nuclei) and BM (defective granulation, maturation arrest at myelocyte stage, and increase in monocytoid forms); cytogenetic abnormalities(e.g.,CSF3R, RUNX1, and ASXL1); and high frequency of somatic mutations in leukemia-associated driver genes. · The most common chromosomal defects in patients with CN at the MDS stage are trisomy 21 and monosomy 7. |
|
GPS
GPS |
|
||
*: Consensus document on the management of febrile neutropenia in paediatric haematology and oncology patients of the Spanish Society of Pediatric Infectious Diseases (SEIP) and the Spanish Society of Pediatric Hematology and Oncology (SEHOP). 23
**: Clinical practice guideline for the prevention of oral and oropharyngeal mucositis in pediatric cancer and hematopoietic stem cell transplant patients: 2021 update. 24
- Acknowledgements
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Clinicians subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Galila Mohamed Mokhtar |
Paediatrics Department, Paediatric Haematology/ Oncology & BMT Unit Faculty of Medicine, Ain Shams University, Cairo, Egypt |
Editor, Clinical expert, GAG member |
|||
|
Prof. Amira Adly |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Dina Ezzat |
Professor of Paediatrics and Paediatric Haematology, Beni-Suef University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Gehan Lotfy |
Professor of Paediatrics , Minia University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Hanafy Hafez |
Professor of Pediatric Oncology, National Cancer Institute, Cairo University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Hoda Hassab |
Professor of Paediatrics and Paediatrics Haematology, Alexandria University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Ilham Youssry |
Professor of Paediatrics and Head of the Paediatric Haematology & BMT unit, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Iman Ragab |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Laila Sherief |
Professor of Paediatrics and Paediatrics Haematology & Oncology, Zagazig University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Marwa Zakaria |
Associate Professor of Paediatrics and Pediatric Hematology & Oncology, Zagazig University, Egypt |
Clinical expert GAG member |
|||
|
Prof. Mervat Hesham |
Professor of Pediatrics and Pediatric Hematology & Oncology, Zagazig University, Egypt |
Clinical expert GAG member |
|||
|
Dr. Naglaa Shaheen |
Consultant of Pediatric Hematology, Health Insurance Organization |
Clinical expert GAG member |
|||
|
Dr. Nouran Yousef |
Lecturer of Paediatrics, Paediatric Diabetes, Endocrinology & Metabolism unit, Ain Shams University, AFCM, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Rasha Abdel-Raouf Afifi |
Professor of Paediatrics and Paediatric Haematology, Cairo University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Dr. Sara Makkeyah |
Associate Professor of Paediatrics, Paediatric Haematology / Oncology Unit & BMT, Ain Shams University, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Prof. Sonia Adolf |
Professor of Paediatrics and Paediatric Haematology, National Research Center, Cairo, Egypt |
Clinical expert GAG member |
|||
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Development/ Adaptation Group (Guideline Methodologists subgroup) |
|||||
|
Name |
Affiliation, Area of expertise / Country / Primary location [work] |
Contribution |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
Overseeing the adolopment process of the guidelines, training and education of new members, revision of the final draft, and organizing online meetings of GDG |
|||
|
Dr. Yasser Sami Amer |
- Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; - Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; - Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland - Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
Overseeing the adolopment process of the guidelines, training and education of new members, participating in writing up the methodology of adaptation process, guideline appraisal, and revision of the final draft |
|||
|
Assistant Professor of Pediatrics Ain Shams University, Egypt |
participating in search and guideline appraisal |
||||
|
- Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. - Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. - Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
Participating in multiple steps of the guideline adaptation process. |
||||
|
Dr. Heba Hussein |
- Associate Professor of Oral Medicine, Oral Diagnosis, and Periodontology Department, Faculty of Dentistry, Cairo University, Cairo, Egypt |
Participating in multiple steps of the guideline adaptation process. |
|||
|
Dr. Lamis Mohsen Elsholkamy
- |
- Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
Dr. Ahmed Mahmoud Youssef
- |
Fellow of Pediatrics, General Organization for Teaching Hospitals and Institutes (GOTHI). - Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
Participating in multiple steps of the guideline adaptation process, Writing the methodology of adaptation process and revised the whole document. |
|||
|
External Reviewers Group (ERG) |
|||||
|
External Reviewer(s) for Clinical Content |
|||||
|
|||||
|
Prof. Nourmin Kaddah |
Professor of Pediatrics & Pediatric hematology Cairo University, Egypt |
||||
|
Prof. Mohamed Ramadan El-Shanshoury |
Professor of Pediatrics Tanta University, Egypt |
||||
|
Prof. Mohamed Tarif Hamza |
Professor of clinical pathology, Ain Shams University, Egypt |
||||
|
International Peer Reviewers |
|||||
|
Cornelia Zeidler |
|
||||
|
External Reviewer(s) for methodology |
|||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
||||
· The GDG/ GAG acknowledge EPG for its help in completing this project.
· We acknowledge the American Society of Clinical Oncology (ASCO), the European-Hematology association - European Network for Innovative Diagnosis and Treatment of Chronic Neutropenias network (EHA-EuNet-INNOCHRON), and the Infectious Diseases Society of America (IDSA) for their cooperation in providing the permission for adapting our guidelines.
· Finally, we wish the best for all our patients and their families who inspired us. It is for them this work is being finalized.
Funding
· This work is not related to any pharmaceutical or industrial company. The members of the GDG/ GAG and their institutes and universities volunteered their participation and contributions.
- Abbreviations
|
|
- Introduction
Neutropenia is defined as a reduction in the absolute neutrophil count (ANC) below the lower limit of the normal range for the age and ethnic origin of the affected subject. In pediatrics, it is important to note that normal values for the ANC vary by age. The lower normal limit in the 1st week of life is 5000/mm3, and this value decreases to 1000/mm3 between the 2nd week of life and 1 year of age. After the age of 1 year, an ANC below 1500/mm3 is defined as neutropenia, between 1000 and 1500/mm3 as mild, between 500 and 1000/mm3 as moderate, and below 500/mm3 as severe neutropenia 1. Certain populations (e.g., African-Americans, Yemenite Jews, and certain Arabs) normally have slightly lower white blood cell count and ANC values 2. Neutropenia is a decrease in circulating (i.e., nonmarginal) neutrophils in the blood 1. The nonmarginal pool constitutes only 4-5% of total body neutrophil stores; while most of the neutrophils in the body are contained in the bone marrow, either as mitotically active (one-third) or postmitotic mature cells (two-thirds) 2.
Neutropenia can be classified as congenital or acquired. Congenital neutropenia is a heterogeneous hereditary group of disorders that are characterized by intermittent episodes, or continuous severe or moderate neutropenia persisting for at least 3 months and can develop at any stage of the proliferation and maturation phases of neutrophils, secondary to genetic causes. Although it occurs only rarely, congenital neutropenia is a significant condition that causes recurrent infections and has a chronic course3,4. Acquired neutropenia is more common than congenital neutropenia, with the most common causes being infections, drugs, and chronic benign neutropenia in infancy and childhood. It is believed that the majority of cases defined as chronic benign neutropenia in infancy and childhood represent an autoimmune neutropenia that is parallel to childhood idiopathic thrombocytopenic purpura 4. Apart from these, bone marrow involvement, nutritional causes, immunological disorders, metabolic disorders, autoimmune lymphoproliferative syndrome, and Evans syndrome are other causes of acquired neutropenia 3,4.
Neutropenic individuals are at increased risk of infection. The greatest susceptibility of infection is seen with ANC below 500 neutrophils/mm3, and when neutrophil production remains low without recovery. Gram-negative bacteria, viruses, fungi, and opportunistic pathogens are also common. The cornerstone for evaluating a patient with neutropenia is a thorough history and physical exam in addition to laboratory data 5. In recent years, advances in genomics have identified novel genes implicated in the pathogenesis and/or evolution of neutropenia, unraveled the underlying pathogenic mechanisms, and opened the way for novel tailored therapies 1-4,6,7. A number of comprehensive reviews have been produced by experts in the field aiming to disseminate the knowledge and guide clinicians for the accurate diagnosis, follow-up, and treatment of neutropenia patients, particularly those with chronic disease 1-4,6-11. Real world data, however, arising from a survey within the Cooperation in Science and Technology European Network for the Innovative Diagnosis and Treatment of Chronic Neutropenias (EuNet-INNOCHRON; https://eunet-innochron.eu/) involving physicians with special interest in neutropenias have shown that the work-up of patients with chronic neutropenia is mostly based on the physicians’ experience and local practices rather than on the guided clinical and laboratory evidence 12. Thus, the diagnosis and monitoring of neutropenic patients remains varied and challenging. Overall, continuous education of hematologists on known and arising neutropenia entities and guided diagnostic, follow-up, and treatment strategies are particularly important.
➡️Purpose and Scope
These guidelines have been developed to standardize the delivery of services and to implement the guidance on the prevention, diagnosis and management of neutropenia in pediatric age groups. It provides guidance to primary health care providers, pediatricians and specially trained nurses.
The guidelines aimed to provide an evidence-based document for the
- Identification of conditions associated with neutropenia.
- Diagnostic approaches in the target population.
- Treatment of neutropenia and the associated complications in the target population.
- Prevention and monitoring of neutropenia.
This version of the guideline includes recommendations and good practice statements for
· Diagnosis of neutropenia in pediatric age groups.
· Management of neutropenia in pediatric age groups.
· Prevention of neutropenia in pediatric age groups
- Methods
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. Keywords used for the search are neutropenia, children, cyclic neutropenia, congenital neutropenia, and febrile neutropenia.
Inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
• Selecting only evidence-based guidelines (guideline must include a report on the methodology of development including the systematic literature searches and explicit links between individual recommendations and their supporting evidence)
• Selecting national and/or international guidelines
• Specific range of dates for publication (using Guidelines published or updated in 2018 and later or the last 7 years)
• Selecting peer-reviewed publications only
• Selecting guidelines written in the English language
• Excluding guidelines written by a single author
The following three categories of databases and websites were searched:
1. CPG databases and libraries (e.g., GIN, ECRI, SIGN, DynaMed, BIGG-REC PAHO)
2. Bibliographic databases (e.g., PubMed, Google Scholar)
3. Specialized professional societies (related to the pediatric subspecialty)
All retrieved Guidelines were screened and appraised using the AGREE II instrument (www.agreetrust.org) 13-16 by at least two members. The panel decided a cut-off point or ranked the guidelines (any guideline scoring above 60% on the rigor dimension was retained)
After reviewing all the previous criteria, the GDG/ GAG recommended using 4 guidelines:
1- Guideline for the Management of Fever and Neutropenia in Pediatric Patients with Cancer and Hematopoietic Cell Transplantation Recipients: 2023 Update. ASCO (2023)
2- The European Guidelines on Diagnosis and Management of Neutropenia in Adults and Children: A Consensus Between the European Hematology Association and the EuNet-INNOCHRON COST Action. EHA-EuNet-INNOCHRON (2023)
3- Guideline for Antibacterial Prophylaxis Administration in Pediatric Cancer and Hematopoietic Stem Cell Transplantation. IDSA (2020)
4- Guideline for the Management of Clostridium Difficile Infection in Children and Adolescents with Cancer and Pediatric Hematopoietic Stem-Cell Transplantation Recipients. ASCO (2018)
We did Adolopment for these guidelines: (Adoption, Adaptation, and Development)17-20
- Adoption for most of the guideline recommendations.
- Development of Good Practice Statement
Contributors to the guideline development process:
Guideline Development Group (GDG)/ Guideline Adaptation Group (GAG):
The GDG/ GAG included two subgroups: the clinicians/ healthcare providers subgroup and the guideline methodologists’ subgroup.
➡️Clinicians Subgroups
The clinicians’ subgroup or clinical panel for this guideline included experts with a range of knowledge, technical skills and diverse perspectives in the field of pediatric hematology.
The main functions of the clinical panel were adolopment of the retrieved Guidelines, determining the scope of the guideline and guideline, reviewing the evidence, and formulating evidence-informed recommendations in case of changing strength of recommendations.
➡️Guideline Methodologists Subgroup
There were 5 guideline methodologists with expertise in guidelines development, adaptation, GRADE and translation of evidence into recommendations. Methodologists provided orientation and overview of evidence-informed guideline development processes using the GRADE approach, guideline adaptation using the Adapted ADAPTE, provided AGREE II assessment of the source guidelines in collaboration with the clinician's subgroup, and generation of the EtD frameworks whenever applicable.
➡️External Review Group:
The External Review Group for this guideline comprises 3 clinical national experts who have interest and expertise in the field of neutropenia as well as eminent international reviewers.
They were identified by the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) as people who can provide valuable insights during the guideline development process.
The External Review Group was asked to comment on (peer review) the final guideline to identify any criticism of the content and to comment on clarity and applicability as well as issues relating to implementation, dissemination, ethics, regulations, or monitoring, but not to change the recommendations formulated by the GDG/ GAG. The members of the External Review Group were required to submit declarations of interest before the peer review process.
➡️Guideline Development/ Adaptation Group meetings:
GDG/ GAG meetings were organized virtually (weekly/bimonthly). Due to the extensive scope of the guideline, EPG was responsible for overseeing the adolopment process. the timetable, and objectives of each meeting. GDG/ GAG meetings were also attended by members of the methodologists. Working rules for each contributor type were outlined by the chair at the start of each meeting, covering aspects such as vocal rights, voting, and evidence to decision and recommendation formulating processes.
➡️Declarations of interests:
Prospective members of the GDG/ GAG were asked to fill in and sign the standard WHO declaration of interest and confidentiality undertaking forms. All guideline members and methodologists were also asked to fill in and sign the standard WHO declaration-of-interests.
Members of the external review group will be asked to fill in and sign the standard WHO declaration-of-interest form before the peer review process.
➡️Evidence for the guideline:
We used the GRADE system (Grading of Recommendations, Assessment, Development and Evaluation) for assigning the quality of evidence and strength of recommendations that includes the following definitions 21.
Description of the interpretation of the GRADE four levels of certainty of evidence:
Table 1. Classification of the Quality of Evidence
|
High |
We are very confident that the true effect lies close to that of the estimate of the effect. |
|
Moderate |
We are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. |
|
Low |
Our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. |
|
Very Low |
We have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of the effect. |
Table 2. Classification of the Strengths of Recommendations
|
Strong |
The desirable effects of an intervention clearly outweigh the undesirable effects (or vice versa), so most patients should receive the recommended course of action. |
|
Conditional |
There is uncertainty about the trade-offs. The clinician and patient need to discuss the patient's values and preferences, and the decision should be individualized. |
➡️Developing good practice statements:
The GDG/ GAG also developed good practice statements for this guideline, which are actionable messages relevant to the guideline questions. The justification for each good practice statement was carefully considered by the GDG/ GAG with an emphasis that they are needed. Good practice statements were developed, guided by the following GRADE criteria:
1- Message is really necessary about actual healthcare practice
2- Have large net positive consequence (relevant outcomes and downstream consequences) (GRADE EtD domains)
3- Collecting and summarizing the evidence is a poor use of time and resources
4- Include a well-documented, clear rationale connecting indirect evidence
5- Are clear and actionable statements.
The GDG/ GAG collectively drafted and finalized good practice statements with relevant justifications and remarks to help with their interpretation, with close support and input from the consultant and guideline methodologists.
We have used the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines (RIGHT-Ad@pt Tool) as a reporting checklist for this guideline adaptation process as recommended by the EQUATOR network.16
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing diagnosis, treatment, and prevention of neutropenia strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
· Select the target populations and evaluate the outcome.
· Identify the local resources to support the implementation.
· Set timelines.
· Distribute the tasks to the members.
· Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
➡️Guideline implementation strategies will focus on the following: -
1. For Practitioners
· Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
· Educational materials: printed or electronic information (software).
· Web-based education: computer-based educational activities.
· A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
· Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
· Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
· Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
· Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
· Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
· Mass media campaigns.
3. For Nurses
· Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
· Educational materials: printed.
· A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
· Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
· Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
· Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
· Administrative policies and procedures.
· Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
· International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
· Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including:
1. Manual for physician for diagnosis and algorithm for management of acute malnutrition
3. Arabic Educational materials for nurses and mothers
Table 1: Important points in history and examination in a neutropenic child25
|
History Previous full blood count available Age at first infection Frequency of infections Sites of infections Recent viral infections Other constitutional symptoms Medications |
|
Family history Neutropenia Frequent or unusual infections Autoimmune disease Early infant death from infection |
|
Physical examination Weight, height, head circumference Skeletal, skin, or nail abnormalities Skin infections Mucositis/ulcers/gingivitis/dental health Perineal or perirectal lesions or fissures Upper respiratory tract infections Bruising or bleeding Hepatomegaly or splenomegaly lymphadenopathy |
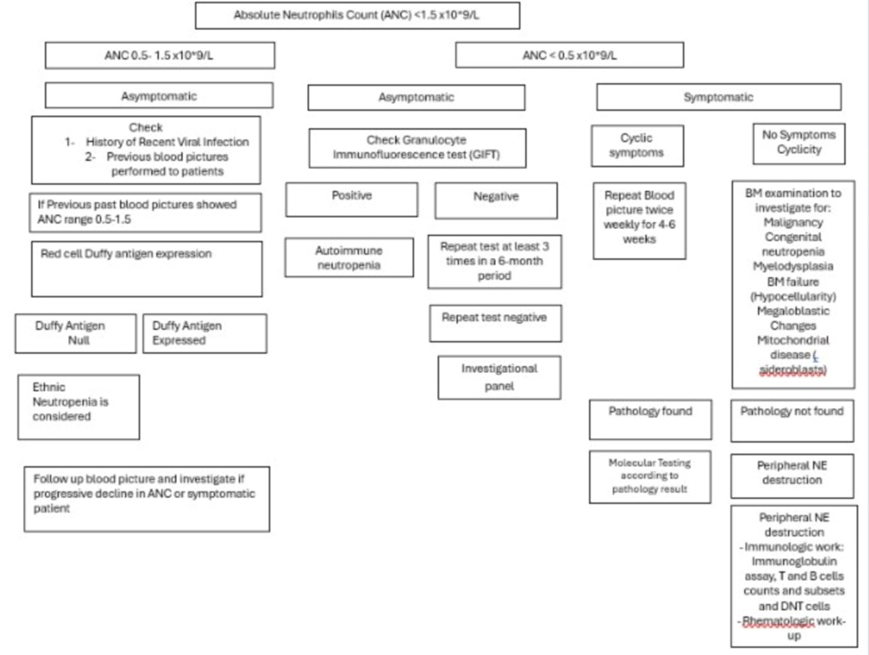
Figure 1: Proposed diagnostic algorithm for congenital neutropenia.
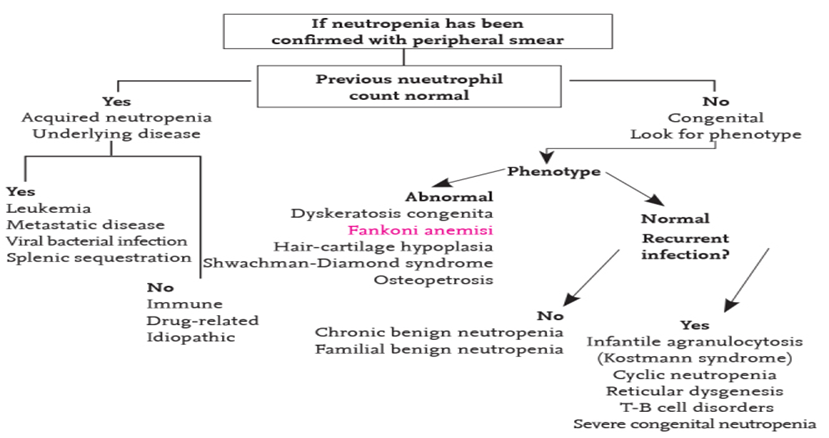
Figure 2: Approach to childhood neutropenia26
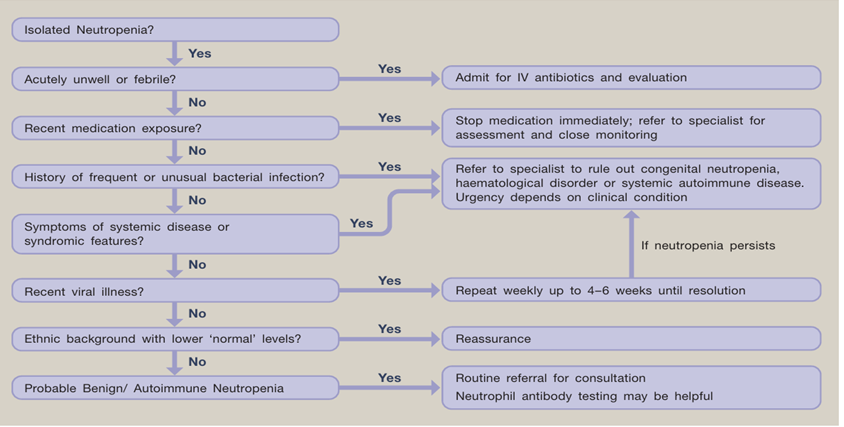
Figure 3: Initial investigations and management in neutropenia25
- Limitations and suggestions for further research needs
Future research recommendations for the management of neutropenia in children in the Egyptian context could include:
· RCT and meta-analysis on the management of primary neutropenia.
· Future research is needed to differentiate between primary and secondary neutropenia and how the management should differ based on the different etiopathogenesis of the different subtypes.
These recommendations aim to address specific challenges and characteristics of the Egyptian context, potentially leading to more effective prevention and management strategies for neutropenia in children.
➡️Challenges
· Scarce evidence-based guidelines on primary neutropenia.
· No data about the normal ranges of neutrophils in the Egyptian population.
· Unclear consensus on the different types of primary neutropenia and their management.
Strengthen the evidence will form the base of the next update of this guideline by generating GRADE summary of finding tables, evidence profiles, and EtD frameworks.
- Monitoring and evaluating the impact of the guideline.
The following are three performance measures or indicators for implementing this adapted CPG for neutropenia in children:
1. Adherence to neutropenia Guidelines
· Numerator: Number of children with neutropenia who received treatment as per guideline recommendations.
· Denominator: Total number of children diagnosed with neutropenia
· Data Source: Hospital or clinic patient records.
2. Duration of Hospital Stay
· Numerator: Total number of hospital stay days for children with neutropenia Denominator: Total number of children admitted with neutropenia
· Data Source: Hospital admission and discharge records.
3. Rate of Readmission
· Numerator: Number of children readmitted with symptoms of neutropenia within a certain period (e.g., 30 days) after discharge.
· Denominator: Total number of children initially admitted with neutropenia
· Data Source: Hospital readmission records.
These key performance indicators are designed to measure the effectiveness and adherence to the guidelines, the efficiency of the treatment in terms of resource utilization (hospital stay), and the success of the treatment in preventing further complications (readmissions).
- Updating of the guideline
The EPG neutropenia GAG has decided to conduct the next review of this adapted CPG for updates after five years. This should be carried out in 2029 after checking for updates in the source CPGs, consultation of expert opinion on the changes needed for updating according to the newest evidence and recommendations published in this area, and the clinical audit and feedback from implementation efforts in the aforementioned local healthcare settings except if any breakthrough evidence-based recommendations are published before that date. The process will be guided by the Checklist for the Reporting of Updated Guidelines (CheckUp) Tool that is freely provided by the AGREE Enterprise and by the Reporting Items for Practice Guidelines in Healthcare (RIGHT) extension for adapted guidelines RIGHT-Ad@pt Checklist.
- References
1. Dinauer MC, Newburger PE, Borregaard N. Phagocyte System and Disorders of Granulopoiesis and Granulocyte Function. In: Orkin SH, Nathan DG, Ginsburg D, Look AT, Fisher DE, Lux S, eds. Nathan and Oski's Hematology and Oncology of Infancy and Childhood E-Book. Saunders; 2014.
2. Lyall EG, Lucas GF, Eden OB. Autoimmune neutropenia of infancy. Journal of Clinical Pathology. 1992;45(5):431. doi:10.1136/jcp.45.5.431
3. Jonsson OG, Buchanan GR. Chronic neutropenia during childhood. A 13-year experience in a single institution. Am J Dis Child. Feb 1991;145(2):232-5. doi:10.1001/archpedi.1991.02160020126032
4. Walkovich K, Connelly JA. Chapter 11 - Disorders of white blood cells. In: Fish JD, Lipton JM, Lanzkowsky P, eds. Lanzkowsky's Manual of Pediatric Hematology and Oncology (Seventh Edition). Academic Press; 2022:207-235.
5. Lichtman MA. Classification and Clinical Manifestations of Neutrophil Disorders. In: Kaushansky K, Lichtman MA, Prchal JT, et al, eds. Williams Hematology, 9e. McGraw-Hill Education; 2015.
6. Lalezari P, Khorshidi M, Petrosova M. Autoimmune neutropenia of infancy. J Pediatr. Nov 1986;109(5):764-9. doi:10.1016/s0022-3476(86)80690-4
7. Wang LY, Wang CL, Chu CC, et al. Primary autoimmune neutropenia in children in Taiwan. Transfusion. May 2009;49(5):1003-6. doi:10.1111/j.1537-2995.2008.02084.x
8. Dale DC. How I diagnose and treat neutropenia. Curr Opin Hematol. Jan 2016;23(1):1-4. doi:10.1097/moh.0000000000000208
9. Hsieh MM, Everhart JE, Byrd-Holt DD, Tisdale JF, Rodgers GP. Prevalence of neutropenia in the U.S. population: age, sex, smoking status, and ethnic differences. Ann Intern Med. Apr 3 2007;146(7):486-92. doi:10.7326/0003-4819-146-7-200704030-00004
10. Segel GB, Halterman JS. Neutropenia in pediatric practice. Pediatr Rev. Jan 2008;29(1):12-23; quiz 24. doi:10.1542/pir.29-1-12
11. Tan S, Kermasson L, Hilcenko C, et al. Somatic genetic rescue of a germline ribosome assembly defect. Nature Communications. 2021/08/19 2021;12(1):5044. doi:10.1038/s41467-021-24999-5
12. Papadaki HA, Mavroudi I, Almeida A, et al. Congenital and Acquired Chronic Neutropenias: Challenges, Perspectives and Implementation of the EuNet-INNOCHRON Action. Hemasphere. Jun 2020;4(3):e406. doi:10.1097/hs9.0000000000000406
13. AGREE Tools. The AGREE Research Trust. 2024. www.agreetrust.org
14. Brouwers MC, Kerkvliet K, Spithoff K. The AGREE Reporting Checklist: a tool to improve reporting of clinical practice guidelines. Bmj. Mar 8 2016;352:i1152. doi:10.1136/bmj.i1152
15. Brouwers MC, Kho ME, Browman GP, et al. AGREE II: advancing guideline development, reporting and evaluation in health care. J Clin Epidemiol. Dec 2010;63(12):1308-11. doi:10.1016/j.jclinepi.2010.07.001
16. Song Y, Alonso-Coello P, Ballesteros M, et al. A Reporting Tool for Adapted Guidelines in Health Care: The RIGHT-Ad@pt Checklist. Ann Intern Med. May 2022;175(5):710-719. doi:10.7326/m21-4352
17. Abdel Baky A, Omar TEI, Amer YS, the Egyptian Pediatric Clinical Practice Guidelines C. Adapting global evidence-based practice guidelines to the Egyptian healthcare context: the Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) initiative. Bulletin of the National Research Centre. 2023/06/13 2023;47(1):88. doi:10.1186/s42269-023-01059-0
18. Alshehri A, Almazrou S, Amer Y. Methodological frameworks for adapting global practice guidelines to national context in the Eastern Mediterranean Region. East Mediterr Health J. Jul 31 2023;29(7):540-553. doi:10.26719/emhj.23.070
19. Amer YS, Elzalabany MM, Omar TI, Ibrahim AG, Dowidar NL. The 'Adapted ADAPTE': an approach to improve utilization of the ADAPTE guideline adaptation resource toolkit in the Alexandria Center for Evidence-Based Clinical Practice Guidelines. J Eval Clin Pract. Dec 2015;21(6):1095-106. doi:10.1111/jep.12479
20. Klugar M, Lotfi T, Darzi AJ, et al. GRADE guidance 39: using GRADE-ADOLOPMENT to adopt, adapt or create contextualized recommendations from source guidelines and evidence syntheses. J Clin Epidemiol. Aug 6 2024;174:111494. doi:10.1016/j.jclinepi.2024.111494
21. Schünemann H., Brożek J., Guyatt G., Oxman A., editors. GRADE handbook for grading quality of evidence and strength of recommendations. . guidelinedevelopment.org/handbook. ; Updated October 2013.
22. Fioredda F, Calvillo M, Bonanomi S, et al. Congenital and acquired neutropenia consensus guidelines on diagnosis from the Neutropenia Committee of the Marrow Failure Syndrome Group of the AIEOP (Associazione Italiana Emato-Oncologia Pediatrica). Pediatr Blood Cancer. Jul 15 2011;57(1):10-7. doi:10.1002/pbc.23108
23. Martínez Campos L, Pérez-Albert P, Ferres Ramis L, et al. Consensus document on the management of febrile neutropenia in paediatric haematology and oncology patients of the Spanish Society of Pediatric Infectious Diseases (SEIP) and the Spanish Society of Pediatric Hematology and Oncology (SEHOP). An Pediatr (Engl Ed). Jun 2023;98(6):446-459. doi:10.1016/j.anpede.2023.03.010
24. Patel P, Robinson PD, Baggott C, et al. Clinical practice guideline for the prevention of oral and oropharyngeal mucositis in pediatric cancer and hematopoietic stem cell transplant patients: 2021 update. Eur J Cancer. Sep 2021;154:92-101. doi:10.1016/j.ejca.2021.05.013
25. Thomas AE, Simpson LA. A step-by-step approach to paediatric neutropenia. Paediatrics and Child Health. 2017/11/01/ 2017;27(11):511-516. doi:https://doi.org/10.1016/j.paed.2017.07.006
26. Celkan T, Koç BŞ. Approach to the patient with neutropenia in childhood. Turk pediatri arsivi. 2015;50(3):136-144. doi:10.5152/turkpediatriars.2015.2295 Accessed 2015/09//. http://europepmc.org/abstract/MED/26568688
https://europepmc.org/articles/pmc4629920?pdf=render
https://doi.org/10.5152/TurkPediatriArs.2015.2295
https://europepmc.org/articles/PMC4629920
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Prof. Galila Mohamed Mokhtar |
Paediatrics Department, Paediatric Haematology/ Oncology & BMT Unit Faculty of Medicine, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Amira Adly |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Dina Ezzat |
Professor of Paediatrics and Paediatric Haematology, Beni-Suef University, Egypt |
None |
Not Applicable |
|
Prof. Gehan Lotfy |
Professor of Paediatrics , Minia University, Egypt |
None |
Not Applicable |
|
Prof. Hanafy Hafez |
Professor of Pediatric Oncology, National Cancer Institute, Cairo University, Egypt |
None |
Not Applicable |
|
Prof. Hoda Hassab |
Professor of Paediatrics and Paediatrics Haematology, Alexandria University, Egypt |
None |
Not Applicable |
|
Prof. Ilham Youssry |
Professor of Paediatrics and Head of the Paediatric Haematology & BMT unit, Cairo University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Iman Ragab |
Professor of Paediatrics, Paediatric Haematology/ Oncology & BMT Unit, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Laila Sherief |
Professor of Paediatrics and Paediatrics Haematology & Oncology, Zagazig University, Egypt |
None |
Not Applicable |
|
Prof. Marwa Zakaria |
Associate Professor of Paediatrics and Pediatric Hematology & Oncology, Zagazig University, Egypt |
None |
Not Applicable |
|
Prof. Mervat Hesham |
Professor of Pediatrics and Pediatric Hematology & Oncology, Zagazig University, Egypt |
None |
Not Applicable |
|
Dr. Naglaa Shaheen |
Consultant of Pediatric Hematology, Health Insurance Organization |
None |
Not Applicable |
|
Dr. Nouran Yousef |
Lecturer of Paediatrics, Paediatric Diabetes, Endocrinology & Metabolism unit, Ain Shams University, AFCM, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Rasha Abdel-Raouf Afifi |
Professor of Paediatrics and Paediatric Haematology, Cairo University, Cairo, Egypt |
None |
Not Applicable |
|
Dr. Sara Makkeyah |
Associate Professor of Paediatrics, Paediatric Haematology / Oncology Unit & BMT, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Prof. Sonia Adolf |
Professor of Paediatrics and Paediatric Haematology, National Research Center, Cairo, Egypt |
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Nanies Soliman |
Associate Professor of Pediatrics Ain Shams University, Egypt |
None |
Not Applicable |
|
Dr. Ranin Soliman
|
1. Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. 2. Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. 3. Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
None |
Not applicable |
|
Dr. Heba Hussein |
Associate Professor of Oral Medicine, Oral Diagnosis, and Periodontology Department, Faculty of Dentistry, Cairo University, Cairo, Egypt |
None |
Not Applicable |
|
Dr. Lamis Mohsen Elsholkamy |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Ahmad Yousef |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
Prof. Nourmin Kaddah |
Professor of Pediatrics & Pediatric hematology Cairo University, Egypt |
None |
Not Applicable |
|
Prof. Mohamed Ramadan El-Shanshoury |
Professor of Pediatrics Tanta University, Egypt |
None |
Not Applicable |
|
Prof. Mohamed Tarif Hamza |
Professor of clinical pathology, Ain Shams University, Egypt |
None |
Not Applicable |
|
External Reviewer for methodology |
|||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia
|
|
|
|
International Peer Reviewers |
|
|
|
|
|
|
None |
Not Applicable |
Web annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for " Guideline for the Management of Neutropenia in Pediatric Age Groups are: neutropenia, pediatric, guidelines…………………………………………………………………………………Annex Table 2. Results of the AGREE II assessment of the four source guidelines for neutropenia guidelines
|
AGREE II/ CPGs |
ASCO 2023 |
EHA-EuNet 2023 |
IDSA 2020 |
ASCO 2018 |
|
Domain 1 (Scope) |
80% |
78% |
98% |
100% |
|
Domain 2 (Stakeholder) |
72% |
70% |
83% |
83.3% |
|
Domain 3 (Rigour) |
77% |
60% |
95% |
84.9% |
|
Domain 4 (Clarity) |
87% |
80% |
95% |
97.2% |
|
Domain 5 (Applicability) |
88% |
57% |
47% |
68.75% |
|
Domain 6 (Independence) |
98% |
100% |
83% |
83.3% |
|
Overall assessment |
82% |
67% |
83% |
88.4% |
|
Recommend for use (Overall assessment) |
YES |
YES |
YES |
YES |
Annex Table 3. Annex Nurses and Parents Educational Guide in Arabic
Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|
