Advanced/Metastatic CRC
| Site: | EHC | Egyptian Health Council |
| Course: | Oncology and Hematological Oncology Guidelines |
| Book: | Advanced/Metastatic CRC |
| Printed by: | Guest user |
| Date: | Wednesday, 6 May 2026, 12:53 AM |
Description
"last update: 7 April 2026" Download Guideline
- Executive Summary
|
Strength of the recommendation |
|
|
Complete work-up should be carried out to achieve an accurate histological diagnosis of the primary tumor, assess the baseline characteristics of the patient and determine the extent of the disease. |
Strong |
|
Besides a comprehensive physical examination, request blood tests including complete blood count and chemistry profile. |
Strong |
|
In addition, serum levels of CEA should be evaluated and monitored during the follow-up period to help evaluate response to treatment. |
Strong |
|
CT of the thoracic, abdominal and pelvic cavities with i.v. contrast administration is the preferred radiological method for the evaluation of the extent of CRC. |
Strong |
|
A Triphasic CT or Dynamic liver MRI is recommended to characterise non-typical liver lesions on CT scans or when liver metastases seem resectable or potentially resectable. |
Strong |
|
FDG-PET/CT scan can be useful, particularly in patients with increased tumour markers without evidence of metastatic disease, or to define the extent of metastatic disease on potentially resectable metastases. |
Conditional |
|
FDG-PET/CT is NOT USEFULL in mucinous and sigent ring differentiation. |
Strong |
|
Testing for KRAS, NRAS exon 2, 3 and 4 and BRAF mutations is recommended in all patients at the time of mCRC diagnosis |
Strong |
|
RAS testing is mandatory before treatment with antiEGFR monoclonal antibodies and can be carried out on either the primary tumour or other metastatic sites. |
Strong |
|
BRAF mutation status should be assessed simultaneously with the evaluation of RAS, for prognostic assessment and for the option of treatment with antiEGFR mAbs. |
Strong |
|
Identification of HER2 amplification by IHC or FISH is recommended in RAS-wt patients to detect those who may benefit from HER2 blockade. |
Strong |
|
2.TREATMENT OF POTENTIALLY RESECTABLE ADVANCED AND METASTATIC DISEASE |
|
|
In patients with resectable metastases, favorable prognostic criteria and a feasible surgical approach, preoperative / post-operative systemic treatment may not be needed. |
Conditional |
|
In patients with resectable metastases, the use of perioperative oxaliplatin-based chemotherapy is recommended where the prognostic situation is unclear. |
Strong |
|
Anti-EGFR monoclonal antibodies in left-sided RAS-wt patients should be used as conversion therapy, when complete resection is the aim. |
Strong |
|
In patients with right-sided and RAS-mutant disease, FOLFOXIRI-bevacizumab and, to a lesser extent, a cytotoxic doublet-bevacizumab should be considered the best choice depending on patients’ ability to tolerate triplet chemotherapy. |
Strong |
|
Conditional |
|
|
In case of peritoneal metastasis only, complete cytoreductive surgery may be carried out. |
Conditional |
|
Local treatment can be used primarily as a metastasis-directed treatment to halt local failure , further dissemination, and/or following systemic therapy as a consolidation treatment, to delay or pause further treatment. |
Conditional |
|
Frequent radiological reevaluations of the potential applicability of surgery or other local treatment techniques should be carried out, generally every 8-12 weeks. |
Strong |
|
Local ablation treatment |
|
|
Conditional |
|
|
Thermal Ablation is a valid treatment option for recurrent disease after surgical resection for small CRLMs. |
Strong |
|
In patients with lung-only metastases or OMD including lung lesions, thermal ablation may be considered along with resection, according to tumor size, number, location, the extent of lung parenchyma loss, or other comorbidities. |
Conditional |
|
SBRT may be considered as a local treatment option. |
Conditional |
|
3.MANAGEMENT OF ADVANCED AND METASTATIC DISEASE WITHOUT POTENTIAL CONVERSION |
|
|
Determining the RAS mutational status on a tumour biopsy is mandatory to guide the best treatment decision. |
Strong |
|
Delivering a biological therapy in combination with chemotherapy in the first-line setting is recommended, unless contraindicated. |
Strong |
|
First-line treatment will consist of doublet of chemotherapy (FOLFOX, FOLFIRI, CAPOX) combined with an anti-VEGF or anti-EGFR mAbs unless contraindicated.. |
Strong |
|
In RAS-wt and BRAF-wt left-sided tumours, doublet chemotherapy plus an anti-EGFR mAbs is the preferred option. |
Strong |
|
In RAS-wt right-sided tumours, chemotherapy + bevacizumab is the recommended treatment unless contraindicated |
Strong |
|
Anti-EGFR mAbs should be combined with the doublets FOLFOX or FOLFIRI. |
Strong |
|
Bevacizumab should be combined with single fluoropyrimidines, irinotecan or oxaliplatin-based doublet of ChT (FOLFOX, CAPOX, FOLFIRI) or triplets (FOLOXIRI). |
Strong |
|
A triplet with FOLFOXIRI plus bevacizumab could also be an option for selective patients with good PS and without comorbidities. |
Conditional |
|
Triplets including FOLFOXIRI should not be used in patients >75 years old, with PS2 or in patients with significant comorbidities.
|
Strong |
|
In patients with comorbidities, older age or with metastatic disease not amenable to a curative treatment strategy and no significant disease-related symptoms, monotherapy with a fluoropyrimidine bevacizumab is recommended |
Strong |
|
In frail or elderly patients unable to tolerate chemotherapy, whose tumours are left-sided and RAS-wt, monotherapy with anti-EGFR mAbs can be considered. |
Conditional |
|
Maintenance and subsequent therapy |
|
|
After first-line therapy with ChT based on oxaliplatin and bevacizumab, maintenance therapy with a fluoropyrimidine is recommended in nonprogressive patients after at least 6 months of treatment. |
Strong |
|
Reintroduction of an initial successful induction therapy may be done after progressive disease while on maintenance therapy. |
Conditional |
|
After failure of first-line oxaliplatin-based therapy, second-line treatment with irinotecan-based or monotherapy is recommended unless contraindicated |
Strong |
|
After failure of first line irinotecan-based therapy , second line treatment with oxaliplatin-based therapy (FOLFOX or CAPOX) is recommended unless contraindicated . |
Strong |
|
In RAS-wt patients not previously treated with an anti- EGFR moAb, treatment with chemotherapy (FOLFIRI or irinotecan) and cetuximab or panitumumab is recommended for left-sided colon tumours. |
Strong |
|
In patients previously treated with chemotherapy alone, a combination of doublet chemotherapy + bevacizumab or anti EGFR (Left side) is recommended. |
Strong |
|
Bevacizumab can be combined with a fluoropyrimidine doublet with oxaliplatin or irinotecan, depending on the first-line chemotherapy backbone delivered. |
Conditional |
|
Reintroduction of the initial induction therapy can be considered after second-line therapy, as long as the patient did not progress during the induction course of first-line chemotherapy. Treatment should be based upon previous treatment lines, AEs, and PS. |
Conditional |
|
Follow up and monitoring |
|
|
History and physical examination and CEA level determination are recommended every 3-6 months for 3 years and every 6-12 months at years 4 and 5 after surgery. |
Strong |
|
Colonoscopy must be carried out at year 1 and every 3-5 years thereafter, looking for metachronous adenomas and cancers. |
Strong |
|
CT scan of chest , abdomen and pelvis every 6-12 months for the first 3 years is recommended in patients who are at higher risk of recurrence according to the TNM classification. |
Strong |
|
Long-term follow-up, rehabilitation and survivorship care programmes should be implemented, aiming at detection of recurrent or new cancers, assessment and management of late and psychosocial effects and implementation of healocal treatmenth promotion measures. |
Strong |
- Recommendations
1. DIAGNOSTIC WORK UP FOR ADVANCED/METASTATIC DISEASE
· A complete work-up should be carried out to achieve an accurate histological diagnosis of the primary tumor, assess the baseline characteristics of the patient and determine the extent of the disease
Strong recommendation, High grade evidence (5)
· Besides a comprehensive physical examination, request blood tests including complete blood count and chemistry profile.
Strong recommendation, High grade evidence (5).
· In addition, serum levels of CEA should be evaluated and monitored during the follow-up period to help evaluate response to treatment.
Strong recommendation, Low grade evidence (6-8)
· CT of the thoracic, abdominal and pelvic cavities with i.v. contrast administration is the preferred radiological method for the evaluation of the extent of CRC.
Strong recommendation, High grade evidence (9,10)
· .A Triphasic CT or Dynamic liver MRI is recommended to characterise non-typical liver lesions on CT scans or when liver metastases seem resectable or potentially resectable.
Strong recommendation, High grade evidence ( 11).
· FDG-PET/CT scan can be useful, particularly in patients with increased tumour markers without evidence of metastatic disease, or to define the extent of metastatic disease on potentially resectable metastases.
Conditional recommendation, High grade evidence (8,12).
· FDG-PET/CT is NOT USEFULL in mucinous and sigent ring differentiation
Strong recommendation, High grade evidence (8,12).
· .Testing for KRAS, NRAS exon 2, 3 and 4 and BRAF mutations is recommended in all patients at the time of mCRC diagnosis.
Strong recommendation, High grade evidence (13-15).
· RAS testing is mandatory before treatment with antiEGFR monoclonal antibodies and can be carried out on either the primary tumour or other metastatic sites.
Strong recommendation, High grade evidence (13-15).
· BRAF mutation status should be assessed simultaneously with the evaluation of RAS, for prognostic assessment and for the option of treatment with antiEGFR mAbs.
Strong recommendation, Low grade evidence (16,17).
· Identification of HER2 amplification by IHC or FISH is recommended in RAS-wt patients to detect those who may benefit from HER2 blockade
Strong recommendation, Low grade evidence (18).
1. TREATMENT OF POTENTIALLY RESECTABLE ADVANCED AND METASTATIC DISEASE
- In patients with resectable metastases , favorable prognostic criteria and a feasible surgical approach, preoperative / post-operative systemic treatment may not be needed.
Conditional recommendation, low grade evidence (19).
- In patients with resectable metastases, the use of perioperative oxaliplatin-based chemotherapy is recommended where the prognostic situation is unclear.
Strong recommendation, high grade evidence (20).
- Anti-EGFR monoclonal antibodies in left-sided RAS-wt patients should be used as conversion therapy, when complete resection is the aim.
Strong recommendation, high grade evidence(21).
- In patients with right-sided and RAS-mutant disease, FOLFOXIRI-bevacizumab and, to a lesser extent, a cytotoxic doublet-bevacizumab should be considered the best choice depending on patients’ ability to tolerate triplet chemotherapy.
Strong recommendation, high grade evidence (21).
- Patients unresponsive to first-line chemotherapy should not be denied resection or ablation of metastases since the outcome of resected patients after second-line treatment could be also favorable.
Conditional recommendation, low grade evidence (22).
- In case of peritoneal metastasis only, complete cytoreductive surgery may be carried out.
Conditional recommendation, high grade evidence (23).
Intent and choice of local treatment
- Local treatment can be used primarily as a metastasis-directed treatment to halt local failure , further dissemination, and/or following systemic therapy as a consolidation treatment, to delay or pause further treatment
Conditional recommendation, low grade evidence (24,25).
- Frequent radiological reevaluations of the potential applicability of surgery or other local treatment techniques should be carried out, generally every 8-12 weeks.
Strong recommendation, very low grade evidence(26).
Local ablation treatment
- In patients with unresectable CRLMs only, or OMD in the liver, Thermal Ablation can be considered for small metastases.
Conditional recommendation, low grade evidence (27-29).
- Thermal Ablation is a valid treatment option for recurrent disease after surgical resection for small CRLMs
Strong recommendation, high grade evidence (30).
- In patients with lung-only metastases or OMD including lung lesions, thermal ablation may be considered along with resection, according to tumor size, number, location, the extent of lung parenchyma loss, or other comorbidities.
Conditional recommendation, low grade evidence (27-29).
- SBRT may be considered as a local treatment option
Conditional recommendation, low grade evidence (31).
2. MANAGEMENT OF ADVANCED AND METASTATIC DISEASE WITHOUT POTENTIAL CONVERSION
First-line therapy
l Determining the RAS mutational status on a tumour biopsy is mandatory to guide the best treatment decision.
Strong recommendation, high grade evidence (32).
l Delivering a biological therapy in combination with chemotherapy in the first-line setting is recommended, unless contraindicated.
Strong recommendation, high grade evidence (33).
- First-line treatment will consist of doublet of chemotherapy (FOLFOX, FOLFIRI, CAPOX) combined with an anti-VEGF or anti-EGFR mAbs unless contraindicated .
Strong recommendation, high grade evidence (34).
- In RAS-wt and BRAF-wt left-sided tumors, doublet chemotherapy plus an anti-EGFR mAbs is the preferred option.
Strong recommendation, high grade evidence (34).
- In RAS-wt right-sided tumors, chemotherapy + bevacizumab is the recommended treatment unless contraindicated ,
Strong recommendation, high grade evidence (35).
- Anti-EGFR mAbs should be combined with the doublets FOLFOX or FOLFIRI.
Strong recommendation, high grade evidence (33,36).
- Bevacizumab should be combined with single fluoropyrimidines, irinotecan or oxaliplatin-based doublet of ChT (FOLFOX, CAPOX, FOLFIRI) or triplets (FOLOXIRI)
Strong recommendation, high grade evidence (37-39).
- A triplet with FOLFOXIRI plus bevacizumab could also be an option for selective patients with good PS and without comorbidities
Conditional recommendation, high grade evidence (37-39).
- Triplets including FOLFOXIRI should not be used in patients >75 years old, with PS2 or in patients with significant comorbidities.
Strong recommendation, very low grade evidence (40).
- In patients with comorbidities, older age or with metastatic disease not amenable to a curative treatment strategy and no significant disease-related symptoms, monotherapy with a fluoropyrimidine bevacizumab is recommended
Strong recommendation, high grade evidence (41).
- In frail or elderly patients unable to tolerate chemotherapy, whose tumours are left-sided and RAS-wt, monotherapy with anti-EGFR mAbs can be considered
Conditional recommendation, very low grade evidence (42).
Maintenance and subsequent therapy
- After first-line therapy with ChT based on oxaliplatin and bevacizumab, maintenance therapy with a fluoropyrimidine is recommended in nonprogressive patients after at least 6 months of treatment.
Strong recommendation, high grade evidence (43).
- Reintroduction of an initial successful induction therapy may be done after progressive disease while on maintenance therapy
.Conditional recommendation, high grade evidence (37).
- After failure of first-line oxaliplatin-based therapy, second-line treatment with irinotecan-based or monotherapy is recommended unless contraindicated
Strong recommendation, high grade evidence (44, 45).
- After failure of first-line Irinotican-based therapy, second-line treatment with Oxaloplatin-based therapy (FOLFOX or CAPOX) is recommended unless contraindicated
Strong recommendation, high grade evidence (22).
- In RAS-wt patients not previously treated with an anti- EGFR moAb, treatment with chemotherapy (FOLFIRI or irinotecan) and cetuximab or panitumumab is recommended for left-sided colon tumours.
Strong recommendation, high grade evidence (46).
- In patients previously treated with chemotherapy alone, a combination of doublet chemotherapy + bevacizumab or anti EGFR (Left side) is recommended.
Strong recommendation, high grade evidence (46).
- Bevacizumab can be combined with a fluoropyrimidine doublet with oxaliplatin or irinotecan, depending on the first-line chemotherapy backbone delivered.
Conditional recommendation, high grade evidence (46).
- Reintroduction of the initial induction therapy can be considered after second-line therapy, as long as the patient did not progress during the induction course of first-line chemotherapy. Treatment should be based upon previous treatment lines, AEs, and PS.
Conditional recommendation, low grade evidence.
➡️ Follow up and monitoring (47-53)
· History and physical examination and CEA level determination are recommended every 3-6 months for 3 years and every 6-12 months at years 4 and 5 after surgery.
Strong recommendation, high grade evidence.
· Colonoscopy must be carried out at year 1 and every 3-5 years thereafter, looking for metachronous adenomas and cancers.
Strong recommendation, moderate grade evidence.
· CT scan of chest , abdomen and pelvis every 6-12 months for the first 3 years are recommended in patients who are at higher risk of recurrence according to the TNM classification.
Stroncommendation, high grade evidence.
· Long-term follow-up, rehabilitation and survivorship care programmes should be implemented, aiming at detection of recurrent or new cancers, assessment and management of late and psychosocial effects and implementation of healocal treatmenth promotion measures.
Strong recommendation, low grade evidence.
- Acknowledgments
- We would like to acknowledge the Oncology Committee of the Egyptian Health Council (EHC) Guidelines for adapting these Guidelines.
- Chair of the Oncology Committee of Egyptian Health Council Guidelines : Prof Hussein Khaled.
- The Oncology Committee Members: Emad Hamada, Samir Shehata, Hesham Elghazaly, Hesham Tawfik, Fouad Abuotaleb, Ebtesam Saad Eldin, Ihab Khalil, Khaled Abdelkarim, Lobna EZZ Elarab, Mary Gamal, Mohamed Abdel Mooti, Mohamed Gamil, Nervana Hussein, Ola Khorshid, Omar Sherif Omar, Rasha Fahmi, Rasha Shalocal treatmentout, Yousri Wasef & Yousri Rostom.
- Chair of the GIT Cancer Scientific Committee: Prof Yousri Rostom
- The gastric cancer Scientific Group Members: (Alphabitical order): Ahmed EL Kassed, Fouad Aboutaleb, Khaled Abdelkarim, Manal EL Mahdy, Omar S. Omar, and Yousri Wasef.
- Abbreviations
5FU fluorouracil
AEs adverse events
CEA carcinoembryonic antigen
ChT chemotherapy
CT computed tomography
CRC colo-rectal cancer
CRLMs colo-rectal liver metastases
EGFR Epidermal growth factor receptor
EMV extramural venous invasion
FDG-PET Fluorodeoxyglucose Positron Emission Tomography
FISH Fluorescence In Situ Hybridization
IHC Immunohistochemistry
mCRC metastatic colo-rectal cancer
MDT mulocal treatmentidisciplinary team
mAb monclonal antibody
MRI magnetic resonance imaging
OMD oligometastatic disease
PS performance status
SBRT Stereotactic Body Radiation Therapy
- Introduction
Colo-rectal cancer is the 7th most common cancer in the Egyptian population with more than 5900 newly diagnosed cases and more than 3000 deaths in 2022 (1).
- Purpose and scope
These guidelines will help to improve the quality of care for Advanced/Metastatic CRC patients via providing a uniform standard of care across the country to help in early diagnosis and treatment for Advanced/Metastatic CRC, with less aggressive treatment options and improved clinical outcomes. These guidelines cover primary diagnosis, staging, treatment and follow-up of Advanced/Metastatic CRC patients.
- Target audience
Clinicians who are involved in the care and treatment of patients with Advanced/Metastatic CRC, including medical oncologists, radiation oncologists, clinical oncologists, onco- and gastrointestinal surgeons, radiologists and pathologists.
- Methodology
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
- Selecting only evidence-based guidelines (guidelines must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence). - Selecting only national and/or international guidelines.
- Specific range of dates for publication (using Guidelines published or updated 2015 and later).
- Selecting peer reviewed publications only.
- Selecting guidelines written in English language.
- Excluding guidelines written by a single author not on behalf of an organization to be valid and comprehensive, a guideline ideally requires mulocal treatmentidisciplinary input.
- Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations.
All retrieved Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. the panel decided a cutoff point or rank the guidelines (any guideline scoring above 50% on the rigor dimension was retained)
The NCCN, ESMO, NICE guidelines are the main sources used while formulating the national guidelines for metastatic colorectal cancer (2-4).
- Evidence assessment
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available through the on the following sites:
. GRADE working group:https://www.gradeworkinggroup.org
. GRADE online training modules: http://cebgrade.mcmaster.ca/
Table 1: Quality of evidence in GRADE
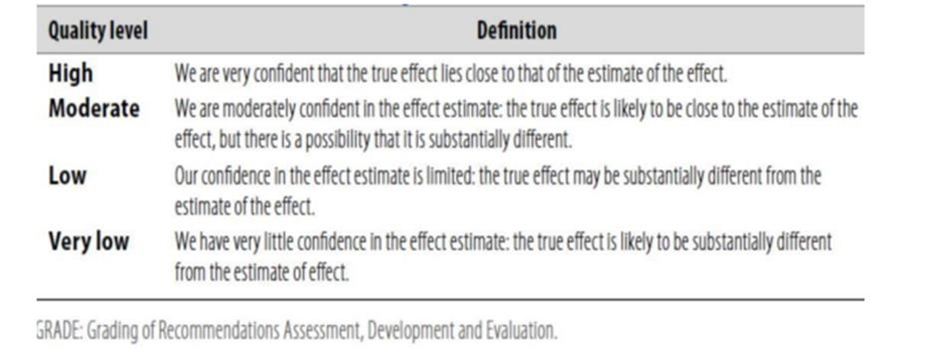
Table 2: Significance of the four levels of evidence

Table 3: Factors that determine how to upgrade or downgrade the quality of evidence.
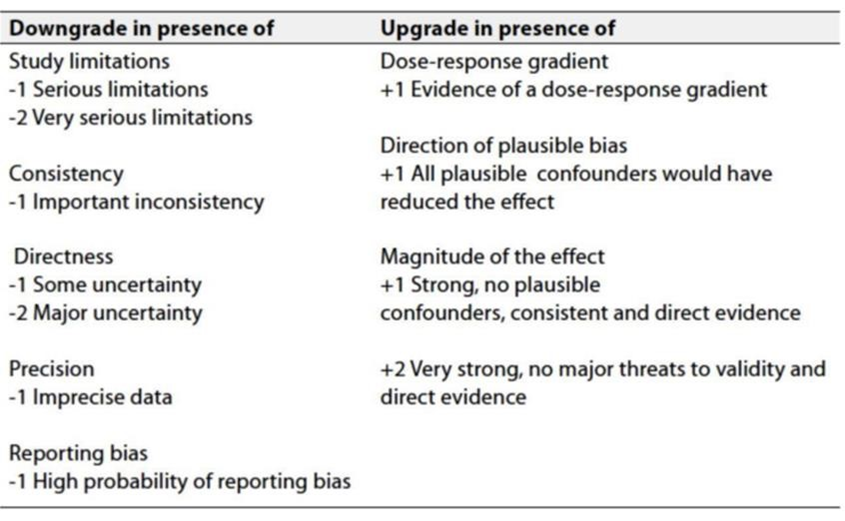
➡️ The strength of the recommendation
The strength of a recommendation communicates the importance of adherence to the recommendation:
Strong recommendations: With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
Conditional recommendations: These are made when there is greater uncertainty about the four factors above (Table 2) or if local adaptation must account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
When not to make recommendations; when there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
- Research Gaps
• Evaluation of real world data on the use on new targeted and immune-therapeutic agents in CRC in Egypt.
• Cost effective analysis of new therapeutic agents in Egypt.
• Define the molecular biologic profiles of our patients.
- Update of the guideline
• This guideline will be updated whenever there is new evidence.
- Annexes
Annex 1.
Annex 2.SYSTEMIC THERAPY FOR ADVANCED OR METASTATIC DISEASE
- mFOLFOX 6
Oxaliplatin 85 mg/m2 IV day 1z Leucovorin 400 mg/m2 IV day 1aa
5-FU 400 mg/m2 IV bolus on day 1, followed by 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) IV continuous infusion
Repeat every 2 weeks
- mFOLFOX 7
Oxaliplatin 85 mg/m2 IV day 1z Leucovorin 400 mg/m2 IV day 1aa
5-FU 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) IV continuous infusion
Repeat every 2 weeks
- FOLFOX + bevacizumab
Bevacizumab mg/kg IV, day 1 Repeat every 2 weeks
- FOLFOX + panitumumab (KRAS/NRAS/BRAF WT)
Panitumumab 6 mg/kg IV over 60 minutes, day 1 ---Repeat every 2 weeks
- FOLFOX + cetuximab (KRAS/NRAS/BRAF WT)
Cetuximab 400 mg/m2 IV over 2 hours first infusion, followed by 250 mg/m2 IV over 60 minutes weekly or Cetuximab 500 mg/m2 IV over 2 hours, day 1, every 2 weeks (preferred for every 2 weeks)
- CAPEOX8
Oxaliplatin 130 mg/m2 IV day 1z
Capecitabine 1000cc mg/m2 twice daily PO for 14 days
Repeat every 3 weeks
- CAPEOX + bevacizumab
Oxaliplatin 130 mg/m2 IV day 1z
Capecitabine 1000cc mg/m2 PO twice daily for 14 days
Bevacizumab 7.5 mg/kg IV day 1 Repeat every 3 weeks
CAPEOX + cetuximab (KRAS/NRAS/BRAF WT)
Cetuximab 400 mg/m2 IV over 2 hours first infusion, followed by 250 mg/m2 IV over 60 minutes weekly or Cetuximab 500 mg/m2 IV over 2 hours, day 1, every 2 weeks (preferred for every 2 weeks)
CAPEOX + panitumumab9-11 (KRAS/NRAS/BRAF WT)
Panitumumab 6 mg/kg IV over 60 minutes, day 1 Repeat every 2 weeks
FOLFIRI
Irinotecan 180 mg/m2 IV over 30–90 minutes, day 1
Leucovorinaa 400 mg/m2 IV infusion to match duration of irinotecan infusion,
day 1
5-FU 400 mg/m2 IV bolus day 1, followed by 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) continuous infusion
Repeat every 2 weeks
- FOLFIRI + bevacizumab
Bevacizumab 5 mg/kg IV, day 1 Repeat every 2 weeks
- FOLFIRI + cetuximab (KRAS/NRAS/BRAF WT)
Cetuximab 400 mg/m2 IV over 2 hours first infusion, followed by 250 mg/m2 IV over 60 minutes weekly or Cetuximab 500 mg/m2 IV over 2 hours, day 1, every 2 weeks (preferred
for every 2 weeks)
- FOLFIRI + panitumumab (KRAS/NRAS/BRAF WT)
Panitumumab 6 mg/kg IV over 60 minutes, day 1 Repeat every 2 weeks
- FOLFIRINOX20
Oxaliplatin 85 mg/m² IV on day 1,z leucovorin 400 mg/m² IV over 2 hours on day 1, irinotecan 165–180 mg/m² IV over 30–90 minutes on day 1, 5-FU 400 mg/m² IV push day 1, 5-FU 1200 mg/m²/day x 2 days (total 2400 mg/m² over 46 hours) continuous infusion.
Repeat every 2 weeks
- Modified FOLFIRINOX
Oxaliplatin 85 mg/m² IV on day 1,z leucovorin 400 mg/m² IV over 2 hours on day 1, irinotecan 150 mg/m² IV over 30–90 minutes on day 1, 5-FU 1200 mg/m²/day x 2 days (total 2400 mg/m² over 46 hours) continuous infusion. Repeat every 2 weeks
- FOLFIRINOX or mFOLFIRINOX + bevacizumab
Bevacizumab 5 mg/kg IV, day 1
Repeat every 2 weeks
- IROX
Oxaliplatin 85 mg/m2 IV
followed by irinotecan 200 mg/m2 over 30–90 minutes every 3 weeks
- IROX + bevacizumab
Bevacizumab 7.5 mg/kg IV on day 1 Repeat every 3 weeks
- Bolus or infusional 5-FU/leucovorin Roswell Park regimen
Leucovorin 500 mg/m2 IV over 2 hours, days 1, 8, 15, 22, 29, and 36
5-FU 500 mg/m2 IV bolus 1 hour after start of leucovorin,
days 1, 8, 15, 22, 29, and 36
Repeat every 8 weeks
- Simplified biweekly infusional 5-FU/leucovorin (sLV5FU2)
Leucovorinaa 400 mg/m2 IV over 2 hours on day 1,
followed by 5-FU bolus 400 mg/m2 followed by 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) continuous infusion Repeat every 2 weeks
Weekly Leucovorin 20 mg/m2 IV over 2 hours on day 1, 5-FU 500 mg/ m2 IV bolus injection 1 hour after the start of leucovorin. Repeat weekly27
or
5-FU 2600 mg/m2 by 24-hour infusion plus leucovorin 500 mg/m2 Repeat every week
- Bolus or infusional 5-FU + bevacizumab
Bevacizumab 5 mg/kg IV on day 1
Repeat every 2 weeks
- Capecitabine
Capecitabine 850–1250 mg/m2 PO twice daily for 14 days Repeat every 3 weeks
- Capecitabine + bevacizumab
Bevacizumab 7.5 mg/kg IV, day 1 Repeat every 3 weeks
l Irinotecan
Irinotecan 125 mg/m2 IV over 30–90 minutes, days 1 and 8 Repeat every 3 weeks
or Irinotecan 180 mg/m2 IV over 30–90 minutes, day 1 Repeat every 2 weeks
or Irinotecan 300–350 mg/m2 IV over 30–90 minutes, day 1 Repeat every 3 weeks
- Irinotecan + cetuximab (KRAS/NRAS/BRAF WT)
Cetuximab 400 mg/m2 first infusion, followed by 250 mg/m2 IV weekly32
or Cetuximab 500 mg/m2 IV over 2 hours, day 1, every 2 weeks16
(preferred for every 2 weeks)
- Irinotecan + panitumumab17,33 (KRAS/NRAS/BRAF WT)
Panitumumab 6 mg/kg IV over 60 minutes every 2 weeks
- Irinotecan + bevacizumab34,bb Irinotecan 180 mg/m2 IV, day 1
Bevacizumab 5 mg/kg IV, day 1 Repeat every 2 weeks
or
Irinotecan 300–350 mg/m2 IV, day 1
Bevacizumab 7.5 mg/kg IV, day 1 Repeat every 3 weeks
Cetuximab (KRAS/NRAS/BRAF WT)
Cetuximab 400 mg/m2 first infusion, followed by 250 mg/m2 IV weekly32
or Cetuximab 500 mg/m2 IV over 2 hours, day 1, every 2 weeks16
(preferred for every 2 weeks)
Panitumumab35 (KRAS/NRAS/BRAF WT)
Panitumumab 6 mg/kg IV over 60 minutes every 2 weeks
- Trastuzumab + pertuzumab
(HER2-amplified and RAS and BRAF WT)
Trastuzumab 8 mg/kg IV loading dose on day 1 of cycle 1, followed by 6 mg/kg IV every 21 days
Pertuzumab 840 mg IV loading dose on day 1 of cycle 1, followed by 420 mg IV every 21 days
- Trastuzumab + lapatinib
(HER2-amplified and RAS and BRAF WT)
Trastuzumab 4 mg/kg IV loading dose on day 1 of cycle 1, followed by 2 mg/kg IV weekly
Lapatinib 1000 mg PO daily
- References
1. https://gco.iarc.who.int/media/globocan/factsheets/populations/818-egypt-fact-sheet.pdf
2. https://www.esmo.org/guidelines/esmo-clinical-practice-guideline-metastatic-colorectal-cancer
3. https://www.nccn.org/professionals/physician_gls/pdf/rectal.pdf
4. https://www.nice.org.uk/guidance/conditions-and-diseases/cancer/colorectal-cancer
5. Hamilton W, Round A, Sharp D, Peters TJ. Clinical features of colorectal cancer before diagnosis: a population-based case-control study. Br J Cancer. 2005;93:399-405.
6. Konishi T, Shimada Y, Hsu M, et al. Association of preoperative and postoperative serum carcinoembryonic antigen and colon cancer outcome. JAMA Oncol. 2018;4(3):309-315.
7. Duffy MJ, van Dalen A, Haglund C, et al. Clinical utility of biochemical markers in colorectal cancer: European Group on Tumour Markers (EGTM) guidelines. Eur J Cancer. 2003;39:718-727.
8. Locker GY, Hamilton S, Harris J, et al. ASCO 2006 update of recommendations for the use of tumor markers in gastrointestinal cancer. J Clin Oncol. 2006;24:531305327.
9. Horton KM, Abrams RA, Fishman EK. Spiral CTof colon cancer: imaging features and role in management. Radiographics. 2000;20:419-430.
10. Nerad E, Lahaye MJ, Maas M, et al. Diagnostic accuracy of CT for local staging of colon cancer: a systematic review and meta-analysis. AJR Am J Roentgenol. 2016;207:984-995.
11. Sahani DV, Bajwa MA, Andrabi Y, et al. Current status of imaging and merging techniques to evaluate liver metastases from colorectal carcinoma. Ann Surg. 2014;259:861-872.
12. Niekel MC, Bipat S, Stoker J. Diagnostic imaging of colorectal liver metastases with CT, MR imaging, FDG PET and/or FDG PET/CT: a meta-analysis of prospective studies including patients who have not previously undergone treatment. Radiology. 2010;257:674-684.
13. Amado RG, Wolf M, Peeters M, et al. Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer.J Clin Oncol. 2008;26(10):1626-1634.
14. Karapetis CS, Khambata-Ford S, Jonker DJ, et al. K-ras mutations and benefit from cetuximab in advanced colorectal cancer. N Engl J Med. 2008;359(17):1757-1765.
15. Douillard JY, Siena S, Cassidy J, et al. Randomised, phase III trial of panitumumab with infusional fluorouracil, leucovorin, and oxaliplatin (FOLFOX4) versus FOLFOX4 alone as first-line treatment in patients with previously untreated metastatic colorectal cancer: the PRIME study. J Clin Oncol. 2010;28(31):4697-4705.
16. Kopetz S, Grothey A, Yaeger R, et al. Encorafenib, binimetinib, and cetuximab in BRAF V600E-mutated colorectal cancer. N Engl J Med.2019;381(17):1632-1643.
17. Schirripa M, Biason P, Lonardi S, et al. Class 1, 2, and 3 BRAF-mutated metastatic colorectal cancer: a detailed clinical, pathologic, and molecular characterization. Clin Cancer Res. 2019;25(13):3954-3961.
18. Sartore-Bianchi A, Trusolino L, Martino C, et al. Dual-targeted therapy with trastuzumab and lapatinib in treatment-refractory, KRAS codon 12/13 wild-type, HER2-positive metastatic colorectal cancer (HERACLES): a proof-of-concept, multicentre, open-label, phase 2 trial. Lancet Oncol. 2016;17(6):738-746.
19. Primrose J, Falk S, Finch-Jones M, et al. Systemic chemotherapy with or without cetuximab in patients with resectable colorectal liver metastasis: the New EPOC randomised controlled trial. Lancet Oncol.2014;15(6):601-611.
20. Primrose J, Falk S, Finch-Jones M, et al. Systemic chemotherapy with or without cetuximab in patients with resectable colorectal liver metastasis: the New EPOC randomised controlled trial. Lancet Oncol.2014;15(6):601-611.
21. Arnold D, Lueza B, Douillard JY, et al. Prognostic and predictive value of primary tumour side in patients with RAS wild-type metastatic colorectal cancer treated with chemotherapy and EGFR directed antibodies in six randomised trials. Ann Oncol. 2017;28(8):1713-1729.
23. Cotte E, Passot G, Mohamed F, et al. Management of peritonealcarcinomatosis from colorectal cancer: current state of practice.
24. Ruers T, Van Coevorden F, Punt CJA, et al. Local treatment of unresectable colorectal liver metastases: results of a randomised phase II trial. J Natl Cancer Inst. 2017;109(9):djx015.
25. Schlijper RCJ, Grutters JPC, Houben R, et al. What to choose as radical local treatment for lung metastases from colo-rectal cancer: surgery or radiofrequency ablation? Cancer Treat Rev. 2014;40(1):60-67.
26. Argilés G, Tabernero J, Labianca R, et al. Localised colon cancer: ESMOClinical Practice Guidelines for diagnosis, treatment and follow-up.Ann Oncol.2020;31(10):1291-1305.
27. Fonck M, Perez JT, Catena V, et al. Pulmonary thermal ablation enables long chemotherapy-free survival in metastatic colorectal cancer patients. Cardiovasc Intervent Radiol. 2018;41(11):1727-1734.
28. Tanis E, Nordlinger B, Mauer M, et al. Local recurrence rates after radiofrequency ablation or resection of colorectal liver metastases. Analysis of the European Organisation for Research and Treatment of Cancer #40004 and #40983. Eur J Cancer. 2014;50(5):912-919.
29. Shady W, Petre EN, Gonen M, et al. Percutaneous radiofrequency ablation of colorectal cancer liver metastases: factors affecting outcomes– a 10-year experience at a single center. Radiology.2016;278(2):601-611.
30. Sofocleous CT, Petre EN, Gonen M, et al. CT-guided radiofrequency ablation as a salvage treatment of colorectal cancer hepatic metastases developing after epatectomy. J Vasc Interv Radiol. 2011;22(6):755-761.
31. Binkley MS, Trakul N, Jacobs LR, et al. Colorectal histology is associated with an increased risk of local failure in lung metastases treated with stereotactic ablative radiation therapy. Int J Radiat Oncol Biol Phys. 2015;92(5):1044-1052.
32. Amado RG, Wolf M, Peeters M, et al. Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. J Clin Oncol. 2008;26(10):1626-1634.
33. Douillard JY, Siena S, Cassidy J, et al. Randomised, phase III trial of panitumumab with infusional fluorouracil, leucovorin, and oxaliplatin (FOLFOX4) versus FOLFOX4 alone as first-line treatment in patients with previously untreated metastatic colorectal cancer: the PRIME study. J Clin Oncol. 2010;28(31):4697-4705.
34. Arnold D, Lueza B, Douillard JY, et al. Prognostic and predictive value of primary tumour side in patients with RAS wild-type metastatic colorectal cancer treated with chemotherapy and EGFR directed antibodies in six randomised trials. Ann Oncol. 2017;28(8):1713-1729.
35. Venook AP, Niedzwiecki D, Innocenti F, et al. Impact of primary (1o) tumour location on overall survival (OS) and progression-free survival (PFS) in patients (pts) with metastatic colorectal cancer (mCRC): analysis of CALGB/SWOG 80405 (Alliance). J Clin Oncol. 2016;34(15_suppl):3504.
36. Van Cutsem E, Lenz H-J, Köhne C-H, et al. Fluorouracil, leucovorin, and irinotecan plus cetuximab treatment and RAS mutations in colorectal cancer. J Clin Oncol. 2015;33(7):692-700.
37. Cremolini C, Antoniotti C, Rossini D, et al. Upfront FOLFOXIRI plus bevacizumab and reintroduction after progression versus mFOLFOX6 plus bevacizumab followed by FOLFIRI plus bevacizumab in the treatment of patients with metastatic colorectal cancer (TRIBE2): a multicentre, open-label, phase 3, randomised, controlled trial. Lancet Oncol. 2020;21(4):497-507.
38. Rossini D, Antoniotti C, Lonardi S, et al. Upfront modified fluorouracil,leucovorin, oxaliplatin, and irinotecan plus panitumumab versus fluorouracil, leucovorin, and oxaliplatin plus panitumumab for patients with RAS/BRAF wild-type metastatic colorectal cancer: the phase III TRIPLETE study by GONO. J Clin Oncol. 2022:JCO2200839.https://doi.org/10.1200/JCO.22.00839.
39. Modest DP, Martens UM, Riera-Knorrenschild J, et al. FOLFOXIRI plus panitumumab as first-line treatment of RAS wild-type metastatic colorectal cancer: the randomised, open-label, phase II VOLFI study (AIO KRK0109). J Clin Oncol. 2019;37(35):3401-3411.
40. Vassilev ZP, Fan X, Xu J, Ostojic H, Barzi A. Use of FOLFOXIRI Plus Bevacizumab and Subsequent Therapies in Metastatic Colorectal Cancer: An Age-Stratified Analysis.Clin Colorectal Cancer. 2024 Sep;23(3):258-271.e1. doi: 10.1016/j.clcc.2024.05.001
41. Van Cutsem E, Cervantes A, Adam R, et al. ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann Oncol. 2016;27(8):1386-1422.
42. Jonker DJ, O’Callaghan CJ, Karapetis CS, et al. Cetuximab for the treatment of colorectal cancer. N Engl J Med. 2007;357(20):2040-2048.
43. Chibaudel B, Maindrault-Goebel F, Lledo G, et al. Can chemotherapy be discontinued in unresectable metastatic colorectal cancer? The GERCOR OPTIMOX2 study. J Clin Oncol. 2009;27(34):5727-5733.
44. Sobrero AF, Maurel J, Fehrenbacher L, et al. EPIC: phase III trial of cetuximab plus irinotecan after fluoropyrimidine and oxaliplatin failure in patients with metastatic colorectal cancer. J Clin Oncol.2008;26(14):2311-2319.
45. Peeters M, Oliner KS, Price TJ, et al. Analysis of KRAS/NRAS mutations in a phase III study of panitumumab with FOLFIRI compared with FOLFIRI alone as second-line treatment for metastatic colorectal cancer. Clin Cancer Res. 2015;21(24):5469-5479.
46. Tol J, Koopman M, Cats A, et al. Chemotherapy, bevacizumab, and cetuximab in metastatic colorectal cancer. N Engl J Med. 2009;360(6):563-572.
47. 41. Wille-Jorgensen P, Syk I, Smedh K, et al. Effect of more vs less frequent follow-up testing on overall and colorectal cancerspecific mortality in patients with stage II or III colorectal cancer: the COLOFOL randomized clinical trial. JAMA. 2018;319: 2095-2103.
48. Alhayek-Aí M, López-Calviño B, Pértega-Díaz S, et al. Intensive followup strategies improve outcomes in nonmetastatic colorectal cancer patients after curative surgery: a systematic review and meta-analysis. Ann Oncol. 2014;26:644-656.
49. Chau I, Allen MJ, Cunningham D, et al. The value of routine serum carcino-embryonic antigen measurement and computed tomography in the surveillance of patients after adjuvant chemotherapy for colorectal cancer. J Clin Oncol. 2004;22:1420-1429.
50. Tsikitis VL, Malireddy K, Green EA, et al. Postoperative surveillance recommendations for early stage colon cancer based on results from the clinical outcomes of surgical therapy trial. J Clin Oncol. 2009;27: 3671-3676.
51. Rex DK, Kahi CJ, Levin B, et al. Guidelines for colonoscopy surveillance after cancer resection: a consensus update by the American Cancer Society and the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2006;130:1865-1871.
52. Howell D, Hack TF, Oliver TK, et al. Models of care for post-treatment follow-up of adult cancer survivors: a systematic review and quality appraisal of the evidence. J Cancer Surviv. 2012;6:359-371.
53. Grunfeld E, Earle CC. The interface between primary and oncology specialty care: treatment through survivorship. J Natl Cancer Inst Monogr. 2010;2010:25-30.