Endovascular Intervention
| الموقع: | EHC | Egyptian Health Council |
| المقرر الدراسي: | طب المخ والأعصاب |
| كتاب: | Endovascular Intervention |
| طبع بواسطة: | Guest user |
| التاريخ: | الأربعاء، 6 مايو 2026، 3:45 AM |
الوصف
"last update:
27 July 2025" Download Guideline
- Executive Summary
|
|
These guidelines are concerned with clinical practice standards of endovascular intervention neurology procedures. It will discuss practicing physician qualifications, specific requirement for treating centers and specific pre & postoperative care and indications of different endovascular intervention neurology procedures.
|
- Recommendations
|
|
I) PHYSICIAN QUALIFICATIONS1- Practicing physician must have a valid license to practice medicine within their respective countries. 2- Practicing physician specialization criteria should be defined at a national level according to national medical regulations. They must have accomplished training in one of the following medical specialties: Neurology, Neurosurgery, Intervention radiology. 3- Practicing physician must have completed an accredited post graduate dedicated training in intervention neurology subspecialty. This program should have not less than 24 months mandatory dedicated training in intervention neurology. Good practice statement II) REQUIREMENTS FOR PRACTICING INSTITUTIONS/DEPARTMENTS1- Intervention neurology practicing must take place in institutions/departments operating in accordance with the national standards of medical service providence. 2- All patients would be treated at a center offering a full spectrum of neuroendovascular care. 3- Ideally, treating centers should have the following requirements at least to provide safe and efficient intervention neurology services: a. Offers full spectrum of neuroendovascular therapy (including aneurysm treatment, surgical and endovascular, arteriovenous malformations, arteriovenous fistulas, etc.) b. At least 250 case per year of stroke patients’ management in a dedicated neuroscience department. c. Dedicated intensive care unit/stroke unit to manage pre- and post-operative patients. d. Standardized care pathways should be implemented with clinical practice guidelines, order sets, and other tools to ensure consistent care delivery and minimize practice variability. This should apply to providers, and nursing and ancillary staff. Strong recommendation & High level of evidence (Pierot et al., 2018) III) PREROCEDURE PATIENT CARE1- Preprocedural documentation for elective diagnostic cervicocerebral/spinal catheter angiography, must contain the following: a. Clinically significant history, including indications for the procedure. b. Clinically significant physical examination and diagnostic imaging findings, including neurological and vascular examinations appropriate to the procedure performed, and a general examination of relevant organ systems. c. Laboratory evaluation as appropriate, including but not limited to measurement of hemoglobin, hematocrit, creatinine, electrolytes, and coagulation parameters. d. Informed consent must be in compliance with all local laws and policies. Strong recommendation & High level of evidence (ACR-ASNR-SIR-SNIS Practice Parameter, 2021) IV) PATIENT
SELECTION, INDICATIONS AND OUTCOMES
|
- Acknowledgements
“We would like to acknowledge the Neurology Committee of National Egyptian Guidelines for adapting & reviewing these guidelines.
Scientific group members: Ahmed Elbassiouny, Wael Osman, Mohammed Elazzazy, Wessam Fathy, Hassan Hosni, Azza Abdlnasser, Mona Ahmed Nada, Magdy Khalaf, Tarek Rageh, Mohamed Foad, Ahmed Fawzi Amin, Khaled Mohamed Ossama, Romany Adly, Amro Said, Ahmed Elsaid, Ahmed Sarhan, Ayman Elsudany, Rady Yousef, Sherif Salah, Ahmed Ateia.- Abbreviations
aSAH aneurysmal subarachnoid hemorrhage
BP blood pressure
CBF cerebral blood flow
CSF cerebrospinal fluid
CT computed tomography
CTA computed tomography angiography
CTP computed tomography perfusion
DCI delayed cerebral ischemia
DSA digital subtraction angiography
EEG electroencephalography
EVD external ventricular drain
GCS Glasgow Coma Scale
ICH intracerebral hemorrhage
ICP intracranial pressure
ICU intensive care unit
LOE Level of Evidence
LOS length of stay
LP lumbar puncture
MCA middle cerebral artery
MMSE Mini-Mental Status Examination
mRS modified Rankin Scale
NIHSS National Institutes of Health Stroke Scale
QOL quality of life
RR relative risk
SAH subarachnoid hemorrhage
TCD transcranial Doppler
TTM therapeutic temperature management
AF - Atrial fibrillation
ASC - Acute stroke center
ASPECTS - Alberta Stroke Program Early Computed Tomography Score
BP - Blood pressure
CT - Computed tomography
CTA - Computed tomography angiography
DOAC - Direct oral anticoagulant
DWI - Diffusion-weighted imaging
FLAIR - Fluid-attenuated inversion recovery
HDL - High density lipoprotein
ICH - Intracerebral haemorrhage
INR - International normalized ratio (for blood clotting time)
LDL - Low density lipoprotein
MCA - Middle cerebral artery
MR - Magnetic resonance
MRA - Magnetic resonance angiography
MRI - Magnetic resonance imaging
mRS - Modified Rankin Scale score
NHS - National Health Service
NICE - National Institute for Health and Care Excellence
NIHSS - National Institute of Health Stroke Scale
PAF - Paroxysmal atrial fibrillation
PFO - Patent foramen ovale
TIA - Transient ischemic attack
TOE - Transesophageal echocardiogram
VA - Vertebral artery
VKA - Vitamin K antagonist
WHO - World Health Organization- Glossary
Acute stroke service - Consists of: a) a comprehensive stroke center (CSC) providing hyperacute, acute and inpatient rehabilitation including thrombectomy (thrombectomy center) and neurosurgery; or b) an acute stroke center (ASC) providing hyperacute, acute and inpatient rehabilitation. All components of a specialist acute stroke service should be based in a hospital that can investigate and manage people with acute stroke and their medical and neurological complications.
Alteplase - A medicine used for thrombolysis.
Anticoagulants - A group of medicines used to reduce the risk of clots by thinning the blood.
Antiplatelets - A group of medicines used to prevent the formation of clots by stopping platelets in the blood sticking together.
Antithrombotics - The generic name for all medicines that prevent the formation of blood clots. This includes antiplatelets and anticoagulants.
Atherosclerosis - Fatty deposits that harden on the inner wall of the arteries (atheroma) and roughen its surface; this makes the artery susceptible to blockage either by narrowing or by formation of a blood clot.
Atrial fibrillation - A heart condition that causes an irregular heartbeat, often faster than the normal heart rate.
Cardiovascular disease - Disease of the heart and/or blood vessels.
Carotid angioplasty - surgical procedure that widens the internal diameter of the carotid artery, after it has been narrowed by atherosclerosis.
Carotid arteries - Main blood vessels in the neck, which supply oxygenated blood to the brain.
Carotid stenosis - The narrowing of the carotid arteries in the neck.
Clipping - A metal device used surgical to close the neck of aneurysm sticking
Coiling - A metal which deploy inside aneurysm to secured
Computed tomography (CT) - An X ray technique used to examine the brain.
Computerized angiography – Anon invasive technique that allow for details of brain blood vessels
Cost-effectiveness - The extent to which the benefits of a treatment outweigh the costs.
Diagnostic cervicocerebral catheter angiography – a complete patient encounter involving percutaneous passage of a catheter into the carotid or the vertebral arteries followed by injection of contrast material and imaging and diagnostic evaluation of the intracranial and extracranial circulation using film or digital imaging systems.
Doppler ultrasound -An imaging technique that measures blood flow and velocity through blood vessels.
EVD – External ventricular drain which used to drain haemorrhage outside ventricle
Hyperlipidemia - Raised levels of lipids (cholesterol, triglycerides or both) in the blood serum.
Hyperlipidemia - Raised levels of lipids (cholesterol, triglycerides or both) in the blood serum.
Hypertension - Raised blood pressure.
Hypertension - Raised blood pressure.
Indicator - a specific, quantifiable, and objective measure of quality.
Ischemic stroke - A stroke that happens when a blood clot blocks an artery that is carrying blood to the brain.
Magnetic resonance imaging (MRI) - A non-invasive imaging technique that allows for detailed examination of the brain.
Major complication –a stroke or other event that results in admission to the hospital for therapy (for outpatient procedures), requires an unplanned increase in the level of care resulting in prolonged hospitalization, or results in permanent adverse sequelae or death.
Mechanical ventilation - Advice used to secured respiration when patient had DCL.
Minor complication – a transient ischemic event or other occurrence that results in no sequelae; however, such an event may require minimal therapy or a short hospital stay for observation (generally overnight).
MRI with diffusion-weighted imaging - This type of scan shows areas of recent ischemic brain damage.
Seizure - abnormal brain discharge cause body seize
Stroke - A clinical syndrome, of presumed vascular origin, typified by rapidly developing signs of focal or global disturbance of cerebral functions lasting more than 24 hours or leading to death.
Stroke – a focal neurological deficit lasting longer than 24 hours, typically documented by imaging findings clinically relevant to the deficit.
Subarachnoid haemorrhage: a haemorrhage in subarachnoid space
Successful examination – a technically successful procedure and set of images resulting in identification or exclusion of the suspected pathology or other pathology capable of being identified with arteriography.
Threshold – a specific level of an indicator that should prompt the performance of a review.
Thrombectomy - The excision of a blood clot from a blood vessel.
Thrombolysis - The use of medicines to break up a blood clot. An example of thrombolysis medicine is alteplase, also sometimes called tPA.
Transient ischemic attack (TIA) – a brief episode of neurological dysfunction caused by focal brain or retinal ischemia, with clinical symptoms typically lasting less than one hour, usually without imaging evidence of infarction (some TIAs are associated with diffusion restriction detected on MRI indicating ischemia or infarction with complete resolution of symptoms within 24 hours.)- Introduction, purpose, scope and audience
INTRODUCTION The field of interventional neurology has expanded dramatically in recent years. Beyond acute ischemic stroke, endovascular therapy may also be used for the treatment of intracranial aneurysms, embolization of arteriovenous malformations and fistulae, preoperative embolization of intracranial and cervical vascular tumors, and stenting of stenotic extracranial and intracranial vessels. The advent of neurointerventional therapy now even includes the possibility of treatment of conditions as diverse as intracranial hypertension, by stenting venous sinuses. SCOPE AND PURPOSE In light of the growth of the field and the increase in the potential to expand the performance of interventional endovascular treatments of stroke and other neurological conditions, this guideline define appropriate clinical indications and specific therapeutic approaches for these procedures in adults (i.e., people aged over 18 years). This guideline is intended to help practicing physicians ensure that patients undergo cervicocerebral/spinal intervention catheter angiography for appropriate reasons, that the methods used and the periprocedural care provided are adequate to minimize complications, and that the quality of the studies obtained is sufficient to answer the clinical questions that prompted them. It will also outline required qualifications needed for either physician and neurointervention centers that will ensure safe & efficient practicing of neurointervention procedures. This guideline is not intended to overrule regulations or standards concerning the provision of services and should be considered in conjunction with them. In considering and implementing this guideline, users are advised to also consult and follow all appropriate legislation, standards and good practice. |
- TARGET AUDIENCE
The guideline is intended for:
· those providing endovascular intervention neurology – neurologists, neurosurgeons and intervention radiologists physicians.
· those commissioning, providing endovascular neuro-intervention services;
· anyone seeking to improve the care of people undergoing endovascular neuro-intervention procedures.
- Methods
We adopted WHO proposed seven distinct steps for development of clinical guidelines to ensure a thorough and rigorous process.
The final research questions and consensus questions are structured using the ‘Population, Intervention, Control, Outcome’ (PICO) format. Each question is assigned to an appropriate topic group according to the scope.
A literature search is undertaken for each individual question to identify studies that help to answer the question and provide evidence that is robust enough to allow recommendations to be made. Literature searching is coordinated by the stroke guideline team. These initial searches look for guidelines, systematic reviews, and meta-analyses only and cover the following databases:
a. Cochrane Database of Systematic Reviews (CDSR)
b. MEDLINE.
A comprehensive search for guidelines was undertaken to identify the most relevant guidelines to consider for adaptation. inclusion/exclusion criteria followed in the search and retrieval of guidelines to be adapted:
· Selecting only evidence-based guidelines (guideline must include a report on systematic literature searches and explicit links between individual recommendations and their supporting evidence)
· Selecting only national and/or international guidelines Specific range of dates for publication (using Guidelines published or updated 2015 and later)
· Selecting peer reviewed publications only
· Selecting guidelines written in English language
· Excluding guidelines written by a single author not on behalf of an organization in order to be valid and comprehensive, a guideline ideally requires multidisciplinary input.
· Excluding guidelines published without references as the panel needs to know whether a thorough literature review was conducted and whether current evidence was used in the preparation of the recommendations
The following characteristics of the retrieved guidelines were summarized in a table:
· Developing organization/authors
· Date of publication, posting, and release
· Country/language of publication
· Date of posting and/or release
· Dates of the search used by the source guideline developers
All Guidelines were screened and appraised using AGREE II instrument (www.agreetrust.org) by at least two members. The panel decided a cut-off point or rank the guidelines (any guideline scoring above 50% on the rigor dimension was retained). The Guideline Development Group has decided to adapt the current guidelines guided by most recent guidelines and standard of practice reported from ACR, ASNR, SIR & SNIS, AHA/ASA[AA1] . (Wojak, J.C. et al., 2015 and Pierot et al., 2018 and ACR-ASNR-SIR-SNIS Practice Parameter, 2021)
EVIDENCE ASSESSMENT
According to WHO handbook for Guidelines we used the GRADE (Grading of Recommendations, Assessment, Development and Evaluation) approach to assess the quality of a body of evidence, develop and report recommendations. GRADE methods are used by WHO because these represent internationally agreed standards for making transparent recommendations. Detailed information on GRADE is available through the GRC secretariat and on the following sites:
■ GRADE working group: https://www.gradeworkinggroup.org/
■ GRADE online training modules: http://cebgrade.mcmaster.ca/
■ GRADE profile software: http://ims.cochrane.org/revman/gradepro
THE STRENGTH OF THE RECOMMENDATION
The strength of a recommendation communicates the importance of adherence to the recommendation.
§ Strong recommendations
With strong recommendations, the guideline communicates the message that the desirable effects of adherence to the recommendation outweigh the undesirable effects. This means that in most situations the recommendation can be adopted as policy.
§ Conditional recommendations
These are made when there is greater uncertainty about the four factors above or if local adaptation has to account for a greater variety in values and preferences, or when resource use makes the intervention suitable for some, but not for other locations. This means that there is a need for substantial debate and involvement of stakeholders before this recommendation can be adopted as policy.
- When not to make recommendations
When there is lack of evidence on the effectiveness of an intervention, it may be appropriate not to make a recommendation.
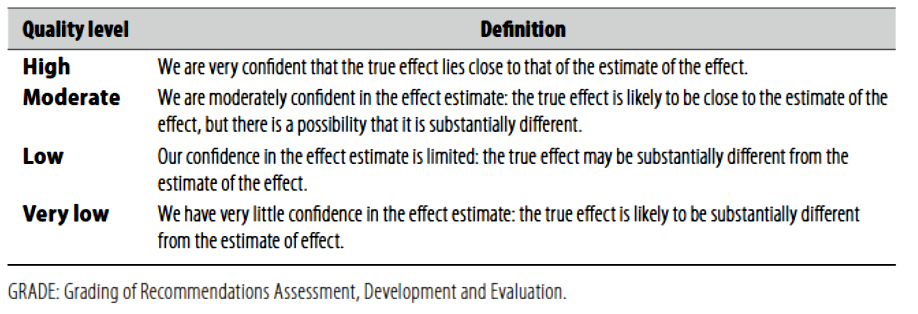
(Table-1) Quality of evidence in GRADE
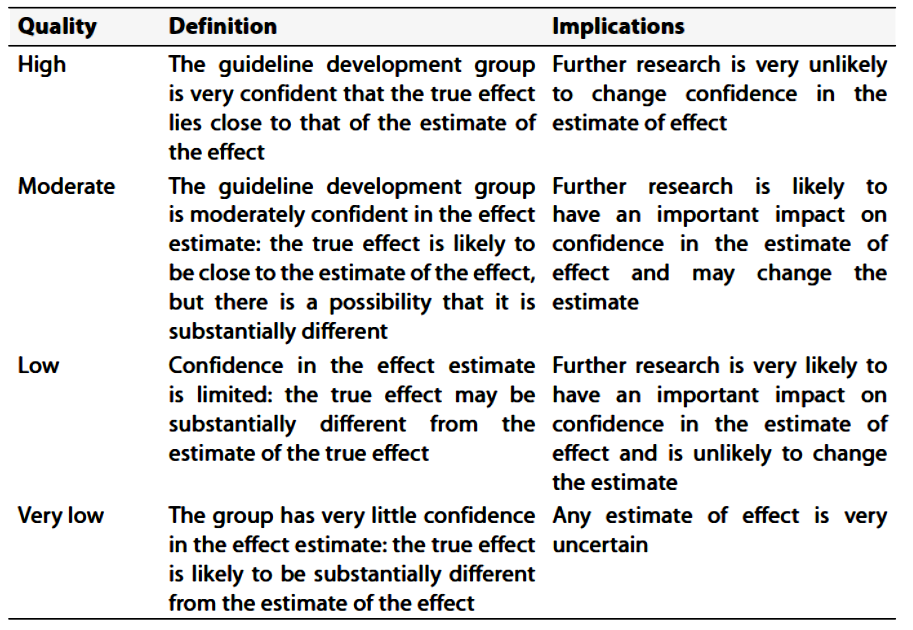
(Table-2) Significance of the four levels of evidence
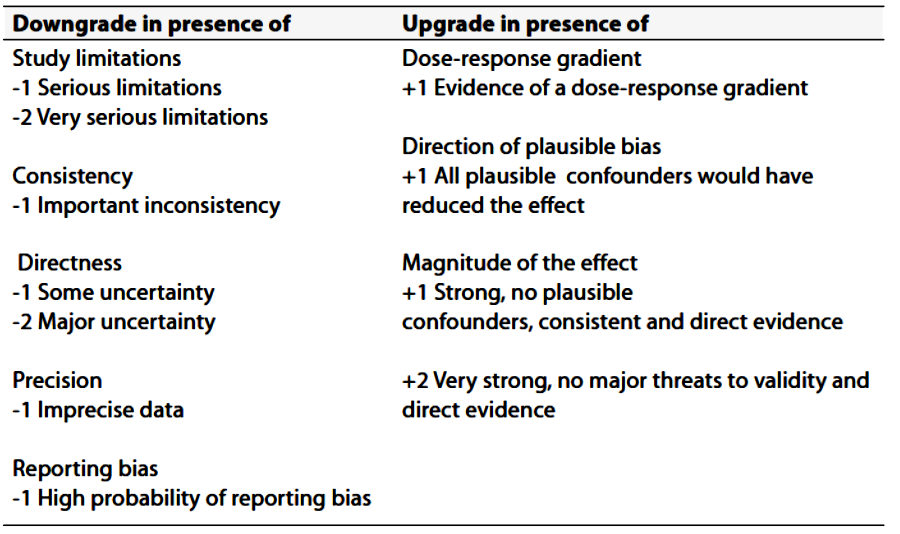
(Table-3) Factors that determine How to upgrade or downgrade the quality of Evidence
- Implementation considerations
|
|
In this section key recommendations are provided to guide the funding, planning and delivery of services along the entire pathway of stroke care. These recommendations will provide the best proposed outline for delivering endovascular intervention neurology services for stroke management. Recommendations: 1. Comprehensive stroke services should include the whole stroke pathway from prevention (including neurovascular services) through pre-hospital and acute care, early rehabilitation, secondary prevention, early supported discharge, community rehabilitation, systematic follow-up, palliative care and long-term support. 2. A public education and professional training strategy should be developed and implemented to ensure that the public and emergency personnel (e.g. staff in emergency call centers) can recognize when a person has a suspected stroke or TIA and respond appropriately. This should be implemented in such a way that it can be formally evaluated. 3. Along the pathway of stroke care, there should be protocols between healthcare providers and social services that enable seamless and safe transfers of care without delay. 4. The provision of comprehensive acute stroke services may require the development of hub-and-spoke models of care (where a few hospitals in a region are designated to provide the hyperacute care for all patients), or telemedicine networks and other forms of cross-site working. 5. The optimal disposition of acute stroke services will depend on the geography of the area served, with the objective of delivering the maximum number of time-critical treatments to the greatest number of people with stroke. 6. Healthcare providers should enact all the secondary stroke prevention measures recommended in this guideline. Effective secondary prevention should be assured through a process of regular audit and monitoring. 7. Healthcare authorities should play an active role in promoting secondary vascular prevention, which is a public health issue as well as being relevant to the individual person with stroke. |
- Clinical indicators for monitoring
|
|
Values outside of the suggested thresholds should trigger a review of policies and/or practices within the department to determine the causes and to implement changes to manage the incidence of related indicators. 1. For quality Improvement Issues related to cervicocerebral/spinal angiography; the following indicators and related thresholds may be used: a. Reported Success Rates of Diagnostic cervicocerebral/spinal catheter angiography is 98% (suggested threshold <98%). b. Reported Overall Procedure complication, either neurologic or systemic occurring within 24 hours of the angiography, is 2% (suggested threshold <2%) 2. For quality improvement issues related to spinal dural AVF endovascular embolization; the following indicators and related thresholds may be used: a. The reported threshold indicators of the efficacy are technical success (81%) with occlusion of the targeted vessels and clinical success (60%) with improvement or stabilization of symptoms. b. The reported overall complication of dural AVF embolization is 10-15% (suggested threshold <5%). 3- For quality improvement issues related to spinal AVM endovascular embolization; the following indicators and related thresholds may be used: a. The reported threshold indicators of the efficacy are technical success (90%) with occlusion of the targeted vessels and clinical success (50%) with improvement or stabilization of symptoms. b. The reported overall complication of dural AVF embolization is 10-15% (suggested threshold <10%). 4- For quality improvement issues related to extracranial tumor endovascular embolization, major complications are rare. However, stroke and intracerebral hemorrhage have been reported in up to 3-6% during intracranial embolization (suggested threshold <5%).
|
- Research needs
|
|
1- Cost effective studies for implementation of endovascular intervention procedures Vs best medical treatment Vs surgical intervention in certain situations in Egyptian population. 2- Needs assessment studies for maximizing numbers of comprehensive stroke centers providing endovascular intervention stroke services. |
- Updating the guideline
|
|
To keep these recommendations valid, all guidelines need to be periodically updated. This will be done whenever strong evidence is available and necessitates recommendation updates. |
- References
|
|
1. ACR-ASNR-SIR-SNIS Practice Parameter for the Performance of Diagnostic Cervicocerebral Catheter Angiography in Adults (amended 2021). Available at https://gravitas.acr.org/PPTS/GetDocumentView?docId=167+&releaseId=2. 2. Albers GW, Marks MP, Kemp S, Christensen S, Tsai JP, Ortega-Gutierrez S, McTaggart R, Torbey MT, Kim-Tenser M, Leslie-Mazwi T, Sarraj A, Kasner SE, Ansari SA, Yeatts SD, Hamilton S, Mylnash M, Heit JJ, Zaharchuk G, Kim S, Carrozzella J, Palesch YY, Demchuk AM, Bammer R, Lavori PW, Broderick JP, Lansberg MG; on behalf of the DEFUSE 3Investigators. Thrombectomy for stroke with perfusion imaging selection at 6–16 hours. N Engl J Med. In press. 3. Amarenco P, Denison H, Evans SR, Himmelmann A, James S, Knutsson M, Ladenvall P, Molina CA, Wang Y, Johnston SC; THALES Steering Committee and Investigators. Ticagrelor added to aspirin in acute non severe ischemic stroke or transient ischemic attack of atherosclerotic origin. Stroke. 2020;51:3504–3513. Doi: 10.1161/STROKEAHA.120.032239 4. Barnett HJM, Taylor DW, Haynes RB, Sackett DL, Peerless SJ, Ferguson GG, Fox AJ, Rankin RN, Hachinski VC, Wiebers DO, et al. Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N Engl J Med. 1991;325:445–453. 5. Brott TG, Hobson RW 2nd, Howard G, Roubin GS, Clark WM, Brooks W, Mackey A, Hill MD, Leimgruber PP, Sheffet AJ, et al; CREST Investigators. Stenting versus endarterectomy for treatment of carotid-artery stenosis. N Engl J Med. 2010;363:11–23. Doi: 10.1056/NEJMoa0912321 6. Buscot MJ, Chandra RV, Maingard J, Nichols L, Blizzard L, Stirling C, Smith K, Lai L, Asadi H, Froelich J, et al. Association of onset-to-treatment time with discharge destination, mortality, and complications among patients with aneurysmal subarachnoid hemorrhage. JAMA Netw Open. 2022;5:e2144039. doi:10.1001/jamanetworkopen.2021.44039 7. Bush CK, Kurimella D, Cross LJ, Conner KR, Martin-Schild S, He J, Li C, Chen J, Kelly T. Endovascular treatment with stent-retriever devices for acute ischemic stroke: a meta-analysis of randomized controlled trials. PLoS One. 2016;11:e0147287. Doi: 10.1371/journal.pone.0147287. 8. Catapano JS, Lang MJ, Koester SW, Wang DJ, DiDomenico JD, Fredrickson VL, Cole TS, Lee J, Lawton MT, Ducruet AF, et al. Digital subtraction cerebral angiography after negative computed tomography angiography findings in non-traumatic subarachnoid hemorrhage. J Neurointerv Surg. 2020;12:526–530. Doi: 10.1136/neurintsurg-2019-015375 9. Chimowitz MI, Lynn MJ, Derdeyn CP, Turan TN, Fiorella D, Lane BF, Janis LS, Lutsep HL, Barnwell SL, Waters MF, et al; SAMMPRIS Trial Investigators. Stenting versus aggressive medical therapy for intracranial arterial steno sis. N Engl J Med. 2011;365:993–1003. Doi: 10.1056/NEJMoa1105335 10. Chimowitz MI, Lynn MJ, Howlett-Smith H, Stern BJ, Hertzberg VS, Frankel MR, Levine SR, Chaturvedi S, Kasner SE, Benesch CG, et al; Warfarin Aspirin Symptomatic Intracranial Disease Trial Investigators. Comparison of warfarin and aspirin for symptomatic intracranial arterial stenosis. N Engl J Med. 2005;352:1305–1316. Doi: 10.1056/NEJMoa043033 11. De Leacy, R. et al. (2022) ‘Endovascular treatment in the multimodality management of brain arteriovenous malformations: Report of the society of neurointerventional surgery standards and Guidelines Committee’, Journal of Neurointerventional Surgery, 14(11), pp. 1118–1124. doi:10.1136/neurintsurg-2021-018632. 12. Dean BL, Flom RA, Wallace RC, et al. Efficacy of endovascular treatment of meningiomas: evaluation with matched samples. AJNR Am J Neuroradiol 1994;15:1675e80. 13. Deng X, Gao F, Zhang D, Zhang Y, Wang R, Wang S, Cao Y, Zhao Y, Pan Y, Liu X, et al. Direct versus indirect bypasses for adult ischemic-type moyamoya disease: a propensity score-matched analysis. J Neurosurg. 2018;128:1785–1791. Doi: 10.3171/2017.2.JNS162405 14. Dippel DW, Majoie CB, Roos YB, van der Lugt A, van Oostenbrugge RJ, van Zwam WH, Lingsma HF, Koudstaal PJ, Treurniet KM, van den Berg LA, Beumer D, Fransen PS, Berkhemer OA; for the MR CLEAN Investigators. Influence of device choice on the effect of intra-arterial treatment for acute ischemic stroke in MR CLEAN (Multicenter Randomized Clinical Trial of Endovascular Treatment for Acute Ischemic Stroke in the Netherlands). Stroke. 2016;47:2574–2581. Doi: 10.1161/STROKEAHA.116.013929. 15. Duffis, E.J. et al. (2012) ‘Head, neck, and Brain tumor embolization guidelines’, Journal of Neurointerventional Surgery, 4(4), pp. 251–255. doi:10.1136/neurintsurg-2012-010350. 16. Farb RI, Vanek I, Scott JN, et al. Idiopathic intracranial hypertension: the prevalence and morphology of sinovenous stenosis. Neurology. 2003;60:1418–24. 17. Giorgianni A, Agosti E, Molinaro S, Terrana AV, Vizzari FA, Nativo L, Garg K, Craparo G, Conti V, Locatelli D, et al. Flow diversion for acutely ruptured intracranial aneurysms treatment: a retrospective study and literature review. J Stroke Cerebrovasc Dis. 2022;31:106284. doi:10.1016/j.jstrokecerebrovasdis.2021.106284 18. Gornik HL, Persu A, Adlam D, Aparicio LS, Azizi M, Boulanger M, Bruno RM, de Leeuw P, Fendrikova-Mahlay N, Froehlich J, et al. First international consensus on the diagnosis and management of fibromuscular dysplasia. Vasc Med. 2019;24:164–189. Doi: 10.1177/1358863X18821816 19. Haussen DC, Grossberg JA, Bouslama M, Pradilla G, Belagaje S, Bianchi N, Allen JW, Frankel M, Nogueira RG. Carotid web (intimal fibromuscular dysplasia) has high stroke recurrence risk and is amenable to stenting. Stroke. 2017;48:3134–3137. Doi: 10.1161/STROKEAHA.117.019020 20. Hoh, B.L. et al. (2023b) ‘2023 guideline for the management of patients with aneurysmal subarachnoid hemorrhage: A guideline from the American Heart Association/American Stroke Association’, Stroke, 54(7). doi:10.1161/str.0000000000000436. 21. Howard BM, Hu R, Barrow JW, Barrow DL. Comprehensive review of imaging of intracranial aneurysms and angiographically negative subarachnoid hemorrhage. Neurosurg Focus. 2019;47:E20. Doi: 10.3171/2019.9.FOCUS19653 22. Howard G, Roubin GS, Jansen O, Hendrikse J, Halliday A, Fraedrich G, Eckstein HH, Calvet D, Bulbulia R, Bonati LH, et al; Carotid Stenting Trialists’ Collaboration. Association between age and risk of stroke or death from carotid endarterectomy and carotid stenting: a meta-analysis of pooled patient data from four randomized trials. Lancet. 2016;387:1305 1311. Doi: 10.1016/S0140-6736(15)01309-4 23. Ido K, Kurogi R, Kurogi A, Nishimura K, Arimura K, Nishimura A, Ren N, Kada A, Matsuo R, Onozuka D, et al; J-ASPECT Study Collaborators. Effect of treatment modality and cerebral vasospasm agent on patient outcomes after aneurysmal subarachnoid hemorrhage in the elderly aged 75 years and older. PLoS One. 2020;15:e0230953. doi:10.1371/journal.pone.0230953 24. Jeon JP, Kim JE, Cho WS, Bang JS, Son YJ, Oh CW. Meta-analysis of the surgical outcomes of symptomatic moyamoya disease in adults. J Neurosurg. 2018;128:793–799. Doi: 10.3171/2016.11.JNS161688 25. Jovin TG, Saver JL, Ribo M, Pereira V, Furlan A, Bonafe A, Baxter B, Gupta R, Lopes D, Jansen O, Smith W, Gress D, Hetts S, Lewis RJ, Shields R, Berry SM, Graves TL, Malisch T, Rai A, Sheth KN, Liebeskind DS, Nogueira RG. Diffusion-weighted imaging or computerized tomography perfusion assessment with clinical mismatch in the tri age of wake up and late presenting strokes undergoing neurointervention with Trevo (DAWN) trial methods. Int J Stroke. 2017;12:641–652. Doi: 10.1177/1747493017710341. 26. Kwon SU, Cho YJ, Koo JS, Bae HJ, Lee YS, Hong KS, Lee JH, Kim JS. Cilostazol prevents the progression of the symptomatic intracranial arterial stenosis: the multicenter double-blind placebo-controlled trial of cilostazol in symptomatic intracranial arterial stenosis. Stroke. 2005;36:782–786. Doi: 10.1161/01.STR.0000157667.06542.b7 27. Lapergue B, Blanc R, Gory B, Labreuche J, Duhamel A, Marnat G, Saleme S, Costalat V, Bracard S, Desal H, Mazighi M, Consoli A, Piotin M; ASTER Trial Investigators. Effect of endovascular contact aspiration vs stent retriever on revascularization in patients with acute ischemic stroke and large vessel occlusion: the ASTER randomized clinical trial. JAMA. 2017;318:443–452. Doi: 10.1001/jama.2017.9644. 28. Leacy RD, Ansari SA, Schirmer CM, Cooke DL, Prestigiacomo CJ, Bulsara KR, et al. Endovascular treatment in the multimodality management of brain arteriovenous malformations: report of the Society of NeuroInterventional Surgery Standards and Guidelines Committee. Journal of NeuroInterventional Surgery. 2022 Nov 1;14(11):1118–24. 29. Lee, S.-K. et al. (2018) ‘Current endovascular strategies for cerebral venous thrombosis: Report of the SNIS standards and Guidelines Committee’, Journal of NeuroInterventional Surgery, 10(8), pp. 803–810. doi:10.1136/neurintsurg-2018-013973. 30. Lemmens R, Hamilton SA, Liebeskind DS, Tomsick TA, Demchuk AM, Nogueira RG, Marks MP, Jahan R, Gralla J, Yoo AJ, Yeatts SD, Palesch YY, Saver JL, Pereira VM, Broderick JP, Albers GW, Lansberg MG; DEFUSE 2, IMS III, STAR, and SWIFT Trialists; DEFUSE 2 IMS III STAR and SWIFT Trialists. Effect of endovascular reperfusion in relation to site of arterial occlusion. Neurology. 2016;86:762–770. Doi: 10.1212/WNL.0000000000002399. 31. Lindgren A, Vergouwen MD, van der Schaaf I, Algra A, Wermer M, Clarke MJ, Rinkel GJ. Endovascular coiling versus neurosurgical clipping for people with aneurysmal subarachnoid haemorrhage. Cochrane Database Syst Rev. 2018;8:CD003085. Doi: 10.1002/14651858.CD003085.pub3 32. Löwhagen Hendén P, Rentzos A, Karlsson JE, Rosengren L, Leiram B, Sundeman H, Dunker D, Schnabel K, Wikholm G, Hellström M, Ricksten SE. General anesthesia versus conscious sedation for endovascular treatment of acute ischemic stroke: the AnStroke trial (Anesthesia During Stroke). Stroke. 2017;48:1601–1607. Doi: 10.1161/ STROKEAHA.117.016554. 33. Marks MP, Lansberg MG, Mlynash M, Kemp S, McTaggart R, Zaharchuk G, Bammer R, Albers GW; DEFUSE 2 Investigators. Correlation of AOL recanalization, TIMI reperfusion and TICI reperfusion with infarct growth and clinical outcome. J Neurointerv Surg. 2014;6:724–728. Doi: 10.1136/neurintsurg-2013-010973. 34. Markus HS, Harshfield EL, Compter A, Kuker W, Kappelle LJ, Clifton A, van der Worp HB, Rothwell P, Algra A; Vertebral Stenosis Trialists’ Collaboration. Stenting for symptomatic vertebral artery stenosis: a preplanned pooled individual patient data analysis. Lancet Neurol. 2019;18:666–673. Doi: 10.1016/S1474-4422(19)30149-8 35. Nagai M, Watanabe E. Benefits of clipping surgery based on three-dimensional computed tomography angiography. Neurol Med Chir (Tokyo).2010;50:630–637. Doi: 10.2176/nmc.50.630 36. Narayanan, S. et al. (2012) ‘Standard of practice: Embolization of spinal arteriovenous fistulae, spinal arteriovenous malformations, and tumors of the spinal axis’, Journal of NeuroInterventional Surgery, 5(1), pp. 3–5. doi:10.1136/neurintsurg-2012-010551. 37. Nogueira RG, Jadhav AP, Haussen DC, Bonafe A, Budzik RF, Bhuva P, Yavagal DR, Ribo M, Cognard C, Hanel RA, Sila CA, Hassan AE, Millan M, Levy EI, Mitchell P, Chen M, English JD, Shah QA, Silver FL, Pereira VM, Mehta BP, Baxter BW, Abraham MG, Cardona P, Veznedaroglu E, Hellinger FR, Feng L, Kirmani JF, Lopes DK, Jankowitz BT, Frankel MR, Costalat V, Vora NA, Yoo AJ, Malik AM, Furlan AJ, Rubiera M, Aghaebrahim A, Olivot JM, Tekle WG, Shields R, Graves T, Lewis RJ, Smith WS, Liebeskind DS, Saver JL, Jovin TG DAWN Trial Investigators. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct [published online ahead of print November 11, 2017]. N Engl J Med. Doi: 10.1056/NEJMoa1706442. http://www. nejm.org/Doi/full/10.1056/NEJMoa1706442. 38. Ogilvy CS, Stieg PE, Awad I, Brown RD, Kondziolka D, Rosenwasser R, et al. Recommendations for the management of intracranial arteriovenous malformations. Stroke [Internet]. 2001 Jun 1;32(6):1458–71. Available from: https://doi.org/10.1161/01.str.32.6.1458 39. Oudshoorn SC, Rinkel GJ, Molyneux AJ, Kerr RS, Dorhout Mees SM, Backes D, Algra A, Vergouwen MD. Aneurysm treatment <24 versus 24-72 h after subarachnoid hemorrhage. Neurocrit Care. 2014;21:4–13. doi:10.1007/s12028-014-9969-8 40. Passero SG, Rossi S. Natural history of vertebrobasilar dolichoectasia. Neurology. 2008;70:66–72. Doi: 10.1212/01.wnl.0000286947.89193.f3 41. Persky MS, Setton A, Niimi Y, et al. Combined endovascular and surgical treatment of head and neck paraganglioma team approach. Head Neck 2002;24:423e31. 42. Phuong Nguyen T, Rehman S, Stirling C, Chandra R, Gall S. Time and predictors of time to treatment for aneurysmal subarachnoid haemorrhage (aSAH): a systematic review. Int J Qual Health Care. 2021;33:mzab019. doi:10.1093/intqhc/mzab019 43. Pierot L, Barbe C, Herbreteau D, Gauvrit JY, Januel AC, Bala F, Ricolfi F, Desal H, Velasco S, Aggour M, et al. Rebleeding and bleeding in the year following intracranial aneurysm coiling: analysis of a large prospective multicenter cohort of 1140 patients: Analysis of Recanalization after Endovascular Treatment of Intracranial Aneurysm (ARETA) Study. J Neurointerv Surg. 2020;12:1219–1225. Doi: 10.1136/neurintsurg-2020-015971 44. Pierot L, Jayaraman MV, Szikora I, AAFITN, ANZSNR, ASNR, CSNR, ESMINT, ESNR, ESO, JSNET, SFNR, SILAN, SNIS, SVIN, WSO, WFITN, et al Standards of practice in acute ischemic stroke intervention: international recommendations Journal of NeuroInterventional Surgery 2018;10:1121-1126. 45. Powers WJ, Clarke WR, Grubb RL Jr, Videen TO, Adams HP Jr, Derdeyn CP; COSS Investigators. Extracranial-intracranial bypass surgery for stroke prevention in hemodynamic cerebral ischemia: the Carotid Occlusion Surgery Study randomized trial. JAMA. 2011;306:1983–1992. Doi: 10.1001/jama.2011.1610 46. Rantner B, Kollerits B, Roubin GS, Ringleb PA, Jansen O, Howard G, Hendrikse J, Halliday A, Gregson J, Eckstein HH, et al; Carotid Stenosis Trialists’ Collaboration. Early endarterectomy carries a lower procedural risk than early stenting in patients with symptomatic stenosis of the in ternal carotid artery: results from 4 randomized controlled trials. Stroke. 2017;48:1580–1587. Doi: 10.1161/STROKEAHA.116.016233 Downloaded from http://ahajournals.org by on January 24, 2025 47. Rothwell PM, Eliasziw M, Gutnikov SA, Fox AJ, Taylor DW, Mayberg MR, Warlow CP, Barnett HJ; Carotid Endarterectomy Trialists’ Collaboration. Analysis of pooled data from the randomized controlled trials of endarterectomy for symptomatic carotid stenosis. Lancet. 2003;361:107–116. Doi: 10.1016/s0140-6736(03)12228-3 48. Rothwell PM, Eliasziw M, Gutnikov SA, Warlow CP, Barnett HJ; Carotid Endarterectomy Trialists Collaboration. Endarterectomy for symptomatic carotid stenosis in relation to clinical subgroups and timing of surgery. Lancet. 2004;363:915–924. Doi: 10.1016/S0140-6736(04)15785-1 49. Ryttlefors M, Enblad P, Kerr RS, Molyneux AJ. International Subarachnoid Aneurysm Trial of neurosurgical clipping versus endovascular coiling: subgroup analysis of 278 elderly patients. Stroke. 2008;39:2720–2726. doi:10.1161/STROKEAHA.107.506030 50. Saver JL, Goyal M, van der Lugt A, Menon BK, Majoie CB, Dippel DW, Campbell BC, Nogueira RG, Demchuk AM, Tomasello A, Cardona P, Devlin TG, Frei DF, du Mesnil de Rochemont R, Berkhemer OA, Jovin TG, Siddiqui AH, van Zwam WH, Davis SM, Castaño C, Sapkota BL, Fransen PS, Molina C, van Oostenbrugge RJ, Chamorro Á, Lingsma H, Silver FL, Donnan GA, Shuaib A, Brown S, Stouch B, Mitchell PJ, Davalos A, Roos YB, Hill MD; HERMES Collaborators. Time to treat ment with endovascular thrombectomy and outcomes from ischemic stroke: a meta-analysis. JAMA. 2016;316:1279–1288. Doi: 10.1001/ jama.2016.13647. 51. Schacht H, Kuchler J, Boppel T, Leppert J, Ditz C, Schramm P, Neumann A. Transluminal balloon angioplasty for cerebral vasospasm after spontaneous subarachnoid hemorrhage: a single-center experience. Clin Neurol Neurosurg. 2020;188:105590. Doi: 10.1016/j.clineuro.2019.105590 52. Schermerhorn ML, Liang P, Eldrup-Jorgensen J, Cronenwett JL, Nolan BW, Kashyap VS, Wang GJ, Motaganahalli RL, Malas MB. Association of trans-carotid artery revascularization vs transfemoral carotid artery stenting with stroke or death among patients with carotid artery stenosis. JAMA. 2019;322:2313–2322. Doi: 10.1001/jama.2019.18441 53. Settecase F, Rayz VL. Advanced vascular imaging techniques. Handb Clin Neurol 2021;176:81–105. 54. Smith LL, Smith DC, Killeen JD, Hasso AN. Operative balloon angioplasty in the treatment of internal carotid artery fibromuscular dysplasia. J Vasc Surg. 1987;6:482–487. Doi: 10.1067/mva.1987.avs0060482 55. Tasar M, Yetiser S. Glomus tumors: therapeutic role of selective embolization. J Craniofac Surg 2004;15:497e505. 56. Ten Brinck MFM, Shimanskaya VE, Aquarius R, Bartels R, Meijer FJA, Koopmans PC, de Jong G, Wakhloo AK, de Vries J, Boogaarts HD. Outcomes after flow diverter treatment in subarachnoid hemorrhage: a meta-analysis and development of a clinical prediction model (OUTFLOW). Brain Sci. 2022;12:394. doi:10.3390/brain sci12030394 57. White JB, Link MJ, Cloft HJ. Endovascular embolization of paragangliomas: a safe adjuvant to treatment. J Vasc Interv Neurol 2008;1:37e41. 58. Wojak, J.C. et al. (2015) ‘Quality Improvement Guidelines for adult diagnostic CERVICOCEREBRAL angiography: Update Cooperative study between the Society of Interventional Radiology (sir), American Society of Neuroradiology (ASNR), and Society of Neurointerventional Surgery (SNIS)’, Journal of Vascular and Interventional Radiology, 26(11), pp. 1596–1608. doi:10.1016/j.jvir.2015.07.002. 59. Yadav JS, Wholey MH, Kuntz RE, Fayad P, Katzen BT, Mishkel GJ, Bajwa TK, Whitlow P, Strickman NE, Jaff MR, et al; Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy Investigators. Protected carotid-artery stenting versus endarterectomy in high-risk patients. N Engl J Med. 2004;351:1493–1501. Doi: 10.1056/NEJMoa040127 60. Zhu D, Yan Y, Zhao P, Duan G, Zhao R, Liu J, Huang Q. Safety and efficacy of flow diverter treatment for blood blister-like aneurysm: a systematic review and meta-analysis. World Neurosurg. 2018;118:e79–e86. doi:10.1016/j.wneu.2018.06.123 |