
- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing community acquired pneumonia diagnosis, treatment, and prevention strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
▪️ Select the target populations and evaluate the outcome.
▪️ Identify the local resources to support the implementation.
▪️ Set timelines.
▪️ Distribute the tasks to the members.
▪️ Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
▪️ Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
▪️ Educational materials: printed or electronic information (software).
▪️ Web-based education: computer-based educational activities.
▪️ A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
▪️ Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
▪️ Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
▪️ Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
▪️ Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
▪️ Mass media campaigns.
3. For Nurses
▪️ Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
▪️ Educational materials: printed.
▪️ A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
▪️Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
▪️ Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
▪️ Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
▪️ Administrative policies and procedures.
▪️ Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
▪️ International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
▪️ Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including:
1. Pathway for assessment of severity of CAP in Primary Care Facilities.
2. Algorithm for treatment of CAP in Acute Care Facilities (Emergency Rooms).
3. Power Point Presentation Lectures for diagnosis, treatment & prevention of CAP
Figure 1. Management of CAP in children
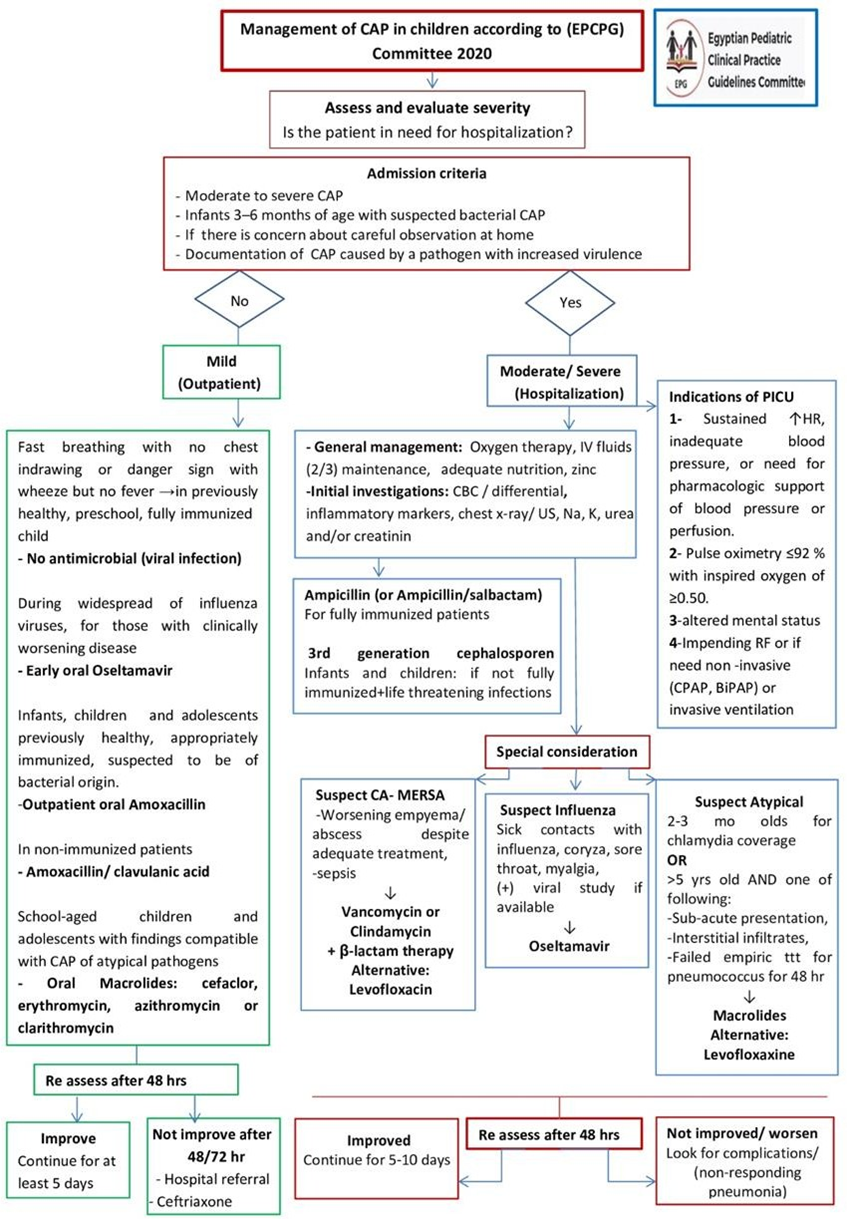
Figure 2. Management of non-responding pneumonia
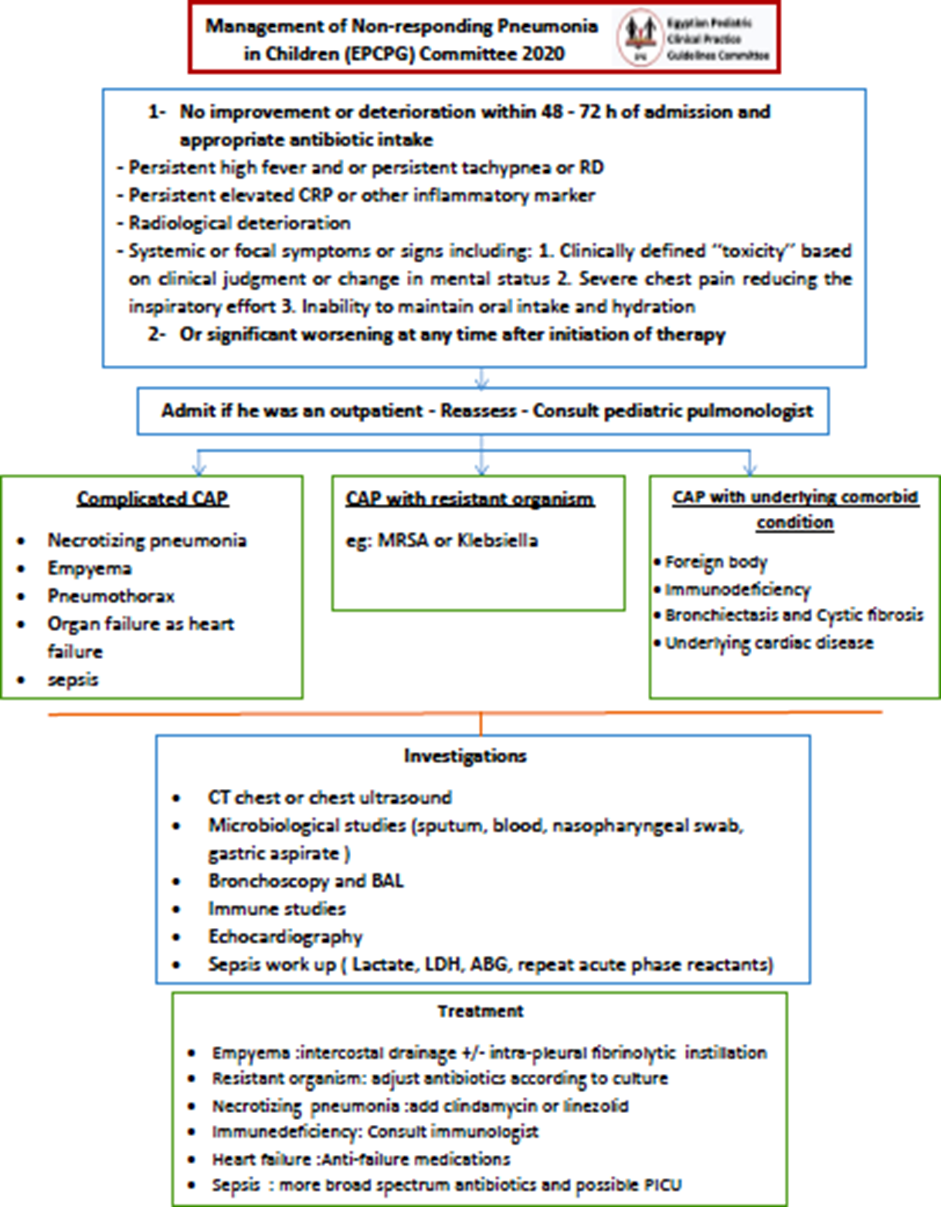
Table 4. Tachypnea According to WHO [29]
|
If the child is: |
The child has fast breathing if you count: |
|
Younger than 2 months 2 months up to 12 months 12 months up to 5 years |
60 breaths per minute or more 50 breaths per minute or more 40 breaths per minute or more. |
Note: The child who is exactly 12 months old has fast breathing if you count 40 breaths per minute or more. A pediatrician measured the respiratory rate by observing the child’s chest movements for one minute while the child rested with no crying or fever.
Table 5. Severity Assessment of Pneumonia [30]
|
Severity Assessment |
Mild |
Moderate |
Severe |
|
Effort of breathing |
Nil or Mild Increase |
Moderate increase |
Severe increase |
|
Respiratory Rate |
Within normal range for age |
Above range for age |
Continuing to rise, and/or evidence of Exhaustion |
|
Oxygen Saturation |
≥95% in room air |
<95% in room air |
Failing to maintain SpO2 ≥95% in 6L Oxygen OR > 90% in air |
|
Circulation |
No tachycardia |
Tachycardia Capillary Refill ≥ 3 sec |
Tachycardia/shock in the Cap Refill ≥ 3 secs |
Table 6. Oral Anti-infective Doses [31]
|
Antibiotics |
Doses |
Notes |
Sensitive Organisms |
|
Amoxicillin |
90 mg/kg/day in 2 doses |
|
Streptococcus pneumoniae Group A Streptococcus Haemophilus influenza |
|
Amoxicillin clavulanate |
Amoxicillin component, 90 mg/kg/day in 2 doses |
|
Streptococcus pneumonia Haemophilus influenza |
|
Azithromycin |
10 mg/kg on day 1, followed by 5 mg/kg/day once daily on days 2–5 |
Maximum of 500 mg on day 1, followed by 250 mg on days 2–5 |
Mycoplasma pneumonia Chlamydia trachomatis or Chlamydia pneumonia |
|
Clarithromycin or oral |
15 mg/kg/day in 2 doses for 7-14 days |
Maximum of 1 g/day |
Mycoplasma pneumonia Chlamydia trachomatis or Chlamydia pneumonia |
|
Erythromycin |
40 mg/kg/day in 4 doses |
|
Mycoplasma pneumonia Chlamydia trachomatis or Chlamydia pneumonia |
|
Doxycycline |
2-4 mg/kg/day in 2 doses |
Children more than7 years old |
Mycoplasma pneumonia Chlamydia trachomatis or Chlamydia pneumonia |
|
Second- or third- generation cephalosporin (cefpodoxime, cefuroxime, cefprozil) |
|
|
Streptococcus pneumonia Haemophilus influenza |
|
Cephalexin |
75–100 mg/kg/day in 3 or 4 doses |
|
Staphylococcus aureus, methicillin susceptible |
|
Levofloxacin |
16–20 mg/kg/day in 2 doses for children 6 months to 5 years old 8–10 mg/kg/day once daily for children 5 to 16 years old |
Maximum daily dose, 750 mg |
Streptococcus pneumonia S. pneumoniae resistant to penicillin Mycoplasma pneumonia Chlamydia trachomatis or Chlamydia pneumonia |
|
Moxifloxacin |
400 mg once daily |
For adolescents with skeletal maturity, |
Mycoplasma pneumonia Chlamydia trachomatis or Chlamydia pneumonia |
|
Linezolid |
30 mg/kg/day in 3 doses for children less than12 years old 20 mg/kg/day in 2 doses for children more than12 years old |
|
Streptococcus pneumoniae S. pneumoniae resistant to penicillin S. aureus, methicillin resistant, susceptible to clindamycin Aureus, methicillin resistant, resistant to clindamycin |
|
Clindamycin |
30–40 mg/kg/day in 3 doses |
Resistance appears to be increasing in certain geographic areas among S. pneumoniae and S. aureus infections. |
S. pneumoniae resistant to Penicillin Group A Streptococcus Staphylococcus aureus, methicillin susceptible S. aureus, methicillin resistant, susceptible to clindamycin |
|
Oseltamivir |
0–8 months old: 6 mg/kg/day in 2 doses; premature infants: 2 mg/kg/day in 2 doses
9–23 months old: 7 mg/kg/day in 2 doses;
more than24 months old: 4 mg/kg/day in 2 doses |
For a 5-day course treatment |
|
Table 7. Parenteral Anti-infective Doses [31]
|
Antibiotics |
Doses |
Notes |
Sensitive Organisms |
|
Ampicillin |
150–200 mg/kg/day every 6 hours 300–400 mg/kg/day (every 6 hours) for S. pneumoniae resistant to penicillin |
|
Streptococcus pneumonia S. pneumoniae resistant to penicillin Group A Streptococcus Haemophilus influenza |
|
Penicillin |
200 000–250 000 U/kg/day every 4–6 h |
|
Streptococcus pneumoniae Group A Streptococcus |
|
Ceftriaxone or
Cefotaxime |
50–100 mg/kg/day every 12–24 hours
150 mg/kg/day every 8 hours |
|
Streptococcus pneumoniae S. pneumoniae resistant to penicillin Group A Streptococcus Haemophilus influenza, |
|
Clindamycin |
40 mg/kg/day every 6–8 hours |
|
Streptococcus pneumoniae S. pneumoniae resistant to penicillin Group A Streptococcus S. aureus, methicillin susceptible S. aureus, methicillin resistant, susceptible to clindamycin |
|
Vancomycin |
40–60 mg/kg/day every 6–8 hours |
|
Streptococcus pneumonia S. pneumoniae resistant to penicillin Group A Streptococcus S. aureus, methicillin susceptible S. aureus, methicillin resistant, susceptible to clindamycin S. aureus, methicillin resistant, resistant to clindamycin |
|
Levofloxacin |
6 months to 5 years: 16–20 mg/kg/day every 12 5–16 years: 8–10 mg/kg/day once daily |
maximum daily dose, 750 mg) |
S. pneumoniae resistant to penicillin Haemophilus influenza Mycoplasma pneumonia Chlamydia trachomatis or Chlamydia pneumonia |
|
Linezolid |
<12 years:30 mg/kg/day every 8 hours
More than 12 years : 20 mg/kg/day |
|
S. pneumoniae resistant to penicillin S. aureus, methicillin resistant, susceptible to clindamycin S. aureus, methicillin resistant, resistant to clindamycin |
|
Cefazolin |
150 mg/kg/day every 8 Hours |
|
Staphylococcus aureus, methicillin susceptible |
|
Semisynthetic penicillin, e.g. oxacillin |
150–200 mg/kg/day every 6–8 hours |
|
Staphylococcus aureus, methicillin susceptible |
|
Ciprofloxacin |
30 mg/kg/day every 12 hours |
|
Haemophilus influenza |
|
Azithromycin |
10 mg/kg / day |
|
Mycoplasma pneumonia Chlamydia trachomatis or Chlamydia pneumonia |
NB: For the child allergic to amoxicillin (choice is depending on the antimicrobial susceptibility of the pathogen). Use alternatives as clindamycin, trimethoprim- sulfamethoxazole or macrolides.
