
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Prof. Abla Saleh Mostafa |
Professor of Pediatrics, Cairo University, Egypt |
None |
Not Applicable |
|
Prof. Amal Ibrahim Hassanain |
Professor of Child Health, National Research Center, Egypt |
None |
Not Applicable |
|
Prof. Dina Hossam-Eldine Hamed |
Ass. Prof. of Pediatrics, Cairo University, Egypt |
None |
Not Applicable |
|
Prof. Dina Tawfeek Sarhan |
Ass. Professor of Pediatrics, Zagazig University, Egypt |
None |
Not Applicable |
|
Prof. Eman Mahmoud Fouda |
Professor of Pediatrics, Ain Shams University, Egypt |
None |
Not Applicable |
|
Prof. Fekry Gharib Bassiouny |
Consultant of Pediatrics and Neonatology, MOHP, Egypt |
None |
Not Applicable |
|
Prof. Hala Gouda Elnady |
Professor of Child Health, National Research Center, Egypt |
None |
Not Applicable |
|
Prof. Hala Hamdi Shaaban |
Professor of Pediatrics, Cairo University, Egypt |
None |
Not Applicable |
|
Prof. Hoda M. Salah El Din Metwally |
Professor of Pediatrics, Faculty of medicine-Girls, Al-Azhar University, Egypt |
None |
Not Applicable |
|
Prof. Magda Hassab Allah Mohamed |
Professor of Pediatrics, Faculty of medicine Girls, Al-Azhar University, Egypt |
None |
Not Applicable |
|
Prof. Maysaa Abdallah Saeed |
Professor of Tropical medicine and infectious disease, Zagazig University, Egypt |
None |
Not Applicable |
|
Prof. Mohamed Mahmoud Rashad |
Professor of Pediatrics, Benha University, Egypt |
None |
Not Applicable |
|
Prof. Mona Mohsen Elattar |
Professor of Pediatrics, Cairo University, Egypt |
None |
Not Applicable |
|
Miss. Nevin Abdalah Kamel |
Head Nurse, Causality Department, Zagazig University Hospital, Egypt |
None |
Not Applicable |
|
Pharmacist, Sarah Naeem Bartella Hebish |
Senior clinical pharmacist, Pediatric oncology, ICU Department, National Cancer Institute, Cairo University, Egypt |
None |
Not Applicable |
|
Prof. Shahenaz Mohamoud Hussien |
Professor of Pediatrics, Al-Azhar University, Egypt |
None |
Not Applicable |
|
Prof. Tarek Hamed |
Professor of Pediatrics, Zagazig University, Egypt |
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Nanis Sulieman |
Associate Professor of Pediatrics Ain Shams University, Egypt |
None |
Not Applicable |
|
Dr. Ranin Soliman
|
1. Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. 2. Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. 3. Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
None |
Not applicable |
|
Dr. Lamis Mohsen Elsholkamy |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Ahmad Yousef |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
Prof. Karima Abd-Alkhalek |
Prof. of Pediatric pulmonology, Ain Shams University, Egypt |
None |
Not Applicable |
|
Prof. Zeinab Radwan |
Prof. of Pediatric pulmonology, Cairo University, Egypt |
None |
Not Applicable |
|
Prof. Tharwt Deraz |
Prof. of Pediatric pulmonology, Ain Shams University, Egypt |
None |
Not Applicable |
|
Prof. Ahmed Al Sawah |
Prof. of Pediatric pulmonology, Al-Azhar University, Egypt |
None |
Not Applicable |
➡️Web annexes
The following annexes can be added as a package of standalone supplementary documents.
Keywords: The MeSH terms for "Guideline for the prevention and management of community-acquired pneumonia in children " on PubMed are: Adolescent, Anti-Bacterial Agents / therapeutic use, Child, Child, Preschool, Community-Acquired Infections / diagnosis, Community-Acquired Infections / drug therapy, Community-Acquired Infections / microbiology, Community-Acquired Infections / prevention & control, Humans, Infant, Infant, Newborn, Pneumococcal Vaccines, Pneumonia / diagnosis, Pneumonia / drug therapy, Pneumonia / microbiology, Pneumonia / prevention & control, Practice Guidelines as Topic, and Vaccines, Conjugate, Child, Child, Preschool, Community-Acquired Infections / drug therapy, Humans, Pneumonia / therapy, Editorial Policies, Evidence-Based Medicine, Practice Guidelines as Topic / standards, and Reproducibility of Results.
The health questions included: 11 questions for diagnosis, 8 for treatment, and two for prevention of CAP in children (see recommendations table).
PIPOH Model Items
• P (patients, target population): This management pathway is intended primarily for use in previously healthy children aged 28 days -12 years presenting with community acquired pneumonia in primary health care centers, governmental hospitals private hospitals or private clinics. Exclusion: Hospital acquired pneumonia (HAP). Ventilator associated pneumonia (VAP), Patients who have underlying serious medical conditions which may affect their respiratory status (e.g. underlying lung disease, immunocompromised child, neuromuscular disorders), and Patients in need for PICU admission. Aspiration pneumonia.
• I (interventions and practices considered/ guidelines category): Community acquired pneumonia management (Diagnosis, treatment, and prevention).
• P (Professionals/ intended or target users and clinical specialties): Physicians: Pediatricians, Primary Health Care Physicians (PHC), Family Practitioners, nurses and clinical pharmacist.
• O (major outcomes considered): Primary outcome: Improvement of symptoms and decrease use of unnecessary medication in children with CAP. Secondary outcome: Improvement of patients’ outcome, decreased rate of hospital admission, decreased need to ICU admission with a net result of decreasing morbidity, mortality and optimizing health resources use.
• H (Healthcare settings): Primary health care centers, Pediatricians at hospitals, and Pediatricians in private clinics in the Egyptian Healthcare system.
Annex Table 2. Inclusion and Exclusion (Eligibility Criteria)
Inclusion/Exclusion Selection Criteria for BTS Guideline 2011
|
|
Include |
Exclude |
|
|
Methods of development |
√ |
----- |
Evidence- Based CPG |
|
√ |
----- |
Consensus -Based CPG(Expert opinion) |
|
|
Author(s) |
√ |
|
Organization |
|
----- |
----- |
Single author |
|
|
Country |
----- |
----- |
National |
|
√ |
----- |
International |
|
|
Date of publication |
2011 |
----- |
Range of years( preferably not older than 5 years) |
|
----- |
----- |
One year |
|
|
Language(s) |
√ |
----- |
English |
|
----- |
----- |
Arabic |
|
|
----- |
----- |
Other |
|
|
Status |
√ |
----- |
Original source CPG |
|
|
----- |
---- |
Adapted |
|
Comments |
It is developed in 2002 & scheduled to be reviewed every 3 years, but the last update was at 2011 |
||
Inclusion/Exclusion Selection Criteria for WHO Guideline 2012-2014
|
|
Include |
Exclude |
|
|
Methods of development |
√ |
------ |
Evidence- Based CPG |
|
--- |
------ |
Consensus -Based CPG(Expert opinion) |
|
|
Author(s) |
√ |
|
Organization |
|
----- |
----- |
Single author |
|
|
Country |
----- |
---- |
National |
|
√ |
----- |
International |
|
|
Date of publication |
2012-2014 |
--- |
Range of years( preferably not older than 5 years) |
|
-- |
---- |
One year |
|
|
Language(s) |
√ |
---- |
English |
|
------ |
---- |
Arabic |
|
|
----- |
----- |
Other |
|
|
Status |
√ |
----- |
Original source CPG |
|
|
--- |
---- |
Adapted |
|
Comments |
Revised at health facilities2014, it contains recommendations for treatment only doesn’t include recommendations for diagnosis or prevention |
||
Inclusion/Exclusion Selection Criteria for IDSA Guideline 2011
|
|
Include |
Exclude |
|
|
Methods of development |
√ |
------ |
Evidence- Based CPG |
|
√ |
------ |
Consensus -Based CPG(Expert opinion) |
|
|
Author(s) |
√ |
|
Organization |
|
----- |
----- |
Single author |
|
|
Country |
----- |
---- |
National |
|
√ |
----- |
International |
|
|
Date of publication |
2011 |
---- |
Range of years( preferably not older than 5 years) |
|
----- |
---- |
One year |
|
|
Language(s) |
√ |
---- |
English |
|
------ |
---- |
Arabic |
|
|
----- |
----- |
Other |
|
|
Status |
√ |
----- |
Original source CPG |
|
|
----- |
---- |
Adapted |
|
Comments |
|
||
Annex Table 3. Results of the AGREE II assessment of the three source guidelines for CAP.
|
AGREE II/ CPGs |
BTS |
IDSA |
WHO |
|
Domain 1 (Scope) |
90.74% |
92.59% |
77.78% |
|
Domain 2 (Stakeholder) |
83.33% |
88.88% |
76.39% |
|
Domain 3 (Rigour) |
81.25% |
77.77% |
76.56% |
|
Domain 4 (Clarity) |
94.44% |
72.22% |
81.95% |
|
Domain 5 (Applicability) |
84.72% |
34.72% |
70.83% |
|
Domain 6 (Independence) |
91.67% |
91.66% |
75% |
|
Overall assessment 1 |
83.33% |
83.33% |
70.83% |
|
Recommend for use (Overall assessment 2) |
Yes with modifications |
Yes with modifications |
Yes with modifications |
Annex Table 4. Annex Nurses and Parents Educational Guide in Arabic

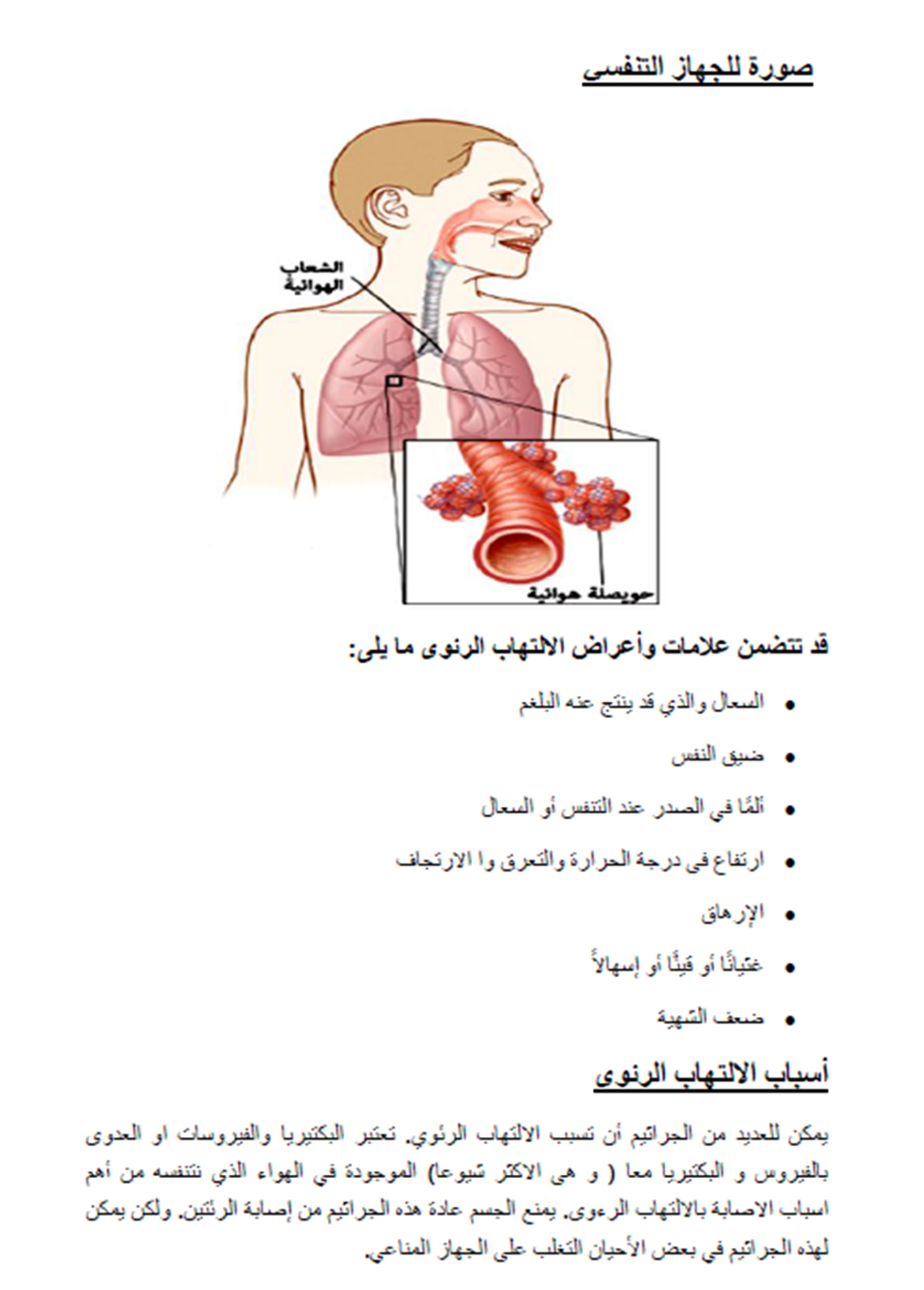



Appendix Table 5. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|
Guideline
Registration: PREPARE-2023CN225.

