- Recommendations
|
Table 3. Recommendations |
|
|
|||
|
A. Diagnosis of PAH deficiency in children |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
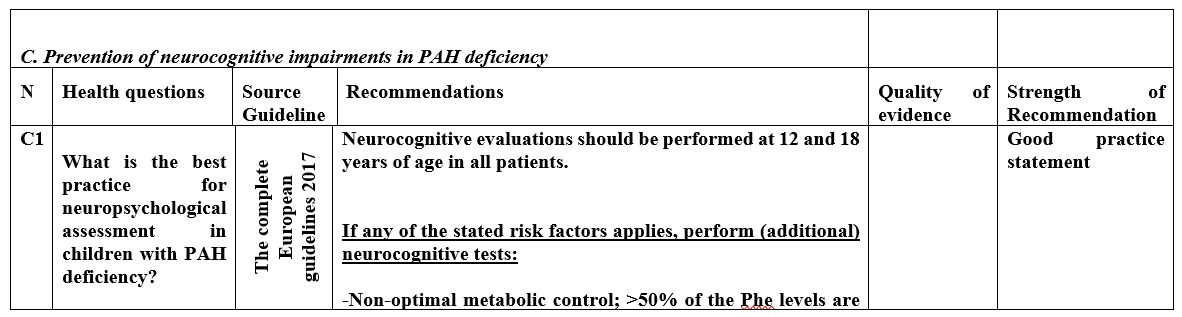
A1 |
What is the best clinical practice in diagnosis of asymptomatic neonates and symptomatic children with PAH deficiency? |
The complete European guidelines 2017 |
Newborn screening should be considered as a national obligation as this has proven to be cost effective.
|
Intermediate
|
strong |
|
In the differential diagnosis of hyperphenylalaninemia, of any degree, BH4 deficiency should be excluded by measuring petrins and dihydropetredine reductase activity in dried blood spot.
As these tests are not available in Egypt, careful follow up of patients neurodevelopment after positive newborn screening is highly recommended |
Low |
Weak (conditional)
|
|||
|
Good practice statement |
|||||
|
A2 |
What is the best clinical practice in classification of diagnosed PAH deficiency patients in pediatric age group?
|
The complete European guidelines 2017 |
To maintain blood levels in the recommended range, patients with PAH deficiency can be classified as either: A- Not requiring treatment B- Requiring diet, BH4 or both.
|
Very low |
Weak (conditional) |
|
A3 |
what is the role of molecular testing in management of diagnosed PAH deficiency in pediatric age group? |
The complete European guidelines 2017 |
Patient genotyping should be considered for diagnosing BH4 responsiveness and may help to define the metabolic phenotype and could help in prevention by offering reproductive options.
|
Very low |
Weak (conditional) |
|
A. Management of PAH deficiency in children |
|
|
|||
|
N |
Health questions |
Source Guideline |
Recommendations |
Quality of evidence |
Strength of Recommendation |
|
B1 |
What is the Phe level and timing at which dietary treatment should be started in asymptomatic neonates and in children with PAH deficiency? (figure (1)
|
The complete European guidelines 2017 |
All patients with untreated blood Phe levels >360 µmol/I should be treated.
Treatment should start as soon as possible, ideally before 10 days of age.
|
Very low |
Weak (conditional)
|
|
No treatment is recommended when untreated blood levels are <360 µmol/I. But, monitoring of blood Phe (at a lower frequency) until 1 year of age as a minimum is recommended to determine whether levels rise above 360 µmol/l. |
Very low |
Weak (conditional)
|
|||
|
Patients with untreated Phe levels 360-600 µmol/I should be treated until the age of 12 years. |
Very low |
Weak (conditional)
|
|||
|
B2 |
What is the target Phe level for optimal management of PAH deficiency in pediatrics age group? Figure (2)
|
The complete European guidelines 2017 |
In treated PKU patients up to the age of 12 years, target Phe level should be 120-360 µmol/L.
|
intermediate |
Strong |
|
In treated PKU patients aged ≥ 12 years target Phe level should be 120-600 µmol/L. |
Very low |
Weak (conditional) |
|||
|
B3 |
What is the best practice in monitoring in PAH deficiency in different pediatric age groups?
|
The complete European guidelines 2017 |
Blood Phe levels should be measured to monitor metabolic control, as they are the most clinically relevant biomarkers
|
intermediate |
Strong |
|
There is insufficient evidence to support routine evaluation of the Phe fluctuations and the measurement of Phe/tyr ratio in PKU
|
Very low |
Weak (conditional) |
|||
|
Frequency of blood PHE measurement should be at minimum: 0-1 year Weekly 1-12 years fortnightly >12 years monthly
|
|
Good practice statement |
|||
|
B4 |
What is the best practice in monitoring growth and development in patients with PAH deficiency in pediatric age group?
|
The complete European guidelines 2017 |
All PKU patients should be followed up in a specialized metabolic center
|
Very low |
Weak (conditional) |
|
An annual nutritional review is required for any patient who is prescribed in low Phe diet or is self-restricting high protein food. Such a clinical review includes a clinical examination including the anthropometric parameters (weight, height, BMI).
It is also recommended that plasma amino acids, plasma homocysteine, and /or methylmalonic acid, hemoglobin, MCV and ferritin are measured.
all other micronutrients (vitamins, minerals including calcium, zinc, selenium) or hormones (parathyroid hormones) can be considered if clinically indicated.
|
low |
Weak (conditional) |
|||
|
|
Outpatient clinic visit should be at minimum, given good clinical and metabolic control: 0-1 year: every 2 months 1-18 years: twice per year
|
|
Good practice statement |
||
|
|
|
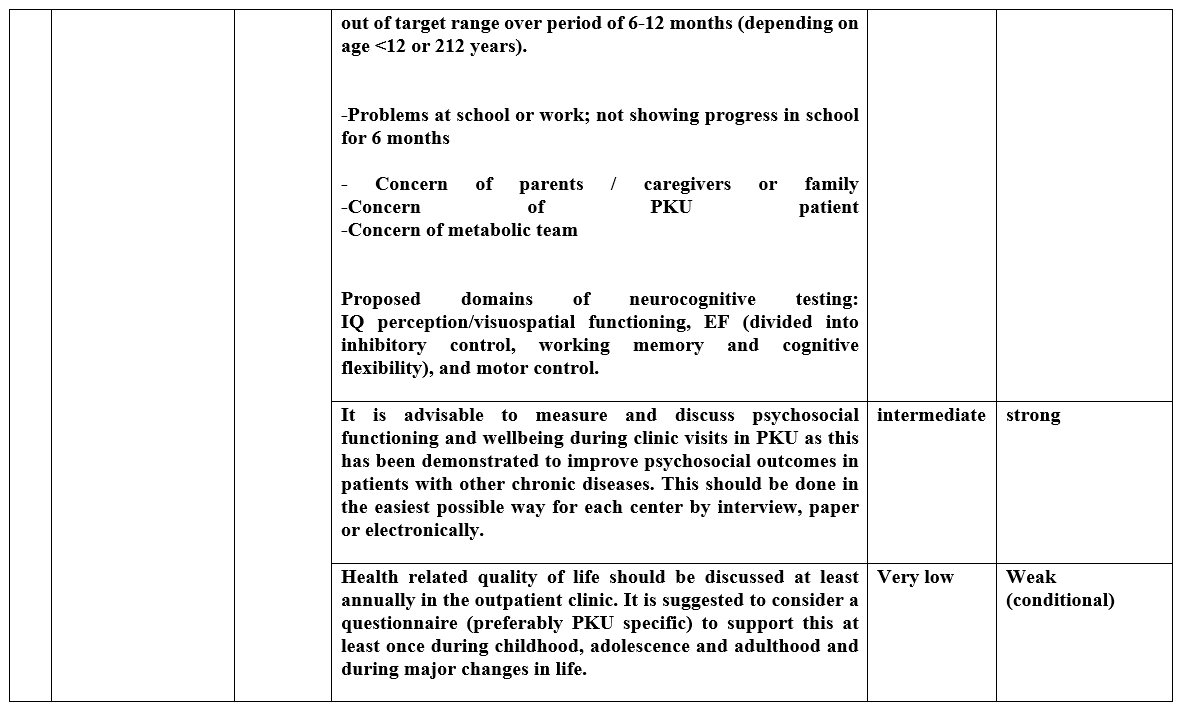
Health professional support is required throughout life to encourage normal healthy feeding behaviors, with a positive acceptance of a low Phe diet
|
low |
Weak (conditional) |
|
|
B5 |
What is the Phe, protein, and caloric requirements for children with PAH deficiency? |
The complete European guidelines 2017 |
Phe intake should be methodically titrated until Phe is consistently maintained within the Phe target range. If there is stability of control, a challenge with additional Phe should be performed systematically to maximize natural protein permitted, Phe deficiency should be avoided.
|
low |
Weak (conditional) |
|
Total protein intake should supply the age-related safe level of protein intake (FAQ/WHO/UNU 2007) with an additional 40% from L- amino acid supplements.
|
Low |
Weak (conditional)
|
|||
|
In PKU, the nutritional intake of energy, macronutrients and micronutrients, should meet the average estimated amounts/ dietary reference values for healthy population.
For all age groups, there should be focus on achieving balanced intake of all nutrients, avoiding catabolism or deficiency but preventing excess of any nutrient that may lead to over nutrition or toxicity.
Supplementary nutrients (vitamins, minerals and LC-PUFA's) added to Phe -free L-amino acid supplements should be in the amounts that will at least meet normal population dietary reference values.
Assessment of dietary intake should be performed in every clinic visit with extra attention directed to patients who are non-adherent, do not have prescribed Phe free L-amino acid supplements (with added micronutrients) or who are at the higher risk of nutritional deficiency
|
Low
|
Weak (conditional)
|
|||
|
|
|
|
Fruits and vegetables (except potatoes containing Phe ≤ 75 mg/100 g of food can be safely given without measurements or estimation in a low Phe-diet without loss of Phe control. Some unrestricted fruits and vegetables should be encouraged in the diet in early life to encourage long-term healthy feeding patterns.
|
Intermediate
|
strong
|
|
The artificial sweetener aspartame, particularly from beverages and tables top sweetener’s is best avoided in patients on a low Phe-diet.
|
Intermediate |
Strong |
|||
|
The Phe-free protein substitute, in the form of L-amino acids, should be provided in any patient with PKU treated with a low Phe diet consuming less than the FAQ/WHO/UNU 2007 safe levels of natural protein intake.
The Phe-free L-amino acid formula should be evenly administered at least 3 times throughout the day
To aid adherence, all patients with PKU should receive a choice of suitable age-appropriate Phe-free L-amino acid supplements.
|
Intermediate |
Strong |
|||
|
B6 |
What is the best practice for introducing breast milk in infants with confirmed PAH deficiency |
The complete European guidelines 2017 |
In infants with PKU Breast-Feeding in
|
low |
Weak (conditional) |
|
Breast feeding in infants diagnosed with phenylketonuria (PKU) 2023 |
Alternating Method: Alternate between breast-feeding and Phe-free L-amino acid infant formula bottle-feeding to achieve acceptable blood Phe control.
|
low |
Weak (conditional) |
||
|
B7 |
Is there specific dietary manipulation needed during management of PKU patients subjected to acute illness?
|
The complete European guidelines 2017 |
In children with PKU, during illness to prevent an excessive rise in blood Phe concentration, it is prudent to encourage the intake of Phe-free L-amino acid supplements and high carbohydrate supplements
|
Very low |
Weak (conditional) |