
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|
Interest identified |
Management plan & decision |
||
|
Prof. Solaf M Elsayed |
Professor of Medical Genetics, Medical Genetics Dept, Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Dr. Shaimaa Gad |
Lecturer of Medical Genetics,Medical Genetics Dept., Ain Shams University, Cairo, Egypt |
None |
Not Applicable |
|
Dr. Heba Allah Hosny |
Medical Genetics, national institute of Neuromotor System, Cairo, Egypt |
None |
Not Applicable |
|
Dr. Rana Mahrous |
Researcher of Medical genetics, National Research Centre, Cairo, Egypt |
None |
Not Applicable |
|
Guideline Adaptation Group (Methodology Subgroup) |
|||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
Dr. Nanis Sulieman |
Associate Professor of Pediatrics Ain Shams University, Egypt |
None |
Not Applicable |
|
Dr. Ranin Soliman
|
1. Assistant Professor of Evidence-based Practice, School of Life and Medical Sciences, University of Hertfordshire, Egypt. 2. Consultant at WHO/EMRO for the Clinical and Public Heath Guideline Adaptation Project in the EMR. 3. Head of Heath Economics and Value Unit, Children’s Cancer Hospital Egypt. |
None |
Not applicable |
|
Dr. Lamis Mohsen Elsholkamy |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Ahmad Yousef |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
Dr. Mona Saber |
Lecturer of Pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
External Review Group |
|||
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
External Reviewer for methodology |
|||
|
|
|
|
|
|
International Peer Reviewers |
|
|
|
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
|
|
|
None |
Not Applicable |
Web annexes
The following annexes can be added as a package of standalone supplementary documents.
➡️Keywords: The MeSH terms for "Guideline for PAH deficiency in children. " on PubMed are: ……Phenylketonuria, phenylalanine hydroxylase deficiency, PKU treatment, PKU management .
Annex Table 2. Results of the AGREE II assessment of the three source guidelines for The complete European guidelines on phenylketonuria: diagnosis and treatment.
|
AGREE II/ CPGs |
………… |
|
Domain 1 (Scope) |
%93 |
|
Domain 2 (Stakeholder) |
%92 |
|
Domain 3 (Rigour) |
%94 |
|
Domain 4 (Clarity) |
%96 |
|
Domain 5 (Applicability) |
%70 |
|
Domain 6 (Independence) |
%100 |
|
Overall assessment. |
%100 |
|
Recommend for use (Overall assessment .) |
YES |
Results of the AGREE II assessment of the three source guidelines for Epilepsy and vaccinations: Italian guidelines.
|
AGREE II/ CPGs |
………… |
|
Domain 1 (Scope) |
%61 |
|
Domain 2 (Stakeholder) |
%37 |
|
Domain 3 (Rigour) |
%65 |
|
Domain 4 (Clarity) |
%89 |
|
Domain 5 (Applicability) |
%22 |
|
Domain 6 (Independence) |
%86 |
|
Overall assessment. |
%83 |
|
Recommendation for use (Overall assessment .) |
YES, with modifications |
PRISMA-ScR-scoping review-Checklist, Breast feeding in infants diagnosed with phenylketonuria (PKU) a scoping review.
|
SECTION |
ITEM |
PRISMA-ScR CHECKLIST ITEM |
REPORTED ON PAGE # |
|
TITLE |
|||
|
Title |
1 |
Identify the report as a scoping review. |
P1 |
|
ABSTRACT |
|||
|
Structured summary |
2 |
Provide a structured summary that includes (as applicable): background, objectives, eligibility criteria, sources of evidence, charting methods, results, and conclusions that relate to the review questions and objectives. |
P1 |
|
INTRODUCTION |
|||
|
Rationale |
3 |
Describe the rationale for the review in the context of what is already known. Explain why the review questions/objectives lend themselves to a scoping review approach. |
P1 |
|
Objectives |
4 |
Provide an explicit statement of the questions and objectives being addressed with reference to their key elements (e.g., population or participants, concepts, and context) or other relevant key elements used to conceptualize the review questions and/or objectives. |
P1 |
|
METHODS |
|||
|
Protocol and registration |
5 |
Indicate whether a review protocol exists; state if and where it can be accessed (e.g., a Web address); and if available, provide registration information, including the registration number. |
Methods P2 |
|
Eligibility criteria |
6 |
Specify characteristics of the sources of evidence used as eligibility criteria (e.g., years considered, language, and publication status), and provide a rationale. |
Methods P2 |
|
Information sources* |
7 |
Describe all information sources in the search (e.g., databases with dates of coverage and contact with authors to identify additional sources), as well as the date the most recent search was executed. |
Methods P2 till September 22 |
|
Search |
8 |
Present the full electronic search strategy for at least 1 database, including any limits used, such that it could be repeated. |
Methods P2 |
|
Selection of sources of evidence† |
9 |
State the process for selecting sources of evidence (i.e., screening and eligibility) included in the scoping review. |
Methods P2 |
|
Data charting process‡ |
10 |
Describe the methods of charting data from the included sources of evidence (e.g., calibrated forms or forms that have been tested by the team before their use, and whether data charting was done independently or in duplicate) and any processes for obtaining and confirming data from investigators. |
Methods P2 |
|
Data items |
11 |
List and define all variables for which data were sought and any assumptions and simplifications made. |
P4 |
|
Critical appraisal of individual sources of evidence§ |
12 |
If done, provide a rationale for conducting a critical appraisal of included sources of evidence; describe the methods used and how this information was used in any data synthesis (if appropriate). |
Not available |
|
Synthesis of results |
13 |
Describe the methods of handling and summarizing the data that were charted. |
Results P3 |
|
RESULTS |
|||
|
Selection of sources of evidence |
14 |
Give numbers of sources of evidence screened, assessed for eligibility, and included in the review, with reasons for exclusions at each stage, ideally using a flow diagram. |
Methods P4 |
|
Characteristics of sources of evidence |
15 |
For each source of evidence, present characteristics for which data were charted and provide the citations. |
Not available |
|
Critical appraisal within sources of evidence |
16 |
If done, present data on critical appraisal of included sources of evidence (see item 12). |
Not available |
|
Results of individual sources of evidence |
17 |
For each included source of evidence, present the relevant data that were charted that relate to the review questions and objectives. |
P4 |
|
Synthesis of results |
18 |
Summarize and/or present the charting results as they relate to the review questions and objectives. |
Results P5 |
|
DISCUSSION |
|||
|
Summary of evidence |
19 |
Summarize the main results (including an overview of concepts, themes, and types of evidence available), link to the review questions and objectives, and consider the relevance to key groups. |
P5 |
|
Limitations |
20 |
Discuss the limitations of the scoping review process. |
Limitation of the study P5 |
|
Conclusions |
21 |
Provide a general interpretation of the results with respect to the review questions and objectives, as well as potential implications and/or next steps. |
Discussion P6 |
|
FUNDING |
|||
|
Funding |
22 |
Describe sources of funding for the included sources of evidence, as well as sources of funding for the scoping review. Describe the role of the funders of the scoping review. |
Funding for the scoping review P7 |
JBI = Joanna Briggs Institute; PRISMA-ScR = Preferred Reporting Items for Systematic reviews and Meta-Analyses extension for Scoping Reviews.
* Where sources of evidence (see second footnote) are compiled from, such as bibliographic databases, social media platforms, and Web sites.
† A more inclusive/heterogeneous term used to account for the different types of evidence or data sources (e.g., quantitative and/or qualitative research, expert opinion, and policy documents) that may be eligible in a scoping review as opposed to only studies. This is not to be confused with information sources (see first footnote).
‡ The frameworks by Arksey and O’Malley (6) and Levac and colleagues (7) and the JBI guidance (4, 5) refer to the process of data extraction in a scoping review as data charting.
§ The process of systematically examining research evidence to assess its validity, results, and relevance before using it to inform a decision. This term is used for items 12 and 19 instead of "risk of bias" (which is more applicable to systematic reviews of interventions) to include and acknowledge the various sources of evidence that may be used in a scoping review (e.g., quantitative and/or qualitative research, expert opinion, and policy document).
From: Tricco AC, Lillie E, Zarin W, O'Brien KK, Colquhoun H, Levac D, et al. PRISMA Extension for Scoping Reviews (PRISMAScR): Checklist and Explanation. Ann Intern Med. 2018;169:467–473. doi: 10.7326/M18-0850.
Annex Table 3. Annex Nurses and Parents Educational Guide in Arabic
Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|
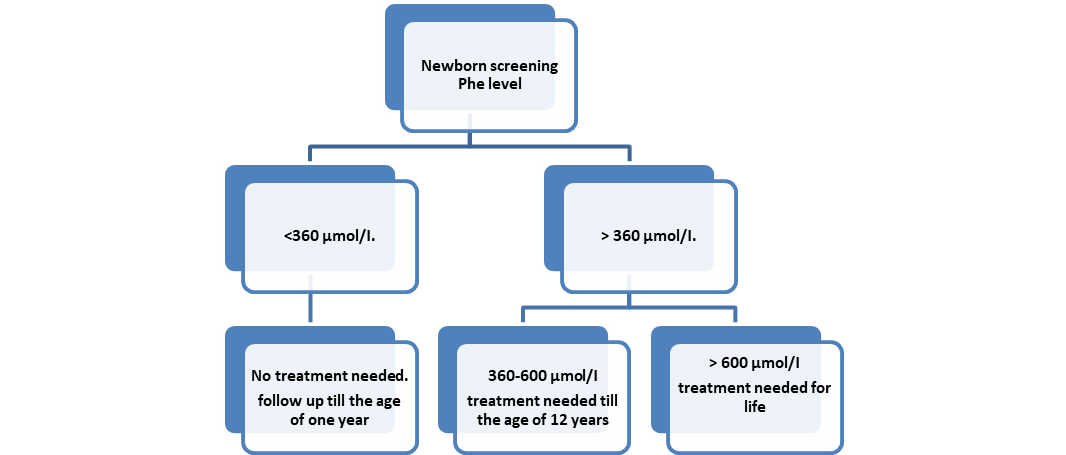
Figure (1): Treatment of PAH deficiency according to Phe level
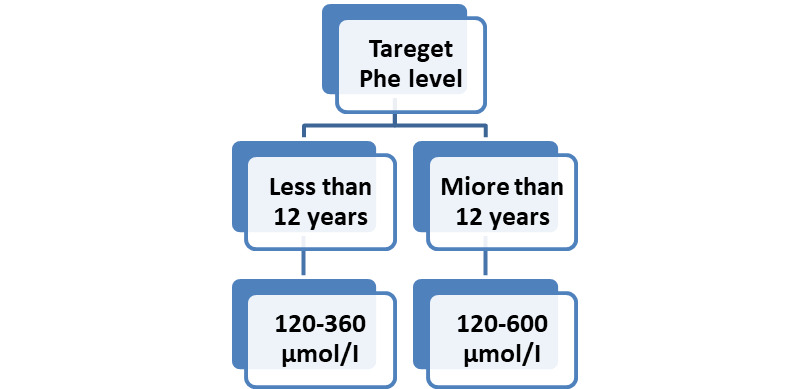
Figure (2): Target Phe levels for optimal management in PAH deficiency
