- Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing strategies for the diagnosis, prevention and management of thrombocytopenia in pediatric age group into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
▪️ Select the target populations and evaluate the outcome.
▪️ Identify the local resources to support the implementation.
▪️ Set timelines.
▪️ Distribute the tasks to the members.
▪️ Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
▪️ Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
▪️ Educational materials: printed or electronic information (software).
▪️ Web-based education: computer-based educational activities.
▪️ A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
▪️ Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
▪️ Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
▪️ Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
▪️ Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
▪️ Mass media campaigns.
3. For Nurses
▪️ Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
▪️ Educational materials: printed.
▪️ A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
▪️ Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
▪️ Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
▪️ Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
▪️ Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
▪️ Administrative policies and procedures.
▪️ Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
▪️ International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
▪️ Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for treatment of CAP in children have been made available in several forms including: algorithms, pathways, tables, and parents’ educational health guide (in Arabic).
Table S1: Modified WHO Bleeding assessment score
Table S2. Causes of neonatal thrombocytopenia 45
|
|
Ill-appearing, Premature |
Well-appearing, mature |
||
|
|
Early Onset |
Late Onset |
Early Onset |
Late Onset |
|
Common |
Sepsis TORCH infection Birth asphyxia DIC NEC |
Sepsis Thrombosis DIC NEC Drug-induced |
Placental insufficiency Autoimmune Alloimmune (FNAIT) Occult infection |
Occult infection NEC |
|
Rare |
Chromosomal disorders • Trisomy 13 • Trisomy 18 • Trisomy 21 • Turner syndrome |
Inborn errors of metabolism Fanconi anemia |
Inherited syndromes • Bernard-Soulier • Wiskott-Aldrich • Thrombocytopenia absent radii • Others Vascular tumors • Kasabach-Merritt |
Inborn errors of metabolism Fanconi anemia |
Sillers L, Van Slambrouck C, Lapping-Carr G. Neonatal Thrombocytopenia: Etiology and Diagnosis. Pediatr Ann. 2015 Jul;44(7):e175-80. doi: 10.3928/00904481-20150710-11. PubMed PMID: 26171707; PubMed Central PMCID: PMCPMC6107300.
Table S3. Suggested thresholds of platelet count for neonatal platelet transfusion 46
|
Platelet count (× 109/l) |
Indication for platelet transfusion |
|
<25 |
Neonates with no bleeding (including neonates with NAIT if no bleeding and no family history of ICH) |
|
<50 |
Neonates with bleeding, current coagulopathy, before surgery, or infants with NAIT if previously affected sibling with ICH |
|
<100 |
Neonates with major bleeding or requiring major surgery (e.g., neurosurgery) |
NAIT, neonatal alloimmune thrombocytopenia; ICH, intracranial hemorrhage.
New HV, Berryman J, Bolton-Maggs PH, Cantwell C, Chalmers EA, Davies T, Gottstein R, Kelleher A, Kumar S, Morley SL, Stanworth SJ; British Committee for Standards in Haematology. Guidelines on transfusion for fetuses, neonates and older children. Br J Haematol. 2016 Dec;175(5):784-828. doi: 10.1111/bjh.14233. Epub 2016 Nov 11. PMID: 27861734.
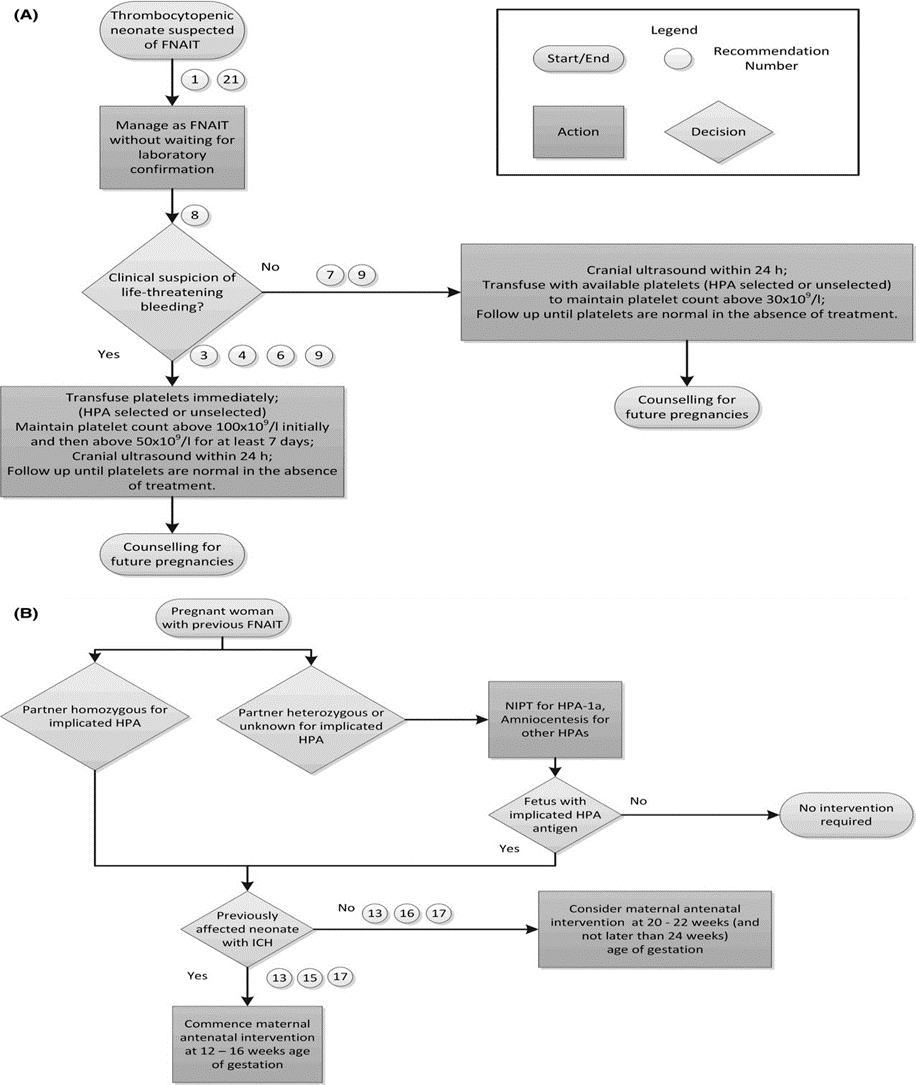
Figure S1: (A) Postnatal Algorithm for FNAIT. (B) Antenatal Algorithm for FNAIT 47
FNAIT, fetal and neonatal alloimmune thrombocytopenia; HLA, human leucocyte antigen; ICH, intracranial hemorrhage; NIPT, non-invasive prenatal testing.
Lieberman L, Greinacher A, Murphy MF, Bussel J, Bakchoul T, Corke S, Kjaer M, Kjeldsen-Kragh J, Bertrand G, Oepkes D, Baker JM, Hume H, Massey E, Kaplan C, Arnold DM, Baidya S, Ryan G, Savoia H, Landry D, Shehata N; International Collaboration for Transfusion Medicine Guidelines (ICTMG). Fetal and neonatal alloimmune thrombocytopenia: recommendations for evidence-based practice, an international approach. Br J Haematol. 2019 May;185(3):549-562. doi: 10.1111/bjh.15813. Epub 2019 Mar 3. PMID: 30828796.
Table S4. ISTH-SSC Bleeding Assessment Tool 48
|
|
SCORE |
||||
|
SYMPTOMS (up to the time of diagnosis) |
0§ |
1§ |
2 |
3 |
4 |
|
Epistaxis |
No/trivial |
- > 5/year |
Consultation only* |
Packing or cauterization or antifibrinolytic |
Blood transfusion or replacement therapy (use of hemostatic blood components and rFVIIa) or desmopressin |
|
Cutaneous |
No/trivial |
For bruises 5 or more (> 1cm) in exposed areas |
Consultation only* |
Extensive |
Spontaneous hematoma requiring blood transfusion |
|
Bleeding from minor wounds |
No/trivial |
- > 5/year |
Consultation only* |
Surgical hemostasis |
Blood transfusion, replacement therapy, or desmopressin |
|
Oral cavity |
No/trivial |
Present |
Consultation only* |
Surgical hemostasis or antifibrinolytic |
Blood transfusion, replacement therapy or desmopressin |
|
GI bleeding |
No/trivial |
Present (not associated with ulcer, portal hypertension, hemorrhoids, angiodysplasia) |
Consultation only* |
Surgical hemostasis, antifibrinolytic |
Blood transfusion, replacement therapy or desmopressin |
|
Hematuria |
No/trivial |
Present (macroscopic) |
Consultation only* |
Surgical hemostasis, iron therapy |
Blood transfusion, replacement therapy or desmopressin |
|
Tooth extraction |
No/trivial or none done |
Reported in ≤25% of all procedures, no intervention** |
Reported in ≥25% of all procedures, no intervention** |
Resuturing or packing |
Blood transfusion, replacement therapy or desmopressin |
|
Surgery |
No/trivial or none done |
Reported in ≤25% of all procedures, no intervention** |
Reported in ≥25% of all procedures, no intervention** |
Surgical hemostasis or antifibrinolytic |
Blood transfusion, replacement therapy or desmopressin |
|
Menorrhagia |
No/trivial |
Consultation only* |
- Time off work/school > 2/year |
- Requiring combined treatment with
antifibrinolytics and hormonal therapy |
- Acute menorrhagia requiring hospital admission and
emergency treatment |
|
Post-partum hemorrhage |
No/trivial or no deliveries |
Consultation only* |
- Iron therapy |
- Requiring blood transfusion, replacement therapy,
desmopressin |
- Any procedure requiring critical care or surgical intervention (e.g. hysterectomy, internal iliac artery legation, uterine artery embolization, uterine brace sutures) |
|
Muscle hematomas |
Never |
Post trauma, no therapy |
Spontaneous, no therapy |
Spontaneous or traumatic, requiring desmopressin or replacement therapy |
Spontaneous or traumatic, requiring surgical intervention or blood transfusion |
|
Hemarthrosis |
Never |
Post trauma, no therapy |
Spontaneous, no therapy |
Spontaneous or traumatic, requiring desmopressin or replacement therapy |
Spontaneous or traumatic, requiring surgical intervention or blood transfusion |
|
CNS bleeding |
Never |
|
|
Subdural, any intervention |
Intracerebral, any intervention |
|
Other bleedings^ |
No/trivial |
Present |
Consultation only* |
Surgical hemostasis, antifibrinolytics |
Blood transfusion or replacement therapy or desmopressin |
In addition to the guidance offered by the table, it is mandatory to refer to the text for more detailed instructions.
§ Distinction between 0 and 1 is of critical importance. Score 1 means that the symptom is judged as present in the patient’s history by the interviewer but does not qualify for a score 2 or more
* Consultation only: the patient sought medical evaluation and was either referred to a specialist or offered detailed laboratory investigation.
** Example: 1 extraction/surgery resulting in bleeding (100%): the score to be assigned is 2; 2 extractions/surgeries, 1 resulting in bleeding (50%): the score to be assigned is 2; 3 extractions/surgeries, 1 resulting in bleeding (33%): the score to be assigned is 2; 4 extractions/surgeries, 1 resulting in bleeding (25%): the score to be assigned is 1.
# If already available at the time of collection.
^ Include: umbilical stump bleeding, cephalohematoma, cheek hematoma caused by sucking during breast/bottle feeding, conjunctival hemorrhage or excessive bleeding following circumcision or venipuncture. Their presence in infancy requires detailed investigation independently from the overall score
Normal range is <4 in adult males, <6 in adult females and <3 in children (3).
Rodeghiero F, Tosetto A, Abshire T, Arnold DM, Coller B, James P, Neunert C, Lillicrap D; ISTH/SSC joint VWF and Perinatal/Pediatric Hemostasis Subcommittees Working Group. ISTH/SSC bleeding assessment tool: a standardized questionnaire and a proposal for a new bleeding score for inherited bleeding disorders. J Thromb Haemost. 2010 Sep;8(9):2063-5. doi: 10.1111/j.1538-7836.2010.03975.x. PMID: 20626619.
Table S5. Differential diagnosis of thrombocytopenia 49
|
Previously diagnosed or possible high risk for conditions that may be associated with immune thrombocytopenia (e.g., infections [HIV, HCV, HBV]), autoimmune/immunodeficiency disorders (CVID, systemic lupus erythematosus, or APS), and malignancy (eg, lymphoproliferative disorders) |
|
Liver disease (including cirrhosis or portal hypertension) |
|
Splenomegaly |
|
Drugs (prescription or nonprescription), including heparin, alemtuzumab, PD-1 inhibitors, abciximab, valproate, alcohol abuse, consumption of quinine (tonic water), exposure to environmental toxins, or chemotherapy |
|
Bone marrow diseases, including myelodysplastic syndromes, leukemias, other malignancies, metastatic disease, myelofibrosis, aplastic anemia, megaloblastic anemia, myelophthisis, and Gaucher disease |
|
Recent transfusions (rare possibility of posttransfusion purpura) and recent vaccinations |
|
Inherited thrombocytopenia: TAR syndrome, radioulnar synostosis, congenital amegakaryocytic thrombocytopenia, Wiskott-Aldrich syndrome, MYH9-related disease, Bernard-Soulier syndrome, type IIB VWD, or platelet-type VWD |
|
Other thrombocytopenic disorders (DIC, TTP, HUS, Evans syndrome) |
APS, antiphospholipid syndrome; CVID, common variable immunodeficiency; DIC, disseminated intravascular coagulation; HUS, hemolytic-uremic syndrome; MYH9, myosin heavy chain 9; PD-1, programmed cell death protein 1; TAR, thrombocytopenia-absent radius; TTP, thrombotic thrombocytopenic purpura; VWD, von Willebrand disease.
Provan D, Arnold DM, Bussel JB, Chong BH, Cooper N, Gernsheimer T, Ghanima W, Godeau B, González-López TJ, Grainger J, Hou M, Kruse C, McDonald V, Michel M, Newland AC, Pavord S, Rodeghiero F, Scully M, Tomiyama Y, Wong RS, Zaja F, Kuter DJ. Updated international consensus report on the investigation and management of primary immune thrombocytopenia. Blood Adv. 2019 Nov 26;3(22):3780-3817. doi: 10.1182/bloodadvances.2019000812. PMID: 31770441; PMCID: PMC6880896.
Table S6 Bleeding assessment tool in pediatric patient with primary immune thrombocytopenia 49
|
Grade |
Bleeding |
Management approach |
|
Grade 1 (minor) |
Minor bleeding, few petechiae (≤100 total) and/or ≤5 small bruises (≤3 cm in diameter), no mucosal bleeding |
Consent for observation |
|
Grade 2 (mild) |
Mild bleeding, many petechiae (>100 total) and/or >5 large bruises (>3 cm in diameter), no mucosal bleeding |
Consent for observation |
|
Grade 3 (moderate) |
Moderate bleeding, overt mucosal bleeding, troublesome lifestyle |
Intervention to reach grade 1 or 2 |
|
Grade 4 (severe) |
Severe bleeding, mucosal bleeding leading to decrease in Hb > 2 g/dL or suspected internal hemorrhage |
Intervention |
This bleeding scale is based on the one used in the previous consensus report,1 updated based on the authors’ opinion. Hb, hemoglobin.
Provan D, Arnold DM, Bussel JB, Chong BH, Cooper N, Gernsheimer T, Ghanima W, Godeau B, González-López TJ, Grainger J, Hou M, Kruse C, McDonald V, Michel M, Newland AC, Pavord S, Rodeghiero F, Scully M, Tomiyama Y, Wong RS, Zaja F, Kuter DJ. Updated international consensus report on the investigation and management of primary immune thrombocytopenia. Blood Adv. 2019 Nov 26;3(22):3780-3817. doi: 10.1182/bloodadvances.2019000812. PMID: 31770441; PMCID: PMC6880896
Table S7 Recommendations for the diagnosis of ITP in children and adults 49
|
Basic evaluation in all patients |
Tests of potential utility in the management of an ITP patient |
Tests of unproven or uncertain benefit* |
|
Patient history |
Glycoprotein-specific antibody (can be used in difficult cases, has poor sensitivity, and is not a primary diagnostic test) |
TPO level |
|
Family history |
Anti-phospholipid antibodies (including anti-cardiolipin and lupus anticoagulant) if there are clinical features of antiphospholipid syndrome |
Reticulated platelets/immature platelet fraction |
|
Physical examination |
Anti-thyroid antibodies and thyroid function |
|
|
CBC and reticulocyte count |
Pregnancy test in women of childbearing potential |
Bleeding time |
|
Peripheral blood film |
Antinuclear antibodies |
Serum complement |
|
Quantitative Ig level measurement† |
Viral PCR for EBV, CMV, and parvovirus |
|
|
Blood group (Rh) |
Bone marrow examination (in selected patients; refer to text) |
|
|
HIV‡ |
Direct antiglobulin test |
|
|
HCV‡ |
H pylori‡ |
|
|
HBV |
|
|
CMV, cytomegalovirus; EBV, Epstein-Barr virus; PCR, polymerase chain reaction; PTT, partial thromboplastin time; Rh, rhesus; TPO, thrombopoietin.
*These tests have no proven role in the differential diagnosis of ITP from other thrombocytopenias and do not guide patient management.
†Quantitative Ig level measurement should be considered in children with ITP and is recommended in children with persistent or chronic ITP as part of the reassessment evaluation.
‡Recommended by the majority of the panel for adult patients in the appropriate geographic setting.
Provan D, Arnold DM, Bussel JB, Chong BH, Cooper N, Gernsheimer T, Ghanima W, Godeau B, González-López TJ, Grainger J, Hou M, Kruse C, McDonald V, Michel M, Newland AC, Pavord S, Rodeghiero F, Scully M, Tomiyama Y, Wong RS, Zaja F, Kuter DJ. Updated international consensus report on the investigation and management of primary immune thrombocytopenia. Blood Adv. 2019 Nov 26;3(22):3780-3817. doi: 10.1182/bloodadvances.2019000812. PMID: 31770441; PMCID: PMC6880896.
Table S8: Therapies for the treatment of ITP 49
|
Clinical situation |
Therapy option |
|
Initial treatment of newly diagnosed ITP |
Corticosteroids |
|
Dexamethasone |
|
|
Methylprednisolone |
|
|
Prednis(ol)one |
|
|
IVIg |
|
|
Anti-D (licensed and available for ITP in only a few countries) |
|
|
Subsequent treatment |
Medical therapies |
|
Medical therapies with robust evidence |
|
|
Rituximab |
|
|
TPO-RAs: eltrombopag, avatrombopag, romiplostim |
|
|
Fostamatinib |
|
|
Medical therapies with less robust evidence |
|
|
Azathioprine |
|
|
Cyclophosphamide |
|
|
Cyclosporine A |
|
|
Danazol |
|
|
Dapsone |
|
|
Mycophenolate mofetil |
|
|
TPO-RA switch |
|
|
Vinca alkaloids |
|
|
Surgical therapies |
|
|
Splenectomy |
|
|
Treatment of patients failing multiple therapies |
Accessory splenectomy |
|
Alemtuzumab |
|
|
Combination of initial and subsequent therapies |
|
|
Combination chemotherapy |
|
|
Clinical trials |
|
|
HSCT |
|
|
Splenectomy, if not already performed |
|
|
Supportive care |
Treatment options for ITP are listed in alphabetical order and do not imply a preferred treatment option.
HSCT, hematopoietic stem cell transplantation; TPO-RA, TPO receptor agonist.
Provan D, Arnold DM, Bussel JB, Chong BH, Cooper N, Gernsheimer T, Ghanima W, Godeau B, González-López TJ, Grainger J, Hou M, Kruse C, McDonald V, Michel M, Newland AC, Pavord S, Rodeghiero F, Scully M, Tomiyama Y, Wong RS, Zaja F, Kuter DJ. Updated international consensus report on the investigation and management of primary immune thrombocytopenia. Blood Adv. 2019 Nov 26;3(22):3780-3817. doi: 10.1182/bloodadvances.2019000812. PMID: 31770441; PMCID: PMC6880896
Table S9. Suggested thresholds of platelet count for platelet transfusion in children with non-immune thrombocytopenia 50
|
Platelet count (× 109/l) |
Clinical situation to trigger platelet transfusion |
|
<10 |
Irrespective of signs of hemorrhage (excluding ITP, TTP/HUS, HIT) |
|
<20 |
Severe mucositis Sepsis Laboratory evidence of DIC in the absence of bleedinga Anticoagulant therapy Risk of bleeding due to a local tumor infiltration Insertion of a non-tunnelled central venous line |
|
<40 |
Prior to lumbar punctureb |
|
<50 |
Moderate hemorrhage (e.g., gastrointestinal bleeding) including bleeding in association with DIC Surgery, unless minor (except at critical sites)
|
|
<75–100 |
Major hemorrhage or significant post-operative bleeding (e.g., post cardiac surgery) Surgery at critical sites: central nervous system including eyes |
· ALL, acute lymphoblastic leukemia; DIC, disseminated intravascular coagulation; HIT, heparin-induced thrombocytopenia; HUS, hemolytic uremic syndrome; ITP, immune thrombocytopenia; LP, lumbar puncture; TTP, thrombotic thrombocytopenic purpura.
· a Note: routine screening by standard coagulation tests not advocated without clinical indication; for laboratory evidence of DIC see Section 5.6.4.
· b It is accepted that prior to lumbar puncture some clinicians will transfuse platelets at higher counts (e.g., 50 × 109/l) in clinically unstable children, non-ALL patients, or for the first LP in newly-diagnosed ALL patients to avoid hemorrhage and cerebrospinal fluid contamination with blasts, or at lower counts (≤20 × 109/l) in stable patients with ALL, depending on the clinical situation. These practices emphasize the importance of considering the clinical setting and patient factors.
New HV, Berryman J,
Bolton-Maggs PH, Cantwell C, Chalmers EA, Davies T, Gottstein R, Kelleher A,
Kumar S, Morley SL, Stanworth SJ; British Committee for Standards in
Haematology. Guidelines on transfusion for fetuses, neonates and older
children. Br J Haematol. 2016 Dec;175(5):784-828. doi: 10.1111/bjh.14233. Epub
2016 Nov 11. PMID: 27861734.