- Instrument & Equipment Processing in Healthcare Facilities
- Contaminated instruments from various hospital or clinic departments should be collected and transported to the CSSD for proper cleaning, disinfection, and sterilization.
- After processing, sterilized instruments should be returned to their respective departments for reuse.
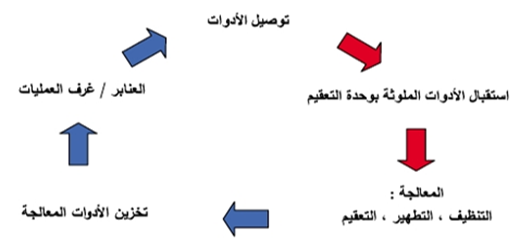
Figure 28
Workflow Diagram for Instrument Reprocessing
Collection and Transportation of Contaminated Instruments:
- Contaminated reusable instruments from hospital departments should be collected in a sealed container filled with water and liquid detergent, labeled with a detailed tag specifying its contents.
- Instruments must be safely transported to the Central Sterile Services Department (CSSD).
- Cleaning contaminated instruments in hospital departments is strictly prohibited.
Handling of Instruments in the CSSD:
- Place contaminated instruments in the designated area within the CSSD.
- Clean all instruments using water and liquid detergent or an enzymatic solution, either manually or using automated systems, while ensuring proper personal protective equipment (PPE) is worn.
- Thoroughly dry all instruments.
- Inspect all equipment to verify cleanliness and detect any damage.
- Send damaged instruments to the maintenance department after high-level disinfection or sterilization.
- Pack clean critical instruments properly before sterilization.
- Sterilize or disinfect instruments as required.
- Verify sterilization effectiveness by checking biological and chemical indicators.
- After reprocessing, return sterilized instruments to hospital departments or store them in a clean, well-ventilated area.
- Strict separation between contaminated and sterile instruments must be maintained within the CSSD.
- Dedicated carts or containers must be used for transporting contaminated instruments—they must not be mixed with those used for sterile instruments.
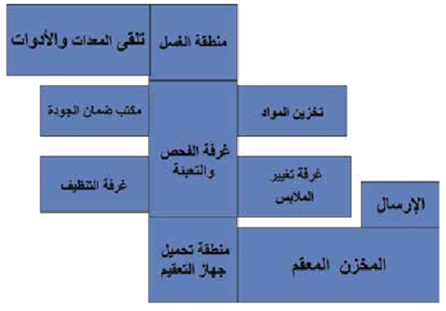
Design of the Central Sterile Services Department (CSSD):
- Physical barriers (easy to clean and disinfect) must separate clean and contaminated areas in the CSSD.
- Airflow direction must move from clean areas to contaminated areas.
- The workflow must be structured to prevent cross-contamination between dirty, clean, disinfected, or sterilized instruments.
- Doors should remain closed to prevent contamination from dust, airborne particles, and insects, while ensuring proper ventilation.
- Adequate hand hygiene facilities must be available.
- A dedicated staff team should be assigned to each processing area.
- If this is not feasible, strict adherence to standard precautions must be maintained for each work zone.
- Separate supplies and storage areas must be allocated for each processing stage.
Important Note:
- All CSSD staff must be vaccinated against Hepatitis B.
- Refer to the Occupational Health and Safety section for further details.
Operations Department General Plan