
- Annexes
Annex Table 1.
Declaration of Conflict of Interests
The members of the guideline development/ adaptation group and the external review group have no academic, financial, or competing interests to declare and none of them were involved in the development of the original source guideline(s).
Any identified potential COI has been reported below.
|
Egyptian Pediatric Clinical Practice Guidelines Committee (EPG) Guideline Adaptation Group (Clinical subgroup) |
|
|||||||
|
Name |
Affiliation, Area of expertise / Role, Country / Primary location [work] |
Declaration of interests |
|
|||||
|
Interest identified |
Management plan & decision |
|
||||||
|
Prof. Mona Mamdouh Hassan |
Pediatrics Department, Consultant, Cairo University |
|
||||||
|
Ass. Prof. Amal Gaber Mohamed |
Pediatrics Department, Consultant, Al-Azhar University |
|
||||||
|
Prof. Amany Kamal El-Hawary |
Pediatrics Department, Consultant, Mansoura University |
|
||||||
|
Prof. Amina M. Abdel Wahab |
Pediatrics Department, Consultant, Suez Canal University |
|
||||||
|
Prof. Ashraf A. Elsharkawy |
Pediatrics Department, Consultant, Mansoura University |
|
||||||
|
Prof. Basma Abd-Elmoez |
Pediatrics Department, Consultant, Minia University |
|
||||||
|
Dr. Eman Elshanawany
|
Pediatrics Department, Consultant, Benha University |
|
||||||
|
Prof. Ghada Anwar |
Pediatrics Department, Cairo University |
|
||||||
|
Prof. Hanaa Abdellateef Mohamad |
Pediatrics Department, Consultant, Assiut University |
|
||||||
|
Ass. Prof. Hanan Hassan Aly |
Pediatrics Department, Consultant, Ain Shams University |
|
||||||
|
Prof. Hoda Atwa |
Pediatrics Department, Consultant, Suez Canal University |
|
||||||
|
Prof. Lubna Fawaz |
Pediatrics Department, Consultant, Cairo University |
|
||||||
|
Dr. Mariam Nader Moawad, |
Department of Pediatrics,Consultant, Armed Forces College of Medicine |
|
||||||
|
Dr. Marian Fares Nashed |
Department of Pediatrics,Consultant, Armed Forces College of Medicine |
|
||||||
|
Ass. Prof. Mona Karem Amin |
Pediatrics Department, Consultant, Suez Canal University |
|
||||||
|
Prof. Nora E Badawi |
Pediatrics Department, Consultant, Cairo University |
|
||||||
|
Ass. Prof. Nouran Y Salah El-Din |
Department of Pediatrics, Ain Shams University |
|
||||||
|
Dr. Ramy Saleh Morsy |
Department of Pediatrics, Consultant, Armed Forces College of Medicine |
|
||||||
|
Prof. Randa M. Matter |
Pediatrics Department, Consultant, Ain Shams University |
|
||||||
|
Ass. Prof. Remon M. Yousef |
Department of Pediatrics, Consultant, Fayoum University |
|
||||||
|
Prof. Sabry M Ghanem |
Department of Pediatrics, Consultant, Al-Azhar University |
|
||||||
|
Prof. Safinaz El Habashy |
Department of Pediatrics, Consultant, Ain Shams University |
|
||||||
|
Ass. Prof. Shaymaa Elsayed Abdel Meguid |
Department of Pediatrics, Consultant, Alexandria University |
|
||||||
|
Prof. Wiam Al Farouk Younis |
Department of Pediatrics,Consultant, Armed Forces College of Medicine |
|
||||||
|
Dr. Hamed Khaled Khalifa |
M.B.B.Ch Misr University for Science and Technology. House Officer, Pediatrics Department, Cairo University.
|
|
||||||
|
Prof. Tarek Omar |
Pediatrics Department, Faculty of Medicine, Alexandria University, Alexandria, Egypt Alexandria Center for Evidence-Based CPG Consultancy Board, EPG, Egypt |
Professor of Pediatrics, Methodology Supervision Subgroup |
||||||
|
|
|
None |
Not Applicable |
|
||||
|
Guideline Adaptation Group (Methodology Subgroup) |
|
|||||||
|
Prof. Ashraf Abdel Baky |
Professor of Pediatrics Ain Shams University, Egypt Founder and Chair of EPG |
None |
Not Applicable |
|
||||
|
Dr. Yasser Sami Amer |
1. Pediatrics Department and Clinical Practice Guidelines and Quality Research Unit, Quality Management Department, King Saud University Medical City, Riyadh, Saudi Arabia; 2. Research Chair for Evidence-Based Health Care and Knowledge Translation, King Saud University, Riyadh, Saudi Arabia; 3. Chair, Adaptation Working Group, Guidelines International Network (GIN), Perth, Scotland 4. Department of Internal Medicine, Ribeirão Preto Medical School, University of São Paulo (FMRP-USP), Ribeirão Preto, São Paulo, Brazil. |
None |
Not Applicable |
|
||||
|
Dr. Nahla Gamaleldin |
Lecturer of pediatrics, Faculty of Medicine, Modern University for Technology and Information (MTI), Egypt |
None |
Not Applicable |
|
||||
|
External Review Group |
|
|||||||
|
Prof. Ghada Anwar |
Pediatrics Department, Cairo University/ Egypt |
|
||||||
|
Prof. Mona Hafez |
Pediatrics Department, Cairo University/ Egypt |
|
||||||
|
Prof. Mona Salem |
Pediatrics Department, Ain Shams University/ Egypt |
|
||||||
|
Prof. Nermin Salah |
Pediatrics Department, Cairo University/ Egypt |
|
||||||
|
Hesham El Hefnawy |
Prof. of diabetes and endocrinology, former dean of the National Institute of Diabetes and Endocrinology, Cairo, Egypt. Head of National Committee of non-communicable disease |
|
||||||
|
External Reviewer for methodology |
|
|||||||
|
Prof. Iván D. Flórez |
Department of Pediatrics, University of Antioquia, Medellín, Colombia, Department of Health Research Methods, Evidence, and Impact, McMaster University, Hamilton, Canada, Leader, AGREE Collaboration (Appraisal of Guidelines for Research & Evaluation) Director, Cochrane Colombia |
|
|||||||
|
Prof. Airton Tetelbom Stein
|
Professor Titular de Saúde Coletiva, Fundação Universidade Federal de Ciências da Saúde de Porto Alegre (UFCSPA), Porto Alegre, Brazil Professor Adjunto, Universidade Luterana do Brasil (Ulbra), Canoas, Brazil Coordenador de Diretrizes Clínicas, Grupo Hospitalar Conceição, Porto Alegre, Brazil 4. Member, Board of Trustees, Guidelines International Network (G-I-N) |
|
|||||||
|
International Peer Reviewers |
|
|
|
|||||
|
|
|
None |
Not Applicable |
|
||||
➡️Web annexes
The following annexes can be added as a package of standalone supplementary documents.
➡️Keywords: The MeSH terms for "Guideline for the prevention and management of DKA’’" on PubMed are: DKA, pediatrics, diagnosis, management
|
Pediatrics Department, Cairo University/ Egypt |
|
|
Prof. Mona Hafez |
Pediatrics Department, Cairo University/ Egypt |
|
Prof. Mona Salem |
Pediatrics Department, Ain Shams University/ Egypt |
|
Prof. Nermin Salah |
Pediatrics Department, Cairo University/ Egypt |
|
Hesham El Hefnawy |
Prof. of diabetes and endocrinology, former dean of the National Institute of Diabetes and Endocrinology, Cairo, Egypt. Head of National Committee of non-communicable disease |
Appendix 1. List of the websites and databases we searched:
CPG databases and libraries:
1. Guidelines International Network (GIN) International Guidelines Library.
https://g-i-n.net/international-guidelines-library/
2. ECRI Guidelines Trust (USA). https://guidelines.ecri.org/
3. National Institute of Clinical and Health Excellence (NICE) UK. http://www.nice.org.uk/guidance/
4. Scottish Intercollegiate Guidelines Network (SIGN) UK. http://www.sign.ac.uk/guidelines/
5. EBSCO DynaMed (USA) https://www.dynamed.com/ (subscription required)
Bibliographic databases:
1. PubMed/ MEDLINE https://pubmed.ncbi.nlm.nih.gov/
2. Embase https://www.embase.com/landing?status=grey (subscription required)
3. CINAHL https://www.ebsco.com/products/research-databases/cinahl-complete
Specialized professional societies:
1. American Academy of Pediatrics (AAP) https://www.aap.org/
2. Canadian Paediatric Society (CPS) https://www.cps.ca/
Royal College of Paediatrics and Child Health (RCPCH) https://www.rcpch.ac.uk/
Annex Table 2. Results of the AGREE II assessment of the three source guidelines for DKA management
|
CPGs AGREE II DOMAINS |
CPG1 |
CPG2 |
CPG3 |
CPG4 |
CPG5 |
|
D1: Scope & Purpose |
60% |
53% |
61% |
99% |
67% |
|
D2: Stakeholder Involvement |
38% |
43% |
36% |
94% |
69% |
|
D3: Rigour of Development |
15% |
9% |
26% |
99% |
60% |
|
D4: Clarity & Presentation |
65% |
68% |
78% |
100% |
94% |
|
D5: Applicability |
9% |
7% |
20% |
96% |
74% |
|
D6: Editorial Independence |
56% |
56% |
4% |
90% |
71% |
|
OA 1 |
33% |
38% |
38% |
100% |
75% |
|
OA 2 (Recommend for use) |
No (4) |
No (4) |
No (4) |
Yes (4) |
Yes (3) Yes with modif. (1) |
|
This table uses the AGREE II Domain Score Color codes (< 40% red; > 41 – 70% yellow; > 71 % green)
|
|||||
Annex Table 3. Annex
Nurses and Parents Educational Guide in Arabic
Appendix Table 4. The RIGHT-Ad@pt checklist |
|||||
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
SNS & NEBMC |
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☐ Yes ☒ No ☐ Unclear |
-- |
|
|
Implementation tools:
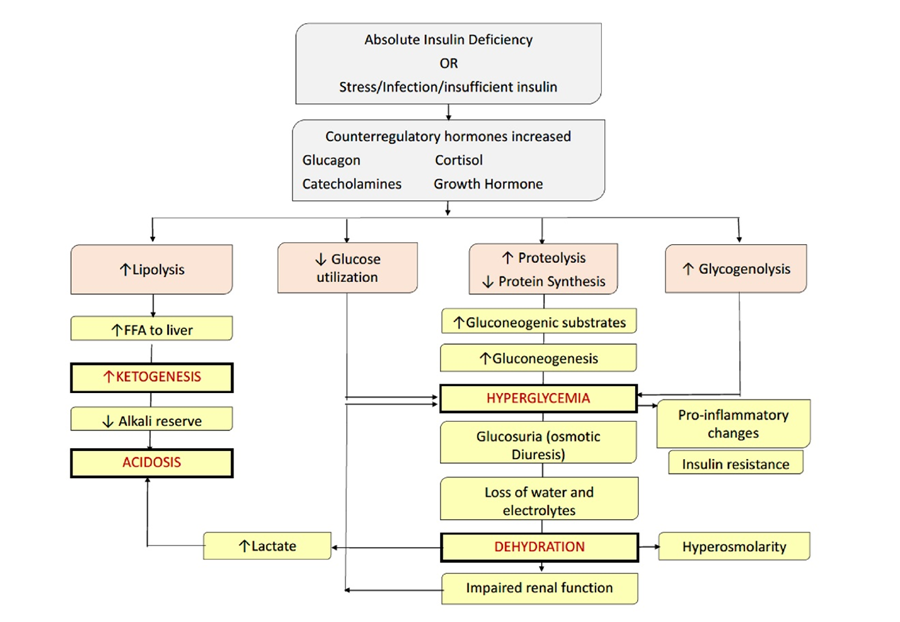
Figure (1): Pathophysiology of diabetic ketoacidosis.
Copyright, 2006 American Diabetes Association. From Diabetes Care,
Vol. 29, 2006:1150–9. ISPAD 2018
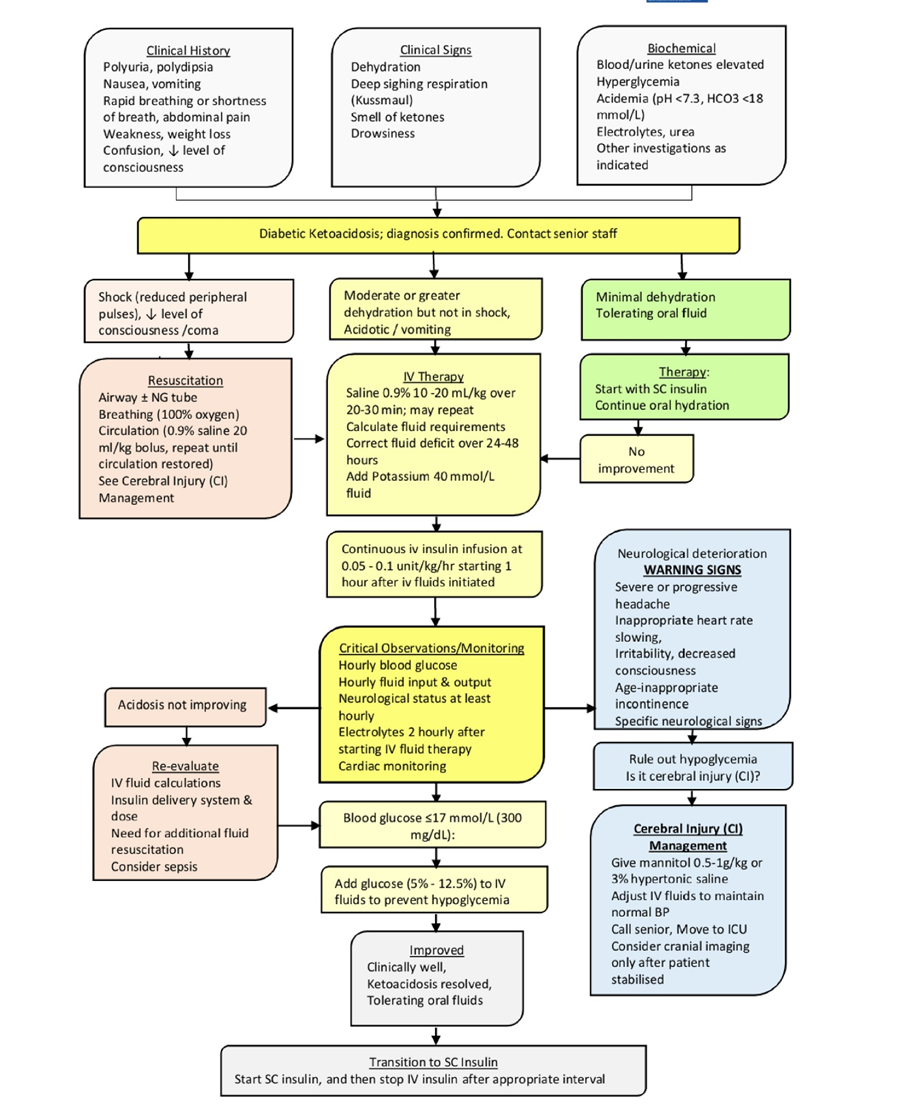
Figure (2): Algorithm for the management of DKA
Pinhas-Hamiel O, Sperling M. Diabetic ketoacidosis. In: Hochberg Z, ed. Practical Algorithms in Pediatric Endocrinology. 3rd, revised edition ed. Basel: Karger; 2017:112-113
· Figure (3): ECG findings in hypo- and hyperkalemia.
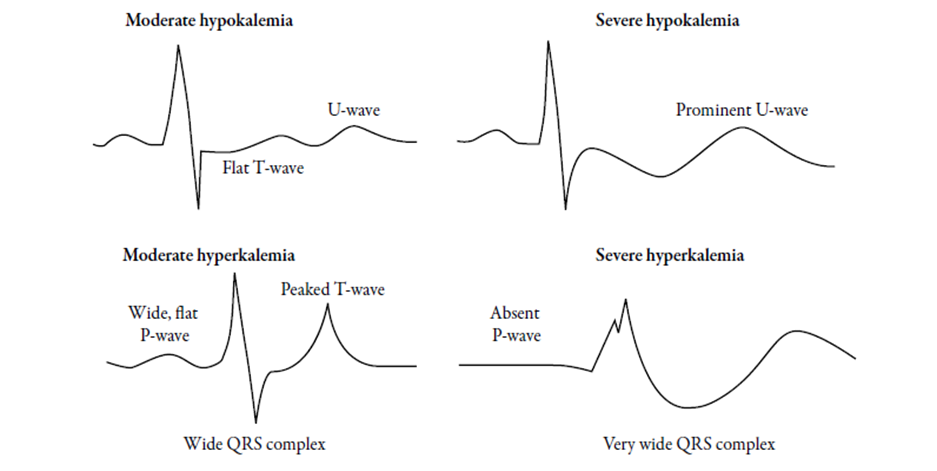
Table (2): Losses of fluids and electrolytes in diabetic ketoacidosis and maintenance requirements in normal children.
(ISPAD clinical practice consensus guidelines 2018, Pediatric Diabetes October 2018; 19 (Suppl.27): 155-177).

Table (3): Glasgow Coma Scale
Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale Lancet 1974: 2: 81–4

Table (4): An alternative example of fluid volumes for the subsequent phase of rehydration. Diabetic Ketoacidosis and Hyperglycemic Hyperosmolar State: A Consensus Statement from the International Society for Pediatric and Adolescent Diabetes. Pediatric Diabetes 2018; 19 (Suppl 27): 155
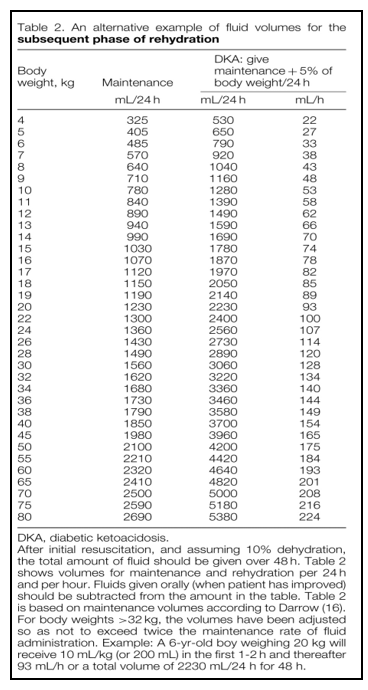
Table (5): Determining Clinical cerebral edema risk.
Muir AB, Quisling RG, Yang MC, Rosenbloom AL. Cerebral Edema in Childhood Diabetic Ketoacidosis: Natural history, radiographic findings, and early identification. Diabetes Care. 2004; 27 (7):1541-1546.
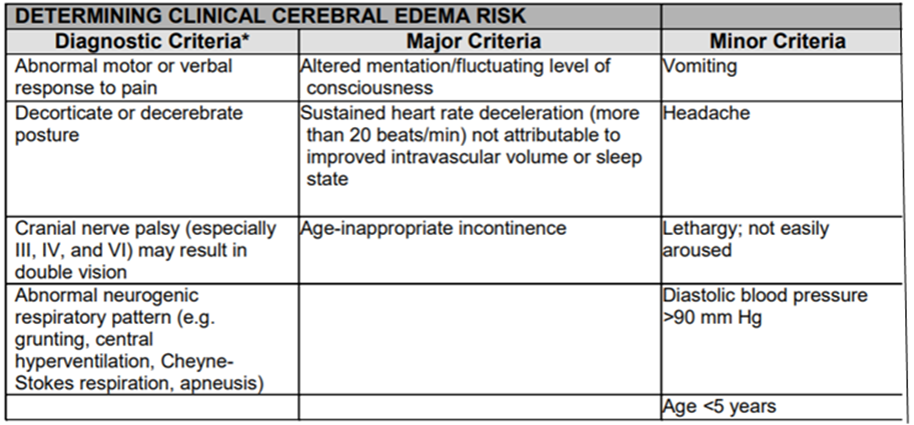
*One diagnostic criterion, or two major criteria, or one major and two minor criteria have a sensitivity of 92%, a specificity of 96% and a false positive rate of only 4% for the early recognition of DKA-related cerebral edema; early enough to allow for effective treatment.
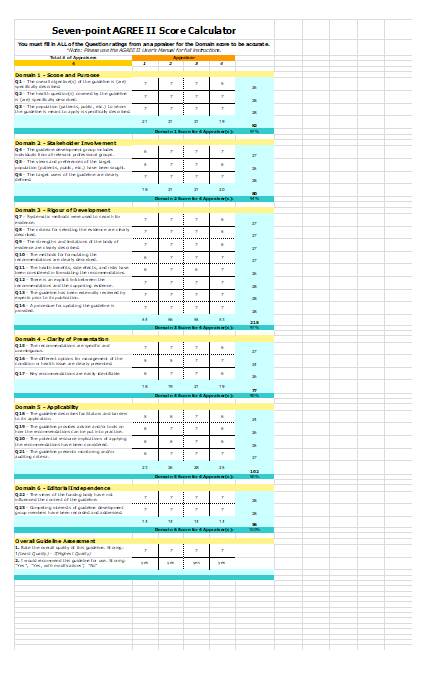
Figure (4): Agree II Score for ISPAD Clinical Consensus Guidelines 20181

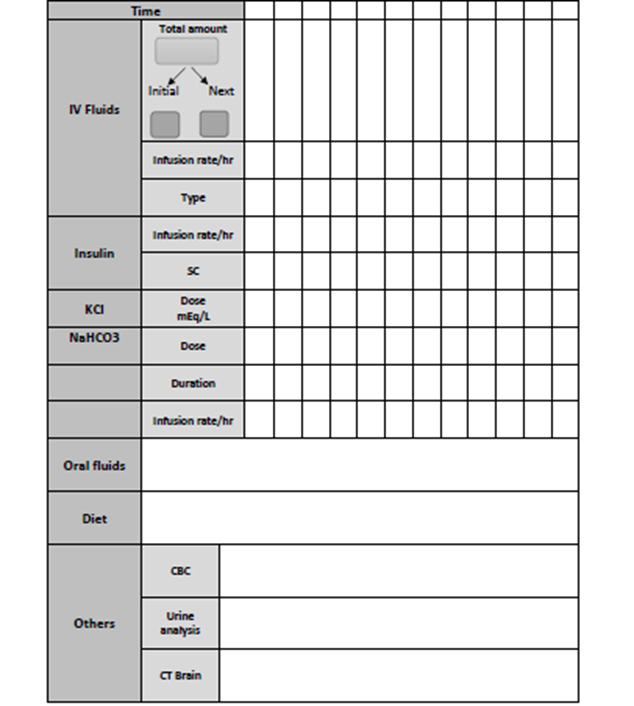
Follow up sheet for DKA ,DEMPU1 Table (6)
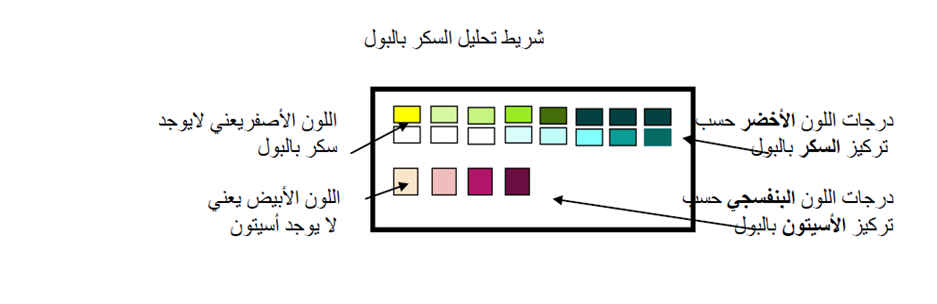
Instructions in Arabic for Family and Patient Education
From DEMPU Diabetes Education Booklet, Cairo University
كتيب التقيف لسكر الأطفال و الشباب بوحدة السكر و الغدد الصماء و الميتابوليزم، مستشفى الأطفال- جامعة القاهرة، اعداد د سحر أبو الليل، مراجعة أ.د.نرمين صلاح.
رقم ايداع2008/ 19133
: ص 28-30





Annex Table 4. The RIGHT-Ad@pt checklist
|
7 sections, 27 topics, and 34 items |
Assessment |
Page(s)* |
Note(s) |
||
|
BASIC INFORMATION |
|||||
|
Title/subtitle |
|||||
|
1 |
Identify the report as an adaptation of practice guideline(s), that is include "guideline adaptation", "adapting", "adapted guideline/recommendation(s)", or similar terminology in the title/subtitle. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
2 |
Describe the topic/focus/scope of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Cover/first page |
|||||
|
3 |
Report the respective dates of publication and the literature search of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
4 |
Describe the developer and country/region of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Executive summary/abstract |
|||||
|
5 |
Provide a summary of the recommendations contained in the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Abbreviations and acronyms |
|||||
|
6 |
Define key terms and provide a list of abbreviations and acronyms (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Contact information of the guideline adaptation group |
|||||
|
7 |
Report the contact information of the developer of the adapted guideline. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
SCOPE |
|||||
|
Source guideline(s) |
|||||
|
8 |
Report the name and year of publication of the source guideline(s), provide the citation(s), and whether source authors were contacted. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Brief description of the health problem(s) |
|||||
|
9 |
Provide the basic epidemiological information about the problem (including the associated burden), health systems relevant issues, and note any relevant differences compared to the source guideline(s).
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Aim(s) and specific objectives |
|||||
|
10 |
Describe the aim(s) of the adapted guideline and specific objectives, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Target population(s) |
|||||
|
11 |
Describe the target population(s) and subgroup(s) (if applicable) to which the recommendation(s) is addressed in the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
End-users and settings |
|||||
|
12 |
Describe the intended target users of the adapted guideline, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
13 |
Describe the setting(s) for which the adapted guideline is intended, and note any relevant differences compared to the source guideline(s). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RIGOR OF DEVELOPMENT |
|||||
|
Guideline adaptation group |
|||||
|
14 |
List all contributors to the guideline adaptation process and describe their selection process and responsibilities. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Adaptation framework/methodology |
|||||
|
15 |
Report which framework or methodology was used in the guideline adaptation process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Source guideline(s) |
|||||
|
16 |
Describe how the specific source guideline(s) was(were) selected. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Key questions |
|||||
|
17 |
State the key questions of the adapted guideline using a structured format, such as PICO (population, intervention, comparator, and outcome), or another format as appropriate. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
18 |
Describe how the key questions were developed/modified, and/or prioritized. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Source recommendation(s) |
|||||
|
19 |
Describe how the recommendation(s) from the source guideline(s) was(were) assessed with respect to the evidence considered for the different criteria, the judgements and considerations made by the original panel. |
☐ Yes ☒ No ☐ Unclear |
|
|
|
|
Evidence synthesis |
|||||
|
20 |
Indicate whether the adapted recommendation(s) is/are based on existing evidence from the source guideline(s), and/or additional evidence. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
21 |
If new research evidence was used, describe how it was identified and assessed. |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Assessment of the certainty of the body of evidence and strength of recommendation |
|||||
|
22 |
Describe the approach used to assess the certainty/quality of the body/ies of evidence and the strength of recommendations in the adapted guideline and note any differences (if applicable) compared to the source guideline(s). |
☐ Yes ☒ No ☐ Unclear |
NA |
|
|
|
Decision-making processes |
|||||
|
23 |
Describe the processes used by the guideline adaptation group to make decisions, particularly the formulation of recommendations.
|
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
RECOMMENDATIONS |
|||||
|
Recommendations |
|||||
|
24 |
Report recommendations and indicate whether they were adapted, adopted, or de novo. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
25 |
Indicate the direction and strength of the recommendations and the certainty/quality of the supporting evidence and note any differences compared to the source recommendations(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
26 |
Present separate recommendations for important subgroups if the evidence suggests important differences in factors influencing recommendations and note any differences compared to the source recommendations(s) (If applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Rationale/explanation for recommendations |
|||||
|
27 |
Describe the criteria/factors that were considered to formulate the recommendations or note any relevant differences compared to the source guideline(s) (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
EXTERNAL REVIEW AND QUALITY ASSURANCE |
|||||
|
External review |
|||||
|
28 |
Indicate whether the adapted guideline underwent an independent external review. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Organizational approval |
|||||
|
29 |
Indicate whether the adapted guideline obtained organizational approval. If yes, describe the process. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
FUNDING, DECLARATION, AND MANAGEMENT OF INTEREST |
|||||
|
Funding source(s) and funder role(s) |
|||||
|
30 |
Report all sources of funding for the adapted guideline and source guideline(s), and the role of the funders. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Declaration and management of interests |
|||||
|
31 |
Report all conflicts of interest of the adapted and the source guideline(s) panels, and how they were evaluated and managed. |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
OTHER INFORMATION |
|||||
|
Implementation |
|||||
|
32 |
Describe the potential barriers and strategies for implementing the recommendations (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Update |
|||||
|
33 |
Briefly describe the strategy for updating the adapted guideline (if applicable). |
☒ Yes ☐ No ☐ Unclear |
|
|
|
|
Limitations and suggestions for further research |
|||||
|
34 |
Describe the challenges of the adaptation process, the limitations of the evidence, and provide suggestions for future research. |
☒ Yes ☐ No ☐ Unclear |
61 |
|
|
