
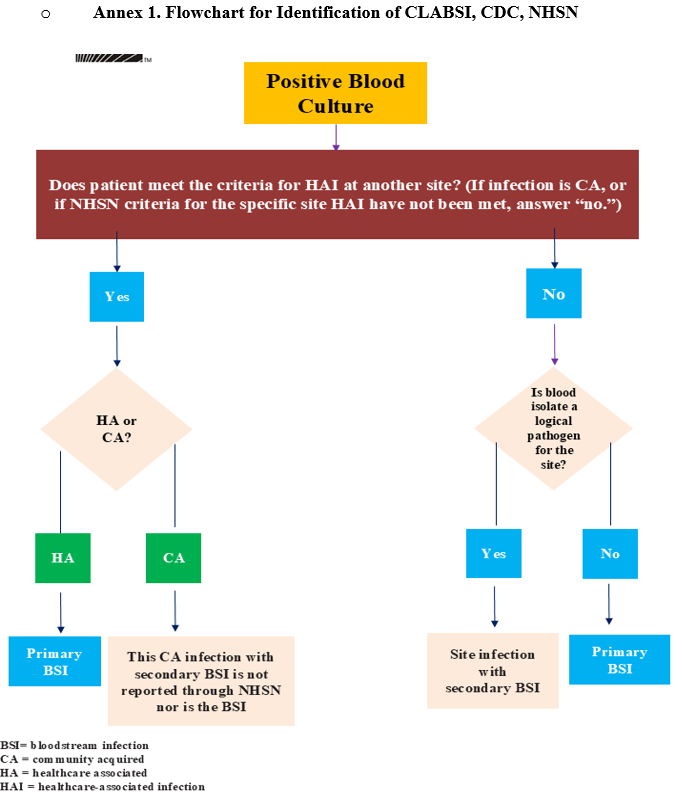
- Annex
Annex 2. Differentiating CLABSI and CRBSI (APIC 2014)
|
Criteria |
CLABSI |
CRBSI |
|
Purpose of definition |
Surveillance |
Clinical diagnosis |
|
Device removal required |
Usually no |
Usually yes |
|
Cultures |
Qualitative blood cultures |
Blood cultures with differential time to positivity |
|
Catheter tip culture recommended |
No |
Yes |
|
Major advantage |
Convenience, lower cost, readily available in most laboratories |
High sensitivity; better positive predictive value |
|
Major disadvantage |
Often unable to distinguish a primary and secondary BSI; may overstate the true incidence of primary CLABSI |
More complex, less convenient, expensive |
Annex 3. Many IV devices are available to
deliver medications, fluids and nutrition into patients' bodies, and monitor
vital signs. Each carries its own benefits and risks for different patients.
Credit: University of Michigan
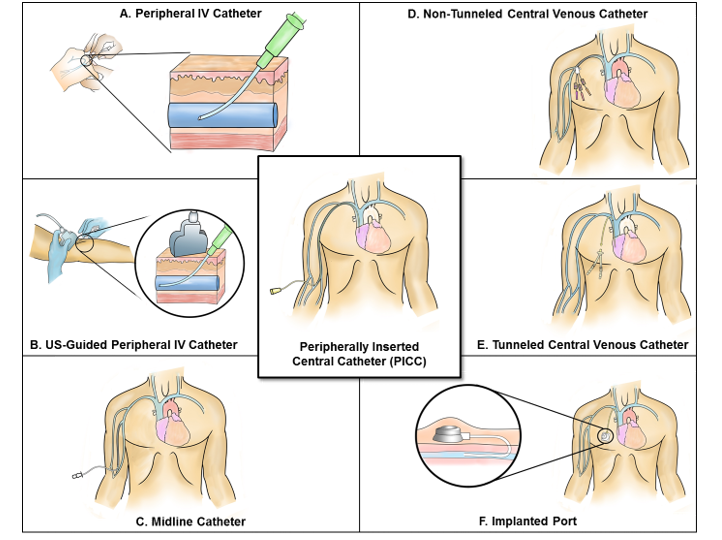
Examples of central lines: Tunnelled and non-tunnelled central venous catheters, implanted ports, pulmonary artery catheters, dialysis or haemofiltration catheters in a great vessel and peripherally inserted central catheters. An introducer is considered a central line if the tip is situated in a great vessel.
Examples of devices that are not considered to be central lines for the purpose of CLABSI surveillance: Arterial catheters, arteriovenous fistulas, arteriovenous grafts, atrial catheters (also known as transthoracic intra-cardiac catheters, those catheters inserted directly into the right or left atrium via the heart wall), extracorporeal membrane oxygenation (ECMO), femoral arterial catheters, intra-aortic balloon pump (IABP) devices, haemodialysis reliable outflow (HeRO) dialysis catheters, peripheral intravenous cannulae or midline catheters, and ventricular assist devices (VAD). Pacemaker wires and other solid or non‑lumen devices inserted into central blood vessels, or the heart are not considered central lines, because fluids are not infused, pushed, or withdrawn through such devices.
