
- Evidence to recommendations
The GDG/ GAG was guided by the results of the AGREE II appraisals of the eligible CPGs and thoroughly reviewed the recommendations of the original source WHO CPGs in consideration of local contextual factors related to the national Egyptian health system like burden of the disease, equity, acceptability, feasibility, and other relevant factors. The GDG decided through an informal consensus process to adopt most recommendation. both ETD and changing strength of recommendation were not done in this guideline).
Also, GDG/ GAG develops group of good practice statements to improve acceptability and feasibility.
➡️Implementation Tools and Considerations
To improve healthcare provision, quality, safety, and patient outcome, evidence-based recommendations must not only be developed, but also disseminated and implemented at national and local levels and integrated into clinical practice.
Dissemination involves educating related healthcare providers to improve their awareness, knowledge and understanding of the guideline’s recommendations. It is one part of implementation, which involved translation of evidence-based guidelines into real life practice with improvement of health outcomes for the patients.
Implementation requires an evidence-based strategy involving professional groups and stakeholders and should consider the local cultural and socioeconomic conditions. Cost-effectiveness of implementation programs should be assessed.
Specific steps need to be followed before clinical practice recommendations can be integrated into local clinical practice, particularly in low resource settings.
Steps of implementing Emergency Management (Diagnosis and Treatment) of Seizures in Children beyond the Neonatal Period strategies into the Egyptian health system:
1. Develop a multidisciplinary working group.
2. Assess the status of nutritional care delivery, care gaps and current needs.
3. Select the material to be implemented, agree on the main goals, identify the key recommendations for diagnosis, treatment and prevention and adapt them to the local context or environment.
4. Identify barriers to, and facilitators of implementation.
5. Select an implementation framework and its component strategies.
6. Develop a step-by-step implementation plan:
· Select the target populations and evaluate the outcome.
· Identify the local resources to support the implementation.
· Set timelines.
· Distribute the tasks to the members.
· Evaluate the outcomes.
7. Continuously review the progress and results to determine if the strategy requires modification.
Guideline implementation strategies will focus on the following: -
1. For Practitioners
· Educational meetings: conferences, lectures, workshops, grand rounds, seminars, and symposia.
· Educational materials: printed or electronic information (software).
· Web-based education: computer-based educational activities.
· A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s).
· Reminders: the provision of information verbally, on papers or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care.
· Optimize professional-patient interactions, through mass media campaigns, reminders, and education materials.
· Practice tools: tools designed to facilitate behavioral/practice changes, e.g., flow charts.
2. For Patients and care givers
· Patient education materials (Arabic booklet): Printed/electronic information aimed at the patient/consumer, family, caregivers, etc.
· Reminders: the provision of information verbally, on papers or electronically to remind a patient/consumer to perform a particular health-related behaviors.
· Mass media campaigns.
3. For Nurses
· Educational meetings: lectures, workshops or traineeships, seminars, and symposia.
· Educational materials: printed.
· A trained person meets with nurses in their practice setting to provide information with the intention of changing the provider’s practice.
· Reminders: the provision of information verbally, on paper or on a computer screen to prompt them to recall information or to perform or avoid a particular action related to patient care.
· Practice tools: tools designed to facilitate behavioral/practice changes.
4. For Stakeholders
Plans have been made to contact with all the health sectors in Egypt including all sectors of the Ministry of Health and Population, National Nutrition Institute, University Hospitals, Ministry of Interior, Ministry of Defense, Non-Governmental Organizations, Private sector, and all Health Care Facilities.
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc.
· Any summary of clinical provision of health care over a specified period may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. Summary may be targeted at the individual practitioner or the organization.
· Administrative policies and procedures.
· Formularies: Drug safety programs, electronic medication administration records.
5. Other activities to assist the implementation of the adapted guideline’s recommendations include:
· International initiative: Dissemination of the presented adapted CPG internationally via sending the final adapted CPG to the Guidelines International Network (GIN) Adaptation Working Group and contacting the CPG developers.
· Gantt chart has been designed to manage the dissemination and implementation stages for the adapted CPG over an accurate time frame (Appendix).
➡️Guideline Implementation Tools
Educational materials based on this Adapted CPG for Emergency Management (Diagnosis and Treatment) of Seizures in Children beyond the Neonatal Period have been made available in several forms including:
Table (13): CPG implementation strategies
|
Focus of Strategy |
Strategies |
|
Practitioners |
· Educational meetings: Conferences, lectures, workshops or traineeships, grand rounds, seminars, and symposia. |
|
· Educational materials: Printed or electronic information. |
|
|
· Web-based education: Computer-based educational activities. |
|
|
· Educational outreach/academic detailing: A trained person meets with providers in their practice setting to provide information with the intention of changing the provider’s practice. The information may include feedback on the performance of the provider(s). |
|
|
· Audit and feedback: Any summary of the clinical provision of health care over a specified period; may include recommendations for clinical action. The information is obtained from medical records, databases, or observations of patients. The summary may be targeted at the individual practitioner or the organization. |
|
|
· Reminders: The provision of information verbally, on paper or on a computer screen to prompt a health professional to recall information or to perform or avoid a particular action related to patient care. |
|
|
· Local opinion leaders: Providers nominated by their colleagues as “educationally influential.” In general, such individuals are identified by their peer colleagues, are trained as change agents, and operate within their communities to teach and enable change. |
|
|
· Patient-mediated interventions: Interventions directed at patients (e.g., mass media campaigns, reminders, education materials) to optimize professional-patient interactions. |
|
|
· Practice tools: Tools designed to facilitate behavioral/practice changes, e.g., flow charts. |
|
|
Patients |
· Patient education materials: Printed/electronic information aimed at the patient, consumer, family, caregivers, etc. |
|
· Mass media campaigns |
|
|
· Reminders: The provision of information verbally, on paper, or electronically to remind a patient/consumer to perform a particular health-related behavior. |
|
|
· Decision-support tools: Aids designed to facilitate shared decisions by patients and their physicians |
|
|
Organizations and regulatory bodies |
· Changes to health care teams: Changing tasks or responsibilities of health professionals or compositions of health professional groups. |
|
· Information and communication technology: Electronic decision support, order sets, care maps, electronic health records, office-based personal digital assistants, etc. |
|
|
· Audit and feedback: Any summary of a clinical provision of health care over a specified period; may include recommendations for clinical action. The information is obtained from medical records, databases, or observations by patients. The summary may be targeted at the individual practitioner or the organization. |
|
|
· Administrative procedures/policies |
|
|
· Formularies: Drug safety programs, electronic medication administration records. |
|
|
· Financial incentives or penalties: The use of remuneration for the performance of certain functions or actions, e.g., screening procedures in primary care. |
|
|
· Mandated practices |
➡️Example of Dissemination and Implementation Proposed Resources
Educational materials based on this Adapted CPG for acute management of seizures in children beyond the neonatal period shall be made available in several forms, including a pocket guide for health care professionals and for parents/caregivers.
proposed Implementation tools:-
• Patient Resources (Information) (Arabic).
• Pocket Guide/Quick Reference Summary (English)
• Power Point Slide Presentation.
• Clinical Algorithm
• Drug Table
1. Pediatric Emergency Triage:
Pocketbook of hospital care for children: Second edition.
Guidelines for the management of common childhood illnesses http://apps.who.int/iris/bitstream/10665/204463/1/9789241510219_eng.pdf?ua=1
Pediatric emergency triage, assessment, and treatment: care of critically-ill children Updated guideline
http://apps.who.int/iris/bitstream/10665/204463/1/9789241510219_eng.pdf?ua=1

2.Quick Summary Guide
|
Guideline Source |
Recommendation |
Level of Evidence |
|
History taking |
||
|
Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics, 2015 |
1. The first stage in clinical management is to recognize if abnormal movement or behavior has an epileptic origin |
Level B |
|
Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics, 2015 |
2. Rule out febrile seizures |
Level B |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
3. Ask about prior history of epilepsy |
Level B |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
4. AED given, AED non-compliance, or known allergies. |
Level C |
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
5. Address history of Ingestion of a toxin or drug abuse as possible etiologies of SE. |
Level C
|
|
Clinical Examination |
||
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
1. Assessment of vital signs: O2 saturation, BP, HR in the immediate (0–2 min) to establish and support baseline vital signs |
Level U |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
2. Neurologic exam is urgent (5–10 min) to evaluate for mass lesion, acute intracranial process |
Level U |
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
3.Address respiratory monitoring to prevent cardiopulmonary arrest, during anticonvulsant drug treatment in status epilepticus in children. |
Level A |
Laboratory Evaluation |
||
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012 |
1. Laboratory tests should be ordered based on individual clinical circumstances that include suggestive historic or clinical findings such as vomiting, diarrhea, dehydration, or failure to return to baseline alertness. |
Level U |
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
2. For all patients, order laboratory tests: blood glucose, complete blood count, calcium (total and ionized), magnesium, AED levels. |
Level U |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012
|
3. Consider based on clinical presentation c) Lumbar puncture (LP). Should be performed in the very young child (<6 months), in the child of any age with persistent (cause unknown) alteration of mental status or failure to return to baseline, or in any child with meningeal signs. If increased intracranial pressure is suspected, the LP should be preceded by an imaging study of the head d) Comprehensive toxicology panel including toxins that frequently cause seizures (i.e. isoniazid, tricyclic antidepressants, theophylline, cocaine, sympathomimetics, alcohol, organophosphates, and cyclosporine e) Other laboratory tests: liver function tests, coagulation studies, arterial blood gases, and inborn errors of metabolism |
Level U
Level U |
|
EEG |
||
|
Evidence-Based Guidelines for EEG Utilization at the University Teaching Hospital (UTH) – 2011 |
V. In the PICU, An EEG is indicated in all unconscious patients suspected of non-convulsive status epilepticus or subclinical seizures. This includes comatose or obtunded inpatients of unclear etiology especially those in whom seizures preceded the onset of coma. VI. EEG is recommended after a first unprovoked seizure as it might offer insights into recurrence risk and/or the need for further neuroimaging. |
Level U |
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
VII. In the treatment of convulsive status epilepticus in children, continuous EEG monitoring is indicated if second therapy fails to stop seizures, and treatment consideration involves the use of anesthetic doses of either thiopental, midazolam, pentobarbital, or propofol. |
Level U |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012 |
VIII. The use of continuous EEG (cEEG) is usually required for the treatment of SE. IX. Continuous EEG monitoring should be initiated within 1 h of SE onset if ongoing seizures are suspected X. The duration of cEEG monitoring should be at least 48 h in comatose patients to evaluate for non-convulsive seizures. XI. The use of this cEEG is encouraged to be available at tertiary care facilities where the optimum standard of care is provided. |
Level U
Guideline Adaptation Group and External Reviewer |
|
Neuroimaging (CT / MRI Brain) |
||
|
Diagnosis and Treatment of the First Epileptic Seizure: Guidelines of the Italian League Against Epilepsy, 2006 |
i. The use of a brain CT scan or MRI in the emergency room is indicated when specific interventions may be needed. ii. A CT scan is strictly indicated when a structural lesion is suspected or when the etiology of the seizure cannot be easily identified. Structural lesions include, among others, post-traumatic complications, cerebral hemorrhage, brain edema, and space-occupying lesions, which may be suggested by post-ictal deficits and/or persisting impairment of consciousness. iii. In the emergency room, MRI is not indicated, except for much selected circumstances, to be evaluated on an individual basis. |
Level 2 |
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
· The etiology of SE should be diagnosed as soon as possible and occur simultaneously and in parallel with treatment for evaluation for mass lesions, meningitis, encephalitis. · All patients with SE should have Head computed tomography (CT) scan · Consider Brain magnetic resonance imaging (MRI)based on clinical presentation |
strong recommendation
Expert opinion Expert opinion |
Therapies |
||
|
Initial Therapy: 0 – 20 minutes |
||
|
1. Benzodiazepines |
||
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
|
· Benzodiazepines are the most efficient group of drugs to be initially used in the treatment of acute convulsion after 5 minutes from the start of seizures
|
Level A
|
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
1. Pre-hospital Treatment: - As early as possible by Trained personal whether medical\Para medical or caretaker. |
Level 1B Level 2B |
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
- Drugs of Choice: - Midazolam (intra-muscular – buccal – nasal) - Diazepam (rectal) - Only one dose Not to be repeated |
Level1B
Level1B Level1B Level B
|
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
|
2. Hospital Emergency Room: · History of pre-hospital use of benzodiazepines must be considered as well as timing. · Doses given within one hour prior to ER presentation must be regarded as an initial dose - Midazolam It is the most appropriate type of benzodiazepines to be used. In the hospital the whole full dose to be given as a single dose better than broken into multiple doses. - Diazepam IV diazepam could be repeated after 5 minutes convulsion continued · Alternatively IF VENOUS ACCESS UNAVAILABLE. - Use midazolam: buccal or intranasal. - Use diazepam: rectal · Follow up for signs of respiratory depression as hypoventilation, pallor decreased oxygen saturation. · Respiratory support and intubation may be needed |
Level A
Level A
Level B
Level A
|
|
2. Non Benzodiazepine Therapy 2.1 Phenytoin and Phenobarbitone |
||
|
First Phase therapy: 5 - 20 minutes |
||
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
· Both phenytoin and phenobarbital are recommended |
Level A
|
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
· Phenytoin followed by phenobarbital is recommended if benzodiazepines fail |
Level 2B |
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
· Phenobarbital is recommended in the absence of benzodiazepines |
Level. A |
|
Second phase therapy: 20 - 40 minutes |
||
|
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
- Phenytoin and Phenobarbital is recommended
- They are not used sequentially but alternating. Generally, phenytoin precedes phenobarbital on account of better safety profile |
Class II a Class II b |
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
· Level B |
|
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016 |
Level B |
|
|
2.2 Valproate |
||
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: (AES guidelines: Epilepsy Currents, Vol. 16, No. 1 (January/February) 2016 pp. 48–61: |
|
Level B
Recommendations of Guideline Adaptation Group and External Reviewers |
|
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
· Valproic acid offers a valid alternative to phenytoin and phenobarbital. It should be used with extreme caution, however, particularly in young children, when a possible metabolic etiology has not been ruled out. |
Level 1B |
|
2.3 Pyridoxine |
||
|
Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics, 2015
Guidelines for the Evaluation and Management of Status Epilepticus, Neurocrit care, 2012) |
· Consider vitamin-dependent diseases (pyridoxine or pyridoxal-phosphate) in the early myoclonic epilepsy group
· Young children with epilepsy (under 3 years) who develop SE lasting 30 minutes, especially if myoclonic should receive pyridoxine trial in case they have pyridoxine dependent/responsive seizures. |
Expert opinion class 4
Level C
|
2.4 Levetiracetam |
||
|
Evidence-Based Guideline: Treatment of Convulsive Status Epilepticus in Children and Adults: Report of the Guideline Committee of the American Epilepsy Society, 2016
Treatment of convulsive status epilepticus in childhood: Recommendations of the Italian League Against Epilepsy - ITALIAN LEAGUE (LICE) 2013 |
· IV Levetiracetam or oral through a nasogastric tube could be given in the second phase therapy for SE
· This drug can be used for continued oral therapy, does not cause significant side effects, and needs no preliminary check of renal function; |
Level U
Level 4 |
3. Overall Seizure Care Pathway
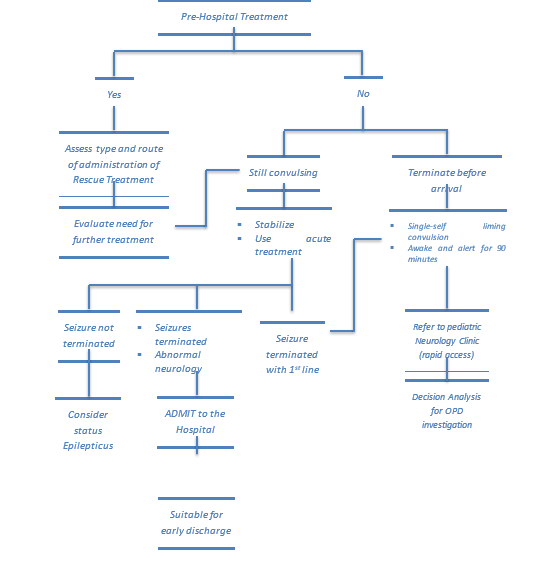
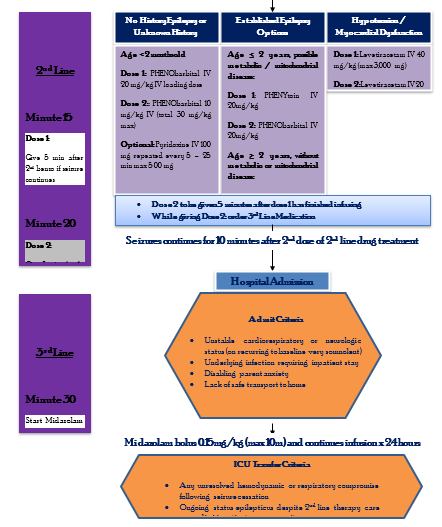
4. Detailed National Seizure Care Pathway

|
Assess Risk of Meningitis or Intracranial Infection |
|
Acute Seizure Management: Diagnostic Tests |
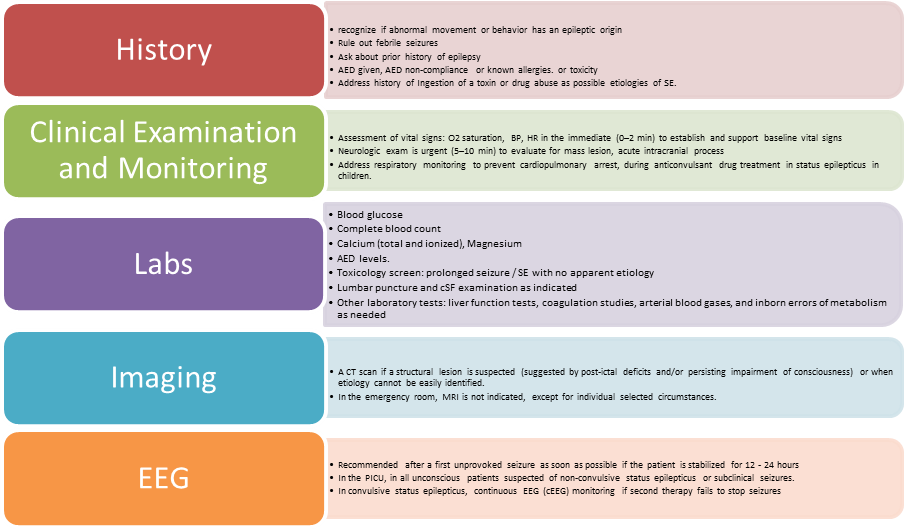
Lumbar puncture (LP) should be performed in:
1. The very young child (<6 months),
2. The child of any age with persistent (cause unknown) alteration of mental status or failure to return to baseline,
3. Any child with meningeal signs.
If increased intracranial pressure is suspected, the LP should be preceded by an imaging study of the head
|
Assess Risk of Meningitis or Intracranial Infection |
||
|
History · >3 days duration of illness · Seen by the previous doctor in previous 24 hours · Drowsiness or vomiting at home · Infants 6 – 12 months old deficient in Hib or pneumococcal vaccines or immunization status cannot be determined · Pretreated with antibiotics |
Physical Signs · Petechiae · Questionable nuchal rigidity · Drowsiness · Convulsing on examination · Weakness or neurological deficit on examination · Signs of infection of head or neck with potential for intracranial extension (e.g. mastoiditis, sinusitis..etc) · Bulging anterior fontanelle |
Complex Features · Focal seizures · Seizure duration > 15 minutes · Multiple seizures in 24 hours
Meningitis Less Likely · Prior febrile seizures · Pre-existing neurological findings |
|
Rationale for Use of Antiepileptic Drugs |
|
Benzodiazepines (1st Line: 2 doses) |

|
2nd Line Medications |
· Phenytoin followed by phenobarbital is recommended if benzodiazepines fail (Level 2B, Grade B).
· Phenobarbital is recommended if no benzodiazepines available (Level A).
· Phenytoin and phenobarbital are not used sequentially but alternating. Generally, phenytoin precedes phenobarbital based on better safety profile (Level B)
· IV valproate is used as an alternative based on its availability especially in tertiary care centers (Expert Opinion)
· IV valproate is used with extreme caution especially in young children when a metabolic etiology has been ruled out (Level 1B, Grade A)
· IV levetiracetam or through a nasogastric tube is an alternative (Level U). It can be used for continued oral therapy, does not cause significant side effects, and needs no preliminary check for renal function (Level 4, Grade C).
|
Consider 2nd Line IV bolus with drug patient is already on Dose 2 to be given 5 minutes after dose 1 has finished infusing |
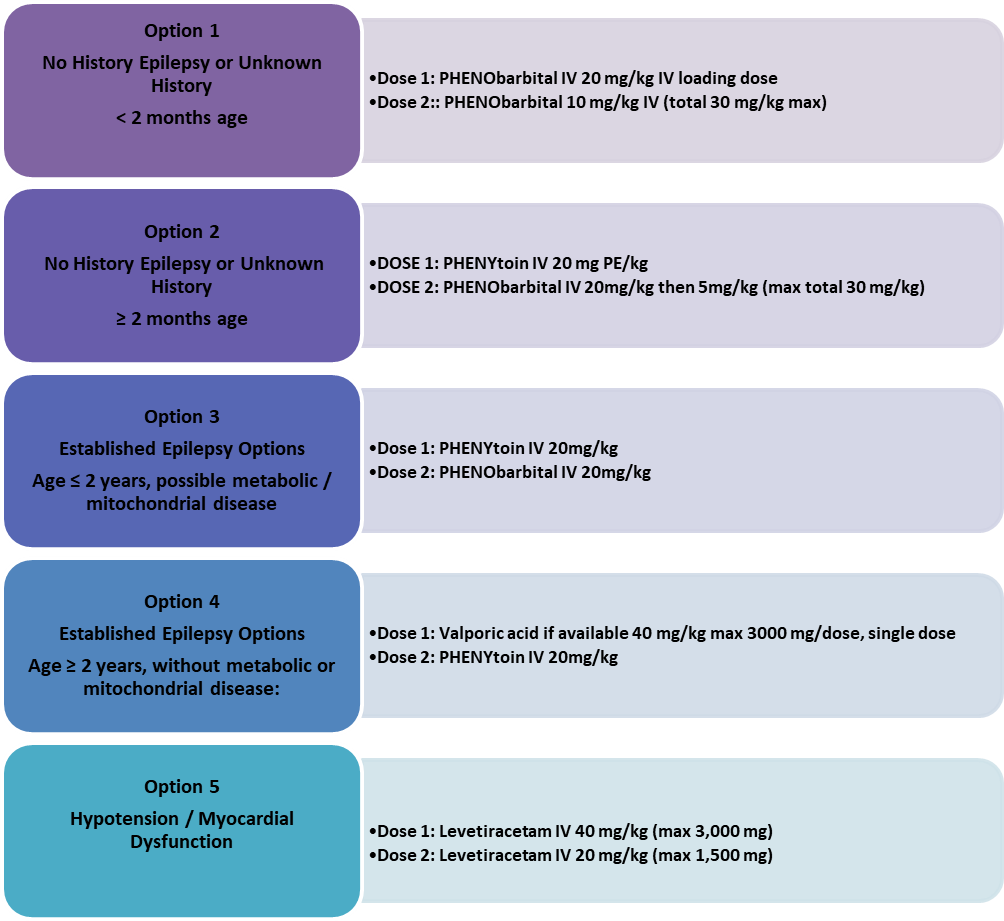
|
Pre-hospital Treatment |
- As early as possible (Level 1B – Grade A and Level 2 – Grade B)
- Better by trained personnel (medical/paramedical) or by a caregiver.
- Drugs of Choice:
- Midazolam:
- Intra-muscular:
|
13 – 40 kg: |
5 mg |
|
>40 kg: |
10 mg |
- Buccal: 0.5 mg/kg (max 10 mg)
- Diazepam:
- Per-rectal:
|
2 – 5 years: |
0.5 mg/kg |
|
6 – 11 years: |
0.3 mg/kg |
|
>12 years: |
0.2 mg/kg |
|
Buccal Midazolam
|
It can be given by the “Syringe Method” where the IV preparation 5 mg/ml trickled from a syringe into which the midazolam has been drawn up using insulin syringe to deliver 1 ml i.e. 5 mg midazolam.
|
Buccal Midazolam |
Buccal Midazolam – Syringe Method:
|
Step 1: Open the ampoule of midazolam |
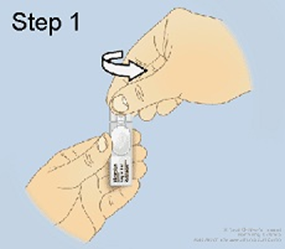
|
|
Step 2: Insert the syringe into the ampoule and hold the ampoule and syringe upright with the ampoule above the syringe. Withdraw the required dose as prescribed by your doctor into a 1mL syringe. Push out any big air bubbles and check the amount of midazolam in the syringe. Remove the ampoule from the syringe. |
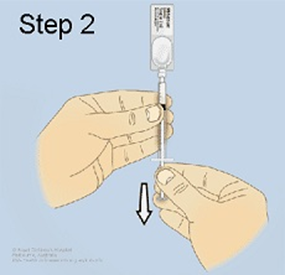
|
|
Step 3: Lay your child on their side (recovery position). Open their lips and slowly trickle the midazolam from the syringe into the inside of the lower cheek The calculated dose is applied to the buccal cavity between the gum and the cheek. Massage the cheek gently after administration. There is no need to open your child's jaw. The solution does not need to be swallowed. |
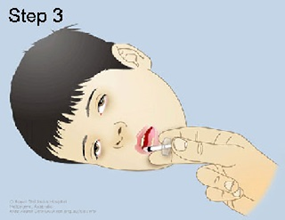
|
|
Step 4: Watch your child's breathing and seizure activity while they remain lying on their side in the first aid position. |
|
|
Step 5: Write down the time that the seizure started, when the midazolam was given and when the seizure stopped. |
|
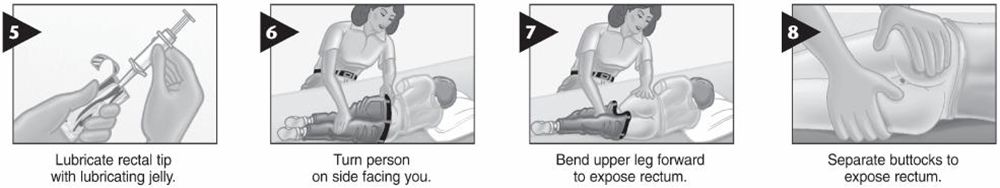
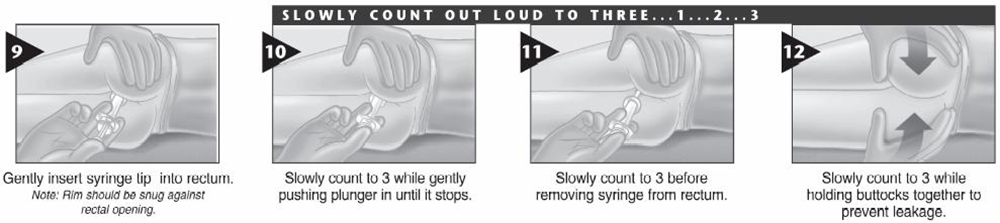
It can be given by the “Syringe Method” where the IV preparation 5 mg/ml trickled from a syringe into which the diazepam has been drawn up using insulin syringe to deliver 1 ml i.e. 5 mg diazepam.

Rectal Diazepam – Syringe Method:
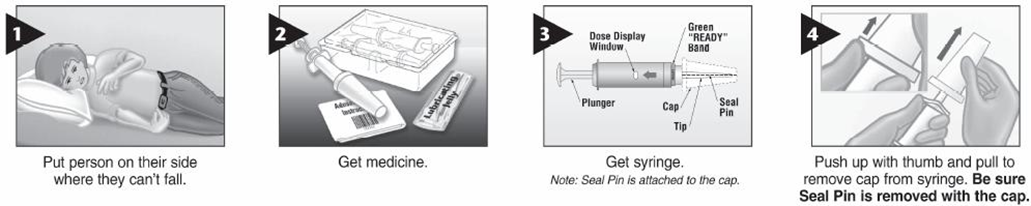
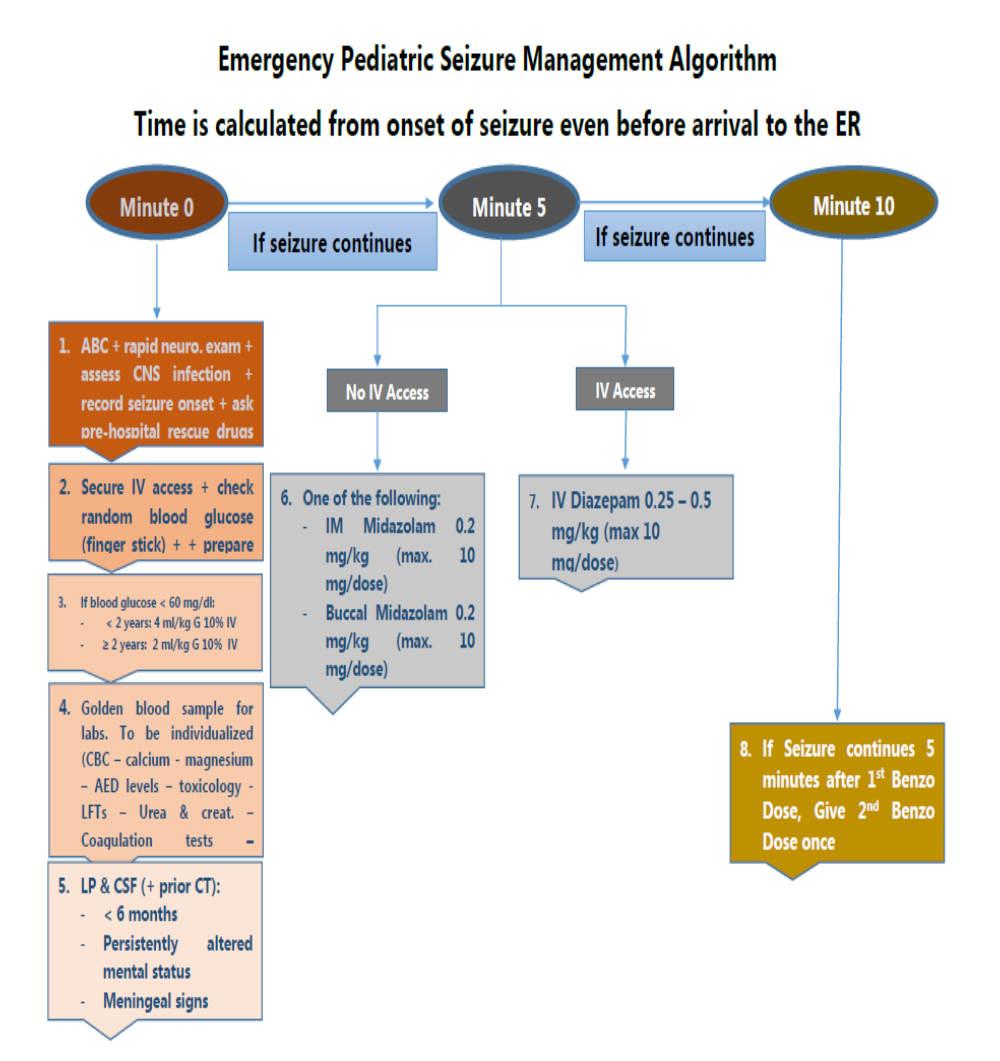
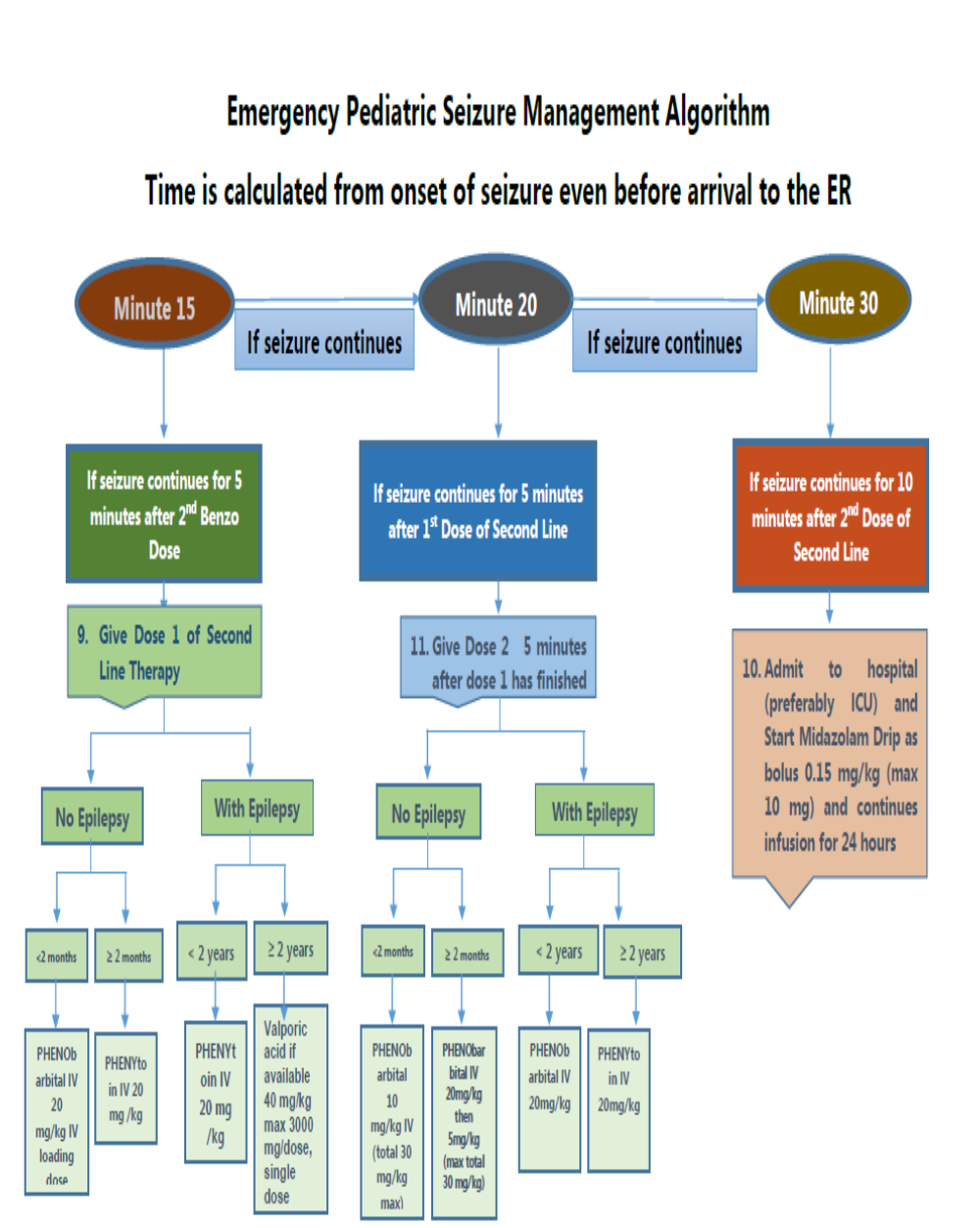
5. Emergency Seizure Management Algorithm
6. Drug Table
7. Table: Intermittent Drug Dosing in Emergency Seizure and Status Epilept
Management
|
Drug |
Initial Dosing |
Administration Rates and Alternative Drug Dosing Recommendations |
Serious Adverse Effects |
Considerations |
|
Diazepam |
IV 0.25 – 0.5 mg/kg max 10 mg/dose, may repeat in 5 min |
IV: Up to 5 mg/min (IVP), maximum 10 mg/dose
Pre-Hospital Treatment: - 2–5 years, 0.5 mg/kg (rectal); - 6–11 years, 0.3 mg/kg (PR); - greater than 12 years, 0.2 mg/kg (rectal) - maximum 20 mg/dose |
Hypotension Respiratory depression signs (hypo-ventilation, pallor, Decrease oxygen saturation. Respiratory support and intubation may be needed). |
- diazepam could be repeated once after 5 minutes convulsion continued - History of pre-hospital use of benzodiazepines must be considered as well as timing. - doses given within one hour before ER presentation must be regarded as an initial dose - Rapid redistribution (short duration), the active metabolite, - IV contains propylene glycol |
|
Midazolam |
0.2 mg/kg IM up to maximum of 10 mg |
10 mg IM (>40 kg); 5 mg IM (13–40 kg);
0.2 mg/kg max 10 mg/dose, (buccal)
|
Hypotension Respiratory depression signs (hypo-ventilation, pallor, Decrease oxygen saturation. Respiratory support and intubation may be needed). |
- Midazolam is the most appropriate type of benzodiazepines to be used - The whole full dose to be given as a single dose better than broken into multiple doses) - The active metabolite, renal elimination, rapid redistribution (short duration) |
|
Phenytoin |
Initial loading dose :20 mg/kg IV, may give an additional 5–10 mg/kg, maximum 1500 mg/dose 10 minutes after the initial loading dose
Maintenance dose: 4-8 mg/kg/day IV divided twice daily |
up to 1 mg/kg/min (if<50kg: give over 20 min, if >50 kg give at rate 50 mg/ min
|
Arrhythmias Hypotension Purple glove syndrome |
- It is preferred to phenobarbital as it has fewer side effects in terms of cardiovascular depression, sedation, and respiratory depression than phenobarbital given after benzodiazepines. - It should be infused using independent venous access in a large-caliber vessel to reduce the risk of phlebitis. It is recommended to monitor heart rate and blood pressure( cardiac arrhythmias, hypotension). Purple glove syndrome, Stevens-Johnson - Phenytoin is contraindicated in patients with Grade II atrioventricular block or severe hypotension - Only compatible in saline in a concentration not exceeding 10 mg/ml - IV contains propylene glycol - If already on phenytoin, halve the loading dose |
|
Phenobarbitone |
20 mg/kg IV, may give an additional 5–10 mg/kg
|
50–100 mg/min IV, may give additional dose 10 min after loading infusion
Must not be faster than 1 mg/ kg/ min (at least 20 min), Must be diluted with water to a concentration, not exceeding10 mg/ ml
(If <30 kg infuse over 20 min) , if > 30 kg infuse at 30 mg / min |
Hypotension Respiratory depression |
- IV contains propylene glycol - If already on phenobarbitone, halve the above loading dose of that drug. - Monitoring of heart rate, electrocardiography, and blood pressure for risk of respiratory depression and hypotension. It should be administered with a resuscitator and/ or trained medical personnel to support advanced ventilation - Respiratory depression is the most clinically significant treatment-emergent adverse event with anticonvulsant drugs in status epilepticus |
|
Valproate |
20–40 mg/kg IV, may give an additional 20 mg/kg |
1.5–3 mg/kg/min Maximum 3000 mg/dose. Single-dose |
Hyperammonemia Pancreatitis Thrombocytopenia Hepatotoxicity |
- Use with caution in patients with traumatic head injury; maybe a preferred agent in patients with glioblastoma multiforme |
|
Pyridoxine |
Loading: administering 100 mg of pyridoxine intravenously If clinical response is not demonstrated, the dose should be repeated every 5 – 25 min up to a maximum of 500 mg.
|
while monitoring the EEG, oxygen saturation, and vital signs In individuals with pyridoxine-dependent epilepsy, clinical seizures generally cease over several minutes. A corresponding change should be observed in the EEG; in some circumstances, the change may be delayed by several hours. |
In some individuals with pyridoxine-dependent epilepsy, significant neurologic, and cardiorespiratory depression follows this trial, making close systemic monitoring essential.
|
|
|
Levetiracetam |
20–60 mg/kg IV twice daily
|
bolus of 13–70 mg/kg – 60 mg/kg, maximum 4500 mg/dose, single-dose, (maximum dose 4 g), typical starting dose 30 mg/kg in 15-min intravenous infusion (from 5 to 60 min) for a total of 100 ml (but at high concentration and low volume: 50 mg/ml) Keppra Injection is for intravenous use only and should be diluted in 100 mL of a compatible diluent before administration. If a smaller volume is required (e.g. pediatric patients), the amount of diluent should be calculated to not exceed a maximum levetiracetam concentration of 15 mg per mL of diluted solution. Consideration Diluents: Sodium chloride (0.9%) injection, USP Lactated Ringer's injection Dextrose 5% injection, USP 2–5 mg/kg/min IV |
The following adverse reactions may be observed: - Behavioral Abnormalities and Psychotic Symptoms - Somnolence and Fatigue - Anaphylaxis and Angioedema - Serious Dermatological Reactions - Coordination Difficulties - Hematologic Abnormalities - Increase in Blood Pressure |
- Minimal drug interactions - Not hepatically metabolized - Levetiracetam in the oral formulation can be administered through a nasogastric tube. - In cases where it was effective, seizures stopped in 25– 30 min after intravenous injection and in 1.5 days when given through a nasogastric tube. - The following calculation should be used to determine the appropriate daily dose of Keppra Injection for pediatric patients - Total daily dose (mL/day) = Daily dose (mg/kg/day) × patient weight (kg)/ 100 mg/mL |
