
"last update: 3 Nov 2025" Download Guideline
- Recommendations
PICO 1: What are the diagnostic criteria of GBS?
Requirements for the diagnosis of sensory- motor or motor GBS are:
• Bilateral flaccid paralysis of extremities
• Absent or decreased deep tendon reflexes in affected limbs
• Progressive worsening for no more than 4 weeks (strong recommendation, high grade evidence)
Evidence Summary:
Features that support diagnosis:
1-Relative symmetry, 2-Relatively mild/absent sensory symptoms and signs,3- Cranial nerve involvement (especially bilateral facial palsy), 4- Autonomic dysfunction, 5- Respiratory insufficiency (due to muscle weakness), 6- Pain (muscular/radicular in back or limb),7- Recent history of infection (<6 weeks).(16)
To ensure the highest level of diagnostic certainty, additional proof is required in the form of increased protein levels in the CSF, along with a mildly increased cell count, and electrophysiological signs of neuropathy.
The maximal duration of progression originates from large studies showing that progression does not exceed 2 weeks in most patients, and almost never exceeds 4 weeks.(9,10)
2- Cerebrospinal fluid analysis:
PICO2: Cerebrospinal fluid (CSF) examination: In patients with clinically suspected GBS, does examination of the CSF compared with no CSF examination influence the diagnostic accuracy, treatment response and patient outcome?
1- We strongly recommend a diagnostic lumbar puncture with subsequent CSF protein analysis and a cell count particularly when the diagnosis is uncertain or when an alternative diagnosis needs to be excluded (strong recommendation, moderate grade evidence)
2- Results supportive of GBS are an increased CSF protein concentration, and a normal or only slightly increased CSF white blood cell count usually <5 cells/μL, rarely 5–50 cells/μL (known as cyto- albuminous dissociation) (Strong recommendation, moderate grade evidence)
3- A CSF white blood cell count of >50 cells/μL should raise suspicion for alternative diagnoses (Strong recommendation, moderate grade evidence)
4- We suggest that for diagnostic certainty in the event of an initially-normal CSF finding, a lumbar puncture may be repeated 7-10 days after the first symptoms appear, if it is deemed necessary due to unclear clinical or electrophysiological results .(Good practice statement)
Evidence summary:
- Normal CSF protein does not exclude GBS. It should be noted that protein concentration is often normal during the first week, and only increases with disease progression so, diagnostic sensitivity of an increased CSF protein depends on the time CSF is examined after onset of weakness. After 7-10 days, it generally reaches a level above that of the reference value, which notably differs in children according to age [11].
- The specificity of a raised CSF protein is unknown, and it does not rule out some mimics of GBS as CSF protein levels can be elevated in other disorders that are similar to GBS.. Therefore, a normal CSF protein reading does not rule out GBS, while an elevated protein level is not solely sufficient to confirm diagnosis. [12]
- Nevertheless, the CSF cell count is usually normal, although occasionally it can be mildly elevated to <50/mm3, rarely more [13]. In such cases, other differential diagnoses as meningitis should be considered as possible causes [13]. Due to the fact that the GBS-related increase in protein is dependent on time, and the standard CSF value for protein depends on age, the cell count can potentially be more valuable as a differential diagnostic factor than protein level.
- Both CSF protein and cell count may be artefactually raised after IVIG [14]
- Since this is an invasive procedure that can cause discomfort and be stressful for children and often has no therapeutic consequences, it is preferred not to be repeated except if unclear clinical or electrophysiological results. [15]
3- Electrophysiological examination:
PICO 3: In patients with clinically suspected GBS, Regarding the added value of electrophysiological examination?
1- We recommend performing electrophysiological examination for confirming GBS diagnosis as it is necessary for obtaining the highest level of diagnostic certainty (strong recommendation, low grade evidence)
If the diagnostic tool is not available, refer the patient to an appropriate clinical centre..
2- We suggest that for confirmation of diagnosis in the event of an initially normal electrophysiological finding, the procedure may be repeated 1 or 2 weeks later if it is deemed necessary because of other unclear data. (conditional recommendation, moderate grade evidence)
Evidence summary:
Prospective and retrospective studies that evaluated up to 84 patients with clinically suspected GBS or up to 66 AIDP cases with a variable number of controls concluded that numerous nerve conduction parameters are abnormal early after disease onset.(19,20) However, some studies included small numbers of patients and/or did not include controls. Sensitivity and specificity vary according to electrodiagnostic criteria and the control groups used.
A typical electrodiagnostic finding is required in order to reach the highest level of diagnostic certainty described by Sejvar et al. [18] Such examinations can be painful and are often difficult to perform in children. Moreover, the necessary expertise is often lacking in pediatric hospitals. Nonetheless, nerve conduction studies and, to a lesser extent, EMG, are required for distinguishing between GBS variants. Notably, due to the latent appearance of pathological spontaneous activity it only makes sense to perform an EMG 2-3 weeks after onset.
Although electrophysiological criteria for the diagnosis of GBS and its variants have been repeatedly defined, they are not always consistently applied and cannot be considered definitive [21,22].
4- MRI brain and spine
PICO (4) In patients with clinically suspected GBS, does using MRI brain and/or spine compared with not using MRI studies influence the diagnostic accuracy .
We recommend performing MRI of the brain and spinal cord when the available findings are ambiguous or if there is evidence of pathology at either the spinal level (e.g., bladder dysfunction at the onset of disease, definable motor or sensory lesion) or central level (encephalopathy, pyramidal signs). (strong recommendation, low grade evidence)
Evidence summary:
Although a spinal MRI is primarily carried out to exclude space-occupying lesions or any other pathology, a contrast enhanced MRI procedure could also support the diagnosis of GBS. A number of publications has reported pathological uptake of contrast agent, particularly by the ventral nerve roots and cranial nerves, hence suggesting that a contrast-enhanced MRI could serve as a diagnostic criterium for GBS in the face of persistently unremarkable CSF and nerve conduction findings.(23,24)
In a retrospective study by Yikilmaz et al. 38 of 40 patients with childhood GBS showed contrast enhancement of the nerve roots; Mulkey et al. reported 22 of 24 patients with spinal nerve root enhancement in an initial MRI(25,26). However, increased uptake of contrast agent is also possible in other inflammatory conditions (serous meningitis, neuroborreliosis) and neurometabolic disorders (Krabbe disease). Since prospective examinations have not yet been carried out, the sensitivity and specificity of contrast-enhanced MRI remains unclear in children and adolescents with GBS.
5- Antecedent events :
PICO (5) In patients with clinically suspected GBS, does enquiring about antecedent events ( infection or vaccination) compared with not enquiring about antecedent events influence the diagnostic test accuracy, treatment response and patient outcome?
Infection:
We suggest that microbiological and serological diagnostics may be carried out as the diagnosis of GBS is more likely if there is a history of recent infection. Asking about these antecedent events may increase diagnostic confidence and is generally more useful when present than absent (Conditional recommendation, moderate grade evidence)
Evidence Summary:
About two- thirds of patients who develop GBS report symptoms of an infection in the 6 weeks preceding the onset of the condition (27) These infections are thought to trigger the immune response that causes GBS. In childhood GBS, a preceding infection is identified as the potential causative agent in 60-70% of cases.
Six pathogens have been temporally associated with GBS in case–control studies: Campylobacter jejuni, cytomegalovirus, hepatitis E virus, Mycoplasma pneumoniae, Epstein–Barr virus and Zika virus.(28,29) It has been suggested that other pathogens are linked to GBS on the basis of evidence from case series or epidemiological studies, but their role in the pathogenesis of GBS is uncertain.
GBS is not or marginally increased in incidence after SARS- CoV- 2 infections although large case– control studies are lacking, from the epidemiological and cohort studies that exist.(30,31)
In general, the absence of an antecedent illness does not exclude a diagnosis of GBS, as putative infections or other immunological stimuli can be subclinical.
Vaccination :
We recommend thorough microbiological and serological diagnostic tests if GBS occurs within 1-6 weeks of vaccination for investigating a potential link with the vaccinating antigen, as well as with alternative causal agents. (Strong recommendation, moderate grade evidence)
Evidence Summary:
The results arising from microbiological and serological diagnostic tests can have substantial legal implications at the social and health care levels.
GBS has been associated temporally with numerous vaccines; however, such temporal association must be differentiated from causality. In general, specific biological markers indicative of a cause-and-effect association with a particular pathogen or vaccine are absent.(32)
Well-conducted case-control studies have been able to show that most vaccines have a very unlikely causal link to GBS. The only exception comes from a swine flu vaccine that was used in an American immunization campaign in 1976-77 and led to a significant increase in the number of GBS cases. This effect was not observed again in later immunization campaigns, even after vaccination during the recent swine flu pandemic in 2009-10.(33)
Furthermore, an accumulation of GBS cases was observed 30-40 years ago following immunization with a rabies vaccine that was grown on mammalian brain tissue. This, however, has not been observed in association with modern rabies vaccines.(34)
6-Serum antibodies
PICO (6) In patients with clinically suspected GBS, does testing for serum antibodies compared with not testing for these antibodies influence the diagnostic test accuracy, particularly for GBS subtypes such as AMAN and Miller Fisher syndrome?
We don’t advise to do routine testing for serum antibodies against gangliosides in most patients with motor-sensory GBS, because of their low–moderate sensitivity and frequent delay in reporting results of antibody assays beyond the therapeutic window (good practice statement)
We suggest testing for serum antibodies against GQ1b in patients suspected to have MFS (or MFS spectrum) especially, when there is some clinical doubt and test results can be obtained within reasonable time (Conditional Recommendation, moderate grade evidence )
Summary of evidence:
Serum antibodies that have been tested in GBS cases include GM1, GM1b, GD1a, GQ1b, GalNAc- GD1a, GT1a, GD1b, GD3, O-GD3, GT3, O-GT3, sulfatide, galactocerebroside, CNTN1, NF155 and NF186, cardiolipin, LM1, sulphated glycolipids, P0 and PMP22. The test accuracy varies depending on GBS subtype, tested antigen and control group. (35,36)
In GBS, the sensitivity of anti-ganglioside antibody panels is reported to vary between 32% and 64%, depending on the presence of a recent infection or GBS subtype (AIDP or AMAN).(37)
For MFS, sensitivity for anti-GQ1b antibodies is high (88%–100%), with a very high specificity (100%). Especially, when there is some clinical doubt and test results can be obtained within reasonable time, testing for anti-GQ1b antibodies is considered helpful. (38,39)
1- Therapeutic recommendations:
PICO (7) In patients with clinically suspected GBS ,Is treatment with intravenous immunoglobulins (IVIG) recommended in children with GBS?
We recommend starting IVIg as soon as possible in patients unable to walk unaided (GBS- DS grade 3 or more) if still within the first 2 weeks from onset of weakness ( strong recommendation- high level of evidence)
• Using the most frequently used and proven effective standard course of IVIg (0.4 g/kg/day for 5 days) rather than a low- dose (0.4 g/kg/day for 3 days) or a high- dose (0.4 g/kg/day for 6 days) regimen or a 2- day regimen (1 g/kg/day) (good practice statement)
We advise to start IVIg (or PE) also in patients who are still able to walk unaided (GBS- DS grade 2) within 4 weeks from onset of weakness, but who have a fast rate of deterioration, a risk of requiring ventilatory support, swallowing difficulties, autonomic disturbances or poor prognostic factors (good practice statement)
Summary of evidence :
In the absence of systematic dose-ranging study data, IVIG is usually administered in children as it is in adults with acute GBS, namely with a single cycle of 2 g/kg body weight distributed over 4 - 5 consecutive days. Reducing the period of IVIG administration to 2 g/kg in 2 days did not show any improved effects but was associated with a higher frequency of relapses [40].
The most common side-effects were headache (12.8% of infusions), fatigue (5.2%), stomach-ache (2.3%) and myalgia (2.3%). Acute side-effects such as skin rash, fever, bronchospasm and thoracic pain occurred more rarely (0.3-0.6%) [41].
An initially progressive disease course with a subsequent plateau phase without recovery is classified as severe GBS, for which the optimal treatment approach remains unclear . However, in children a satisfactory recovery can still be expected even after a prolonged plateau-phase of up to 90 days[42].
8-Plasmapheresis, plasma exchange
PICO ( 8) Is treatment with plasmapheresis recommended in children with GBS?
The same indications as listed in the IVIG in cases when IVIG is not available. We recommend four to five exchanges over 1– 2 weeks with a total exchanged volume of 12– 15 L in patients who are severely disabled (unable to walk unaided, bedridden or ventilated). (strong recommendation, high grade evidence)
We suggest PE (four to five exchanges over 1– 2 weeks) in patients who are still ambulatory, but who have a fast rate of deterioration, a risk of requiring ventilatory support, swallowing difficulties or other poor prognostic factors. These patients are considered at high risk of further deterioration, which may potentially be prevented by starting treatment early( good practice statement)
Summary of evidence
The best effect of PE is seen when therapy is started within seven days of symptom onset, although an effect can still be detected if applied up to 30 days post-onset.
The given dosages for PE mostly comprise 4-5 cycles with an exchange volume of 200-250 ml/kg body weight over 7-14 days.
Albumin is a more favorable exchange fluid than FFP [43].
There is no preference for treatment with either IVIg or PE. Therefore, the choice may depend on the availability or the experience.(50) However, IVIg is generally associated with few adverse events and is readily available in most hospitals. PE requires special facilities, good intravenous access and has a slightly higher adverse event (AE) rate [42].
The combination of PE followed by IVIG did not yield any significant additional benefits, although an effect in individual patients cannot be ruled out.
Application of PE within 1-2 weeks of the preceding IVIG therapy is not indicated.
PICO (9). In patients diagnosed with GBS, does treatment with corticosteroids (IV or oral), alone or in combination with IVIg or PE, influence the disease course ?
We don’t recommend the use of oral corticosteroids or IV methylprdnisone (IVMP) for the treatment of GBS.(strong recommendation, moderate level of evidence)
Summary of evidence:
There is a probable lack of efficacy of IVMP (moderate certainty evidence from a trial with 242 patients),(44)
The probable lack of efficacy of the combination of IVIg and IVMP compared with IVIg and placebo (moderate certainty evidence from a trial with 225 patients)(45)
The probable harm (delayed recovery) of oral corticosteroids (low certainty evidence from 4 trials with a total of 120 patients) using various oral regimens of the equivalent of 40 mg prednisolone daily for at least 2 weeks, and the high certainty evidence of adverse effects (diabetes more common), despite that hypertension was less common in the corticosteroid-treated patients, which is crucial in decision making. (46,47,48)
PICO (10 ). Does treatment with pharmacological therapies other than PE, IVIg (as eculizumab, mycophenolate, interferons or other) influence disease course ?
We don’t recommend the use of immunosuppressive treatment approaches as an alternative to immunomodulation with plasmapheresis and IVIG. (conditional recommendation, low grade evidence)
Summary of evidence:
As there is evidence for complement activation from pathological studies and from animal models of GBS.(53) Eculizumab, a complement blocking agent, was beneficial in an animal model.(54)
In two small phase 2 trials (including a total of 41 patients), no beneficial effects of eculizumab could be demonstrated (very low to low certainty evidence).(55)
PICO (11) In patients diagnosed with GBS, what is indication of ICU admission and mechanical ventillation?
We advise using a prognostic model at hospital admission to quantify the risk of requiring mechanical ventilation. This can be quantified using the modified Erasmus GBS Respiratory Insufficiency Score (mEGRIS)( table5) OR
Using the following risk factors for requiring mechanical ventilation during hospital admission. These include:
- Rapid progression of limb weakness during hospital admission
- GBS- DS grade 4 (unable to walk 5 m even with aid)
- Neck flexion, facial or bulbar weakness
- The inability to cough
- Autonomic instability such as fluctuations in blood pressure or heart rate. (good practice statement)
• We advise regularly assessing any decline in respiratory function by measuring forced vital capacity (FVC), and single breath count (SBC). (good practice statement) Fig 2
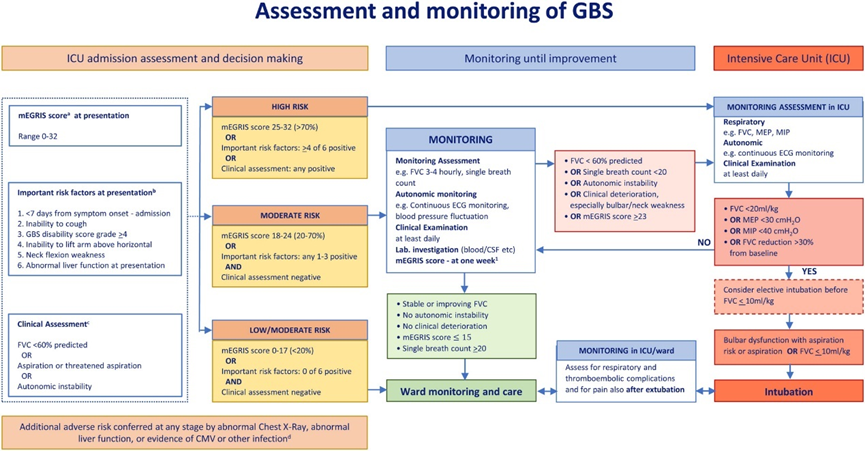
Fig.2: Assessment and monitoring of GBS
Frequent monitoring of the following quantitative measures of ventilatory function:
a- FVC should be checked between three and six times a day, depending on severity, and until significant worsening seems unlikely. While the patient is still declining and there is a reduction in FVC, 4- hourly monitoring is likely appropriate.
b- A fall of the FVC > 30% below the predicted baseline should alert concern, a fall of >30% in 24 h likely indicates immediate transfer to ICU, or a 50% decline in under 24 h likely indicates the need for ventilation.
c- Single breath count <20 (inability to count in a single breath out loud from 1 to 20 indicates the need to transfer to ICU.
Summary of evidence :
According to the published case series on GBS in childhood, 15-25% of patients were dependent on mechanical ventilation. Respiratory failure in children particularly needs to be reckoned with when the disease progresses quickly and the upper extremities and cranial nerves are affected (56,57) Moreover following discharge from the ICU the possibility of secondary deterioration in the ensuing weeks should be kept in mind.
PICO (12). In patients initially diagnosed with GBS, does the presence of certain clinical symptoms or laboratory features compared with their absence predict the subsequent diagnosis of A-CIDP as confirmed by neurological worsening at >8 weeks from onset?
We suggest changing the diagnosis from GBS to A-CIDP after a few weeks from onset in some patients initially diagnosed with GBS, especially if the patient worsens after initial improvement or stabilization (known as a treatment-related fluctuation, TRF), or presents as mild or slowly progressive GBS and continues to worsen.(conditional recommendation, low grade evidence)
• A-CIDP is more likely if there are three (or more) TRFs.
• A-CIDP cannot be confirmed unless there is further worsening at least 8 weeks after onset.
• In case of a TRF, consider re-treatment with IVIg or PE.(good practice statement)
Evidence summary:
In a prospective study of 170 patients initially diagnosed as GBS, 16 (9%) had a TRF and another 8 (5%) were subsequently diagnosed with A-CIDP (58). Confirmation of these results however warrants additional studies in larger numbers of patients.
A-CIDP is likely in patients initially diagnosed with GBS, if there is further worsening after 8 weeks from onset (sensitivity 100%, specificity 92%)(51,52), or when there are three (or more) TRFs (episodes of worsening following treatment-induced improvement/stabilisation) (sensitivity 52%, specificity 96%) (49,59).
Reduced MNCV < 90% of lower limit of normal (or <85% if small distal CMAP) was more frequently found in A-CIDP than in GBS . Patients with A-CIDP tested with nerve US within 4 weeks of onset had greater enlargement of peripheral nerves compared with those with GBS (sensitivity 88%, specificity 84%) (60).. Some patients suspected to have A-CIDP may have an autoimmune nodopathy (61). These patients have a poorer response to conventional therapies for CIDP, and there is anecdotal evidence that these patients may response to rituximab. There is no evidence from an RCT, but observational data indicate that a repeated course of IVIg or PE can be effective in case of a TRF(62)
Rationale: It is important to diagnose A-CIDP because treatment differs from GBS. A-CIDP however should not be overdiagnosed in severely weak patients with slow or no improvement. If in doubt, the presence of muscle wasting and denervation on electromyography indicates that GBS-related axonal degeneration is more likely than A-CIDP.
