
- Appendix 1
Major study results
The Diabetic Retinopathy Study (DRS) was designed to investigate the value of laser photocoagulation surgery for patients with severe nonproliferative diabetic retinopathy (NPDR) and proliferative diabetic retinopathy (PDR). The results are shown in Table A4-1.
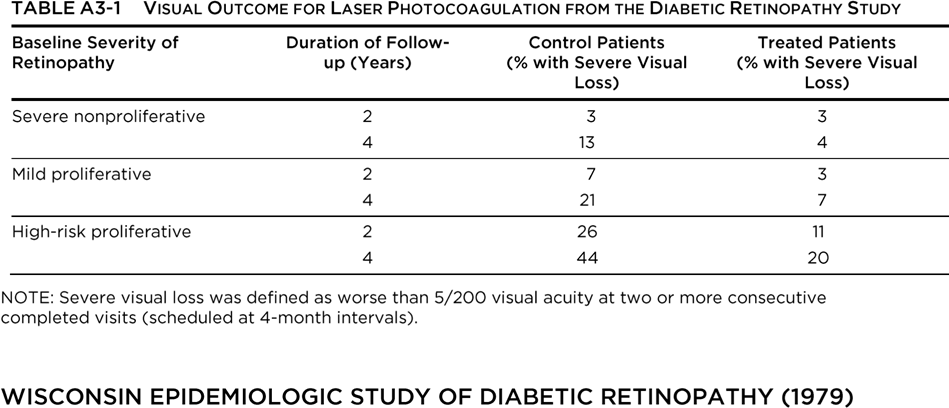
The Wisconsin Epidemiologic Study of Diabetic Retinopathy (WESDR) began in 1979. It was initially funded by the National Eye Institute, which is part of the National Institutes of Health. The purpose of the WESDR is to describe the frequency and incidence of complications associated with diabetes (eye complications such as diabetic retinopathy and visual loss, kidney complications such as diabetic nephropathy, and amputations), and to identify risk factors (such as poor glycemic control, smoking, and high blood pressure) that may contribute to the development of these complications.
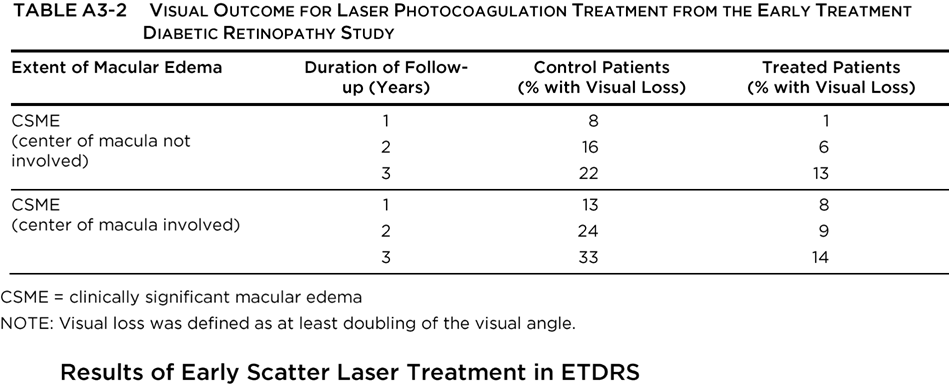
The Early Treatment Diabetic Retinopathy Study (ETDRS) investigated the value of photocoagulation surgery for patients with NPDR or PDR without high-risk characteristics. The results for eyes with macular edema are shown in Table A4-2. Visual loss was defined as at least doubling of the visual angle (e.g., 20/20 to 20/40, or 20/50 to 20/100). In eyes with NPDR or non-high-risk PDR, early panretinal photocoagulation (PRP) was compared with deferral of photocoagulation, and although there was a beneficial treatment effect, the outlook for maintaining vision was good in both groups. The 5-year rates of severe visual loss or vitrectomy ranged from 2% to 6% in eyes assigned to early photocoagulation and from 4% to 10% in eyes assigned to deferral. Early PRP was associated with side effects (small decreases in visual acuity and visual field) in some eyes, and the ETDRS concluded that deferral of photocoagulation was preferable at least until retinopathy was approaching the highrisk stage. Eyes approaching that stage had a 50% risk of reaching it within 12 to 18 months. Eyes in this category had very severe NPDR or non-high-risk PDR characterized by NVD less than one-quarter to one-third disc area and/or NVE, without vitreous or preretinal hemorrhage.
Recent additional analyses of visual outcome in ETDRS patients with severe NPDR to nonhigh-risk PDR suggest that the recommendation to consider PRP before the development of high-risk PDR is particularly appropriate for patients with type 2 diabetes. The risk of severe vision loss or vitrectomy was reduced by 50% in patients who were treated early compared with those who deferred treatment until high-risk PDR developed.
For patients with type 1 diabetes, the timing of the PRP will depend on the compliance with follow-up, status and response to treatment of the fellow eye, impending cataract surgery, and/or pregnancy status.
The Diabetic Retinopathy Vitrectomy Study (DRVS) investigated the role of vitrectomy in managing eyes with very severe PDR. The benefit of early vitrectomy for severe vitreous hemorrhage (defined as hemorrhage obscuring the macula or major retinal vessels for 3 disc diameters from the macular center) was seen in type 1 patients, but no such advantage was found in type 2 patients, who did not benefit from earlier surgery. Early vitrectomy was beneficial among patients with visual acuity of 5/200 or worse and severe vitreous hemorrhage with reduced vision for at least 1 month and without previous treatment or complications such as retinal detachment or neovascularization of the iris. Overall, at 2 years after surgery, 25% of the early vitrectomy group and 15% of the deferral group had visual acuity of 20/40 or better. The advantage was most pronounced in patients with type 1 diabetes (36% vs. 12% for early vitrectomy versus deferral of vitrectomy, respectively) and was not statistically significant for patients with type 2 diabetes.
The DRVS showed that early vitrectomy was beneficial for patients with visual acuity of 20/400 or better plus 1 of the following: (1) severe neovascularization and fibrous proliferation; (2) fibrous proliferation and moderate vitreous hemorrhage; or (3) moderate neovascularization, severe fibrous proliferation, and moderate vitreous hemorrhage. Among such patients, 44% with early vitrectomy and 28% in the observation group had visual acuity of 20/40 or better at 4 years of follow-up.
The results of the DRVS should be interpreted in light of subsequent advances in vitreoretinal surgery, such as the introduction of small-gauge vitrectomy technology, endoscopic and indirect ophthalmoscopic laser photocoagulation surgery, and advanced instrumentation. The use of longacting intraocular gases such as sulfur hexafluoride (SF6) and perfluoropropane (C3F8), the use of viscodissection, and the use of heavier-than-water liquids such as perfluoro-octane are advances in vitreoretinal surgery that developed after the DRVS. Thus, the results may actually be better than those reported in the DRVS. Early vitrectomy should be considered for selected patients with type 2 diabetes, particularly those in whom severe vitreous hemorrhage prohibits laser therapy photocoagulation of active neovascularization.
The FIELD study was a randomized controlled trial that evaluated long-term fenofibrate therapy for the reduction of cardiovascular events in 9795 patients with type 2 diabetes mellitus. Fenofibrate did not significantly reduce the risk of the primary outcome of coronary events. It did reduce total cardiovascular events, mainly due to fewer nonfatal myocardial infarctions and revascularizations. The higher rate of starting statin therapy in patients allocated to receive placebo might have masked a moderately larger treatment benefit.
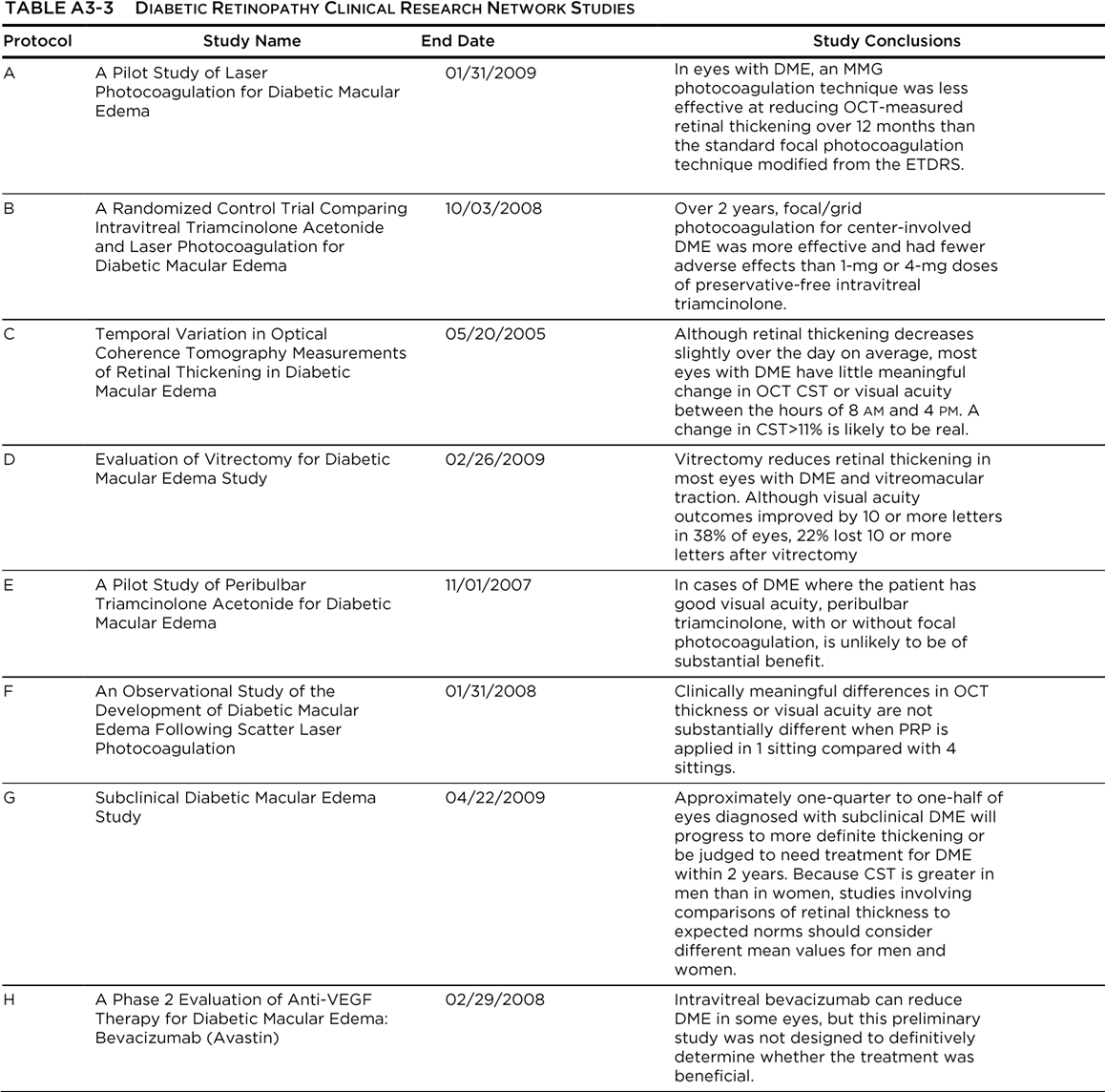
The Diabetic Retinopathy Clinical Research Network (DRCR.net) is a collaborative network dedicated to facilitating multicenter clinical research of diabetic retinopathy, diabetic macular edema
(DME), and associated conditions. The DRCR.net supports the identification, design, and implementation of multicenter clinical research initiatives focused on diabetes-induced retinal disorders. Principal emphasis is placed on clinical trials, but epidemiologic outcomes and other research may be supported as well. The DRCR.net was formed in 2002 and currently includes over 115 participating sites (offices) with over 400 physicians throughout the United States. The DRCR.net is funded by the National Eye Institute (NEI), which is a part of the National Institutes of Health, the branch of government that funds medical research. The DRCR.net has completed multiple clinical trials evaluating the role of anti-vascular endothelial growth factor (anti-VEGF), laser treatment, and corticosteroids in DME, anti-VEGF efficacy in PDR and vitreous hemorrhage, and even diabetes education effectiveness on DME (See Table A3-3). Most importantly, DRCR.net Protocol T (Comparative Effectiveness Study of Intravitreal Aflibercept, Bevacizumab, and Ranibizumab for Diabetic Macular Edema) compared the effectiveness of ranibizumab, aflibercept, and bevacizumab in the treatment of DME.96 This study found that all three drugs resulted in improvement in visual acuity at 1 year with similar safety profiles. However, the mean visual acuity using aflibercept was better for eyes with visual acuity of 20/50 or worse at 1 year. At 2 years, the mean visual acuity in the aflibercept was no longer superior to ranibizumab, although it remained superior to bevacizumab. Another important treatment comparison was done in Protocol I: Intravitreal Ranibizumab for Diabetic Macular Edema with Prompt vs. Deferred Laser Treatment. Three-year results were reported in 2012. The study utilized ranibizumab monthly until improvement no longer occurred (with resumption if the condition worsened) and random assignment to focal/grid laser treatment promptly or deferred (≥24 weeks). The 3-year results suggest that focal/grid laser treatment at the initiation of intravitreal ranibizumab is no better, and worse for vision outcomes, than deferring laser treatment for ≥24 weeks in eyes with DME involving the fovea and with vision impairment.94
A previous publication from Protocol I results confirmed the 1-year results that intravitreal ranibizumab with prompt or deferred laser was more effective through 2 years compared with prompt laser alone for the treatment of DME involving the central macula. Laser was not associated with endophthalmitis, the rare but potentially devastating complication of injecting ranibizumab. In pseudophakic eyes, results with intravitreal triamcinolone plus prompt laser appeared similar to results in the ranibizumab arms and were more effective than laser alone, but the triamcinolone plus prompt laser arm had an increased risk of IOP elevation.189
Most
recently, the DRCR.net Protocol S evaluated the effects of anti-VEGF versus
PRP.291 In a randomized, multicenter, noninferiority trial, 394 eyes
of 305 adults with PDR were randomized to receive either PRP or anti-VEGF therapy.
Ranibizumab 0.5 mg was given at baseline and as frequently as every 4 weeks
based on a structured retreatment design. Eyes in both groups were allowed
ranibizumab if DME was present. In eyes with PDR, ranibizumab was not inferior
to PRP in terms of visual acuity outcomes at 2 years. Mean visual acuity
improvement was +2.8 letters for ranibizumab and +0.2 letters for PRP-treated
eyes (P<0.001). When the totality
of the visual acuity data was included (area under the curve analysis), eyes
given ranibizumab had overall better visual acuity outcomes than eyes treated
with PRP. There was less mean reduction in peripheral visual field (-23 dB vs.
-422 dB; P<0.001) with ranibizumab
than with PRP treatment. The rates for vitrectomy were more frequent (15% vs. 4%;
P<0.001), and DME development was
more frequent (28% vs. 9%; P<0.001)
in the PRP group than in the ranibizumab group. Moreover, rates of active
neovascularization or rates of regression of neovascularization were similar
between the two groups.


The RISE and RIDE trials were parallel phase III multicenter double-masked sham injection– controlled randomized studies conducted at private and university-based retina specialty clinics in the
United States and South America. (See Glossary.)
The phase III results for both studies were published in 2012. The studies utilized monthly intravitreal ranibizumab (0.5 or 0.3 mg) or sham injections, with macular laser available if needed. The study concluded that ranibizumab rapidly and sustainably improved vision, reduced the risk of further vision loss, and improved macular edema in patients with DME, with low rates of ocular and nonocular side effects.
READ-2 was a phase II multicenter randomized controlled trial that compared 0.5 mg injections of ranibizumab versus focal laser treatment over 2 years in patients with type 1 or type 2 diabetes mellitus and DME. Patients randomized to one arm of the trial received ranibizumab at baseline, and at 1, 3 and 5 months after baseline; a second arm received laser treatment at baseline and at 3 months (if needed); the third arm received both ranibizumab and laser treatment at baseline and 3 months. From month 5, all subjects received ranibizumab every 2 months and/or maintenance laser treatment every 3 months.
At 24 months, differences between the groups were not statistically significant, and all groups experienced improved visual acuity. Patients receiving combined ranibizumab and laser treatment required fewer injections than patients receiving ranibizumab alone.
BOLT was a phase II 2-year randomized controlled trial that compared intravitreal 1.25 mg bevacizumab injections and focal laser treatment in patients with persistent DME and visual impairment. Bevacizumab patients received an injection every 6 weeks, whereas laser patients were treated every 4 weeks.
At 2 years, visual acuity results were substantially better in the bevacizumab group compared with the laser group, with significant differences in the proportions of patients gaining 10 letters and 15 letters. No patients lost 10 or more letters in the bevacizumab group, compared with 14% of patients treated
with laser.
These studies compared the efficacy and safety of intravitreal aflibercept injection (IAI) with macular laser photocoagulation surgery for DME. Visual improvement were observed in the IAI treatment regimens over laser control at 52, 100 and 148 weeks. Incidence of adverse events was consistent with the known safety profile of IAI.
The DRCR.net compared the efficacy and safety of bevacizumab, ranibizumab, and aflibercept in a multicentered, randomized clinical trial. At the primary endpoint at 1 year, the mean change in vision was greater for aflibercept than for either of the other two drugs. However, the mean visual acuity changes were dependent on the baseline visual acuity. For eyes with milder visual acuity loss, the drugs resulted in similar visual outcomes (8.0 with aflibercept, 7.5 with bevacizumab, and 8.3 with ranibizumab; P>0.50 for each pairwise comparison). However, for eyes with 20/50 or worse vision, the mean visual acuity in eyes treated with aflibercept had greater improvements in vision (18.9 with aflibercept, 11.8 with bevacizumab, and 14.2 with ranibizumab; P<0.001 for aflibercept vs. bevacizumab, P=0.003 for aflibercept vs. ranibizumab, and P=0.21 for ranibizumab vs.
bevacizumab). There were no significant differences in rates of adverse events. However, at 2 years, the mean visual acuity results were similar for ranibizumab and aflibercept, although aflibercept results remained significantly better than bevacizumab results. There was a slightly higher rate of Antiplatelet Trialists' Collaboration (APTC) events with ranibizumab compared with the other two drugs at the 2-year endpoint. All three drugs improved visual acuity at 2 years, and the number of injections decreased in year 2 compared to year 1.
