
- Lab Diagnosis
Samples are taken from infected gills.
· Presumptive diagnosis
o Isolation, Identification
Branchiomyces spp. could grow on Sabouraud dextrose agar (SDA) media (with duck extract decoction 10%+ citric acid 10%); the primary growth was observed from 2 to 3 days post-incubation at room temperature characterized by whitish raised hairy growth. Microscopically, through a wet mount preparation from the culture growth, the hyphae appeared broad, non-septated, intermingled with each other, and branched at their tips (Fig, 2; A, B, C, D, E).

Fig.2: A) primary growth of Branchiomyces sanguinis growth on SDA media observed from 2 to 3 days post-incubation. B) Typical growth of colonies was noticed from 7 to 9 days showing creamy velvety heaped at the center with hairy in the periphery and firmly attached to the media. C) Wet mount preparation from the culture growth, the hyphae appeared broad, non-septated, intermingled with each other, and branched at their tips. D) A portion of the colony stained with lactophenol cotton blue (LPCB) reveals thick hyphae that are branching at their tips (arrowhead) (Mahboub and Shaheen, 2021). Showed E) Thick hyphae with opalescence organelles (Shaheen unpublished data).
· Histopathological examination
o Gills specimens were examined for histopathological changes. The specimen was rapidly fixed in 10% natural formalin buffered phosphate for 24 hours, washed under running tap water and dehydrated through ascending grads of ethanol. It was cleaned in chloroform and embedded in paraffin wax at 60°C. The sections of 5 μm thickness were stained by Hematoxylin and eosin stain (Eyarefe et al., 2008; Adeshina et al., 2019). Using special stains for fungi (periodic acid Schiff’s (PAS) or silver stains), oomycete hyphae and spores can be easily seen in the gill vasculature and in extravascular gill tissues, (Fig.3, A, B).
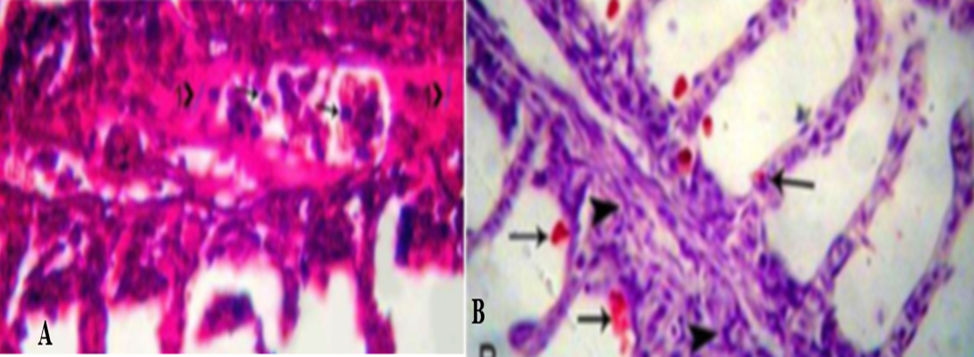
Fig.3: A) Periodic Acid-Schiff -stained paraffin section, s pores (arrowhead) and hyphae (head 1) stain purple and present inside the gill vessels. Also, some secondary lamellae show shortening, others reveal desquamation in the epithelium (head 2). B) H&E-stained section of gills Infected with B. demingrans reveal bright red spores (arrowhead) locate in-between the gill filaments, with aggregation of macrophages, lymphocytes and fibroblasts inside the gill vessels (head) (Mahboub and Shaheen, 2021).
· Confirmatory Diagnosis
· Molecular diagnosis
PCR tests for detection of DNA in samples (a pure isolate of Branchiomycosis sp. On SDA with 10% duck excrement decoction) were performed using two universal primers to ensure that Branchiomyces sp. is related to fungi.
· Control
o Prevention measures
- Quarantine for the newly introduced fish
- Proper drainage, drying, and disinfectant of the pond (Perfect disinfections)
- Increasing water exchange in the Pond.
- Feed well balanced ration.
- Controlling the addition of fertilizers(chemical).
- Addition of copper sulphate in four treatments 2-3 kg/hectare, at monthly intervals starting in mid-May and ending in mid-August.
- Addition of CaO (quick lime) 150-200 kg/hectare, at 2-week intervals during the summer.
o Control measures and treatment
- The dead and dying fish must be removed and burned in a lime pit (burying and burning).
- Avoid moving infected fish to prevent spreading of infection.
- Feeding fish should be stopped.
- Increase the pond water exchange to ensure maximum oxygenation.
- Addition of copper sulfate 2-3 kg/hectare, at monthly intervals starting in mid-May and ending in mid-August (0.3 mg/L),
- Using CaO (quick lime) 150-200 kg/hectare daily and the pH should not exceed 9.0
- The ponds must be drained, dried and disinfected with quick lime.
· Zoonotic importance
No zoonotic importance
