
- Annexes
Annex 1.
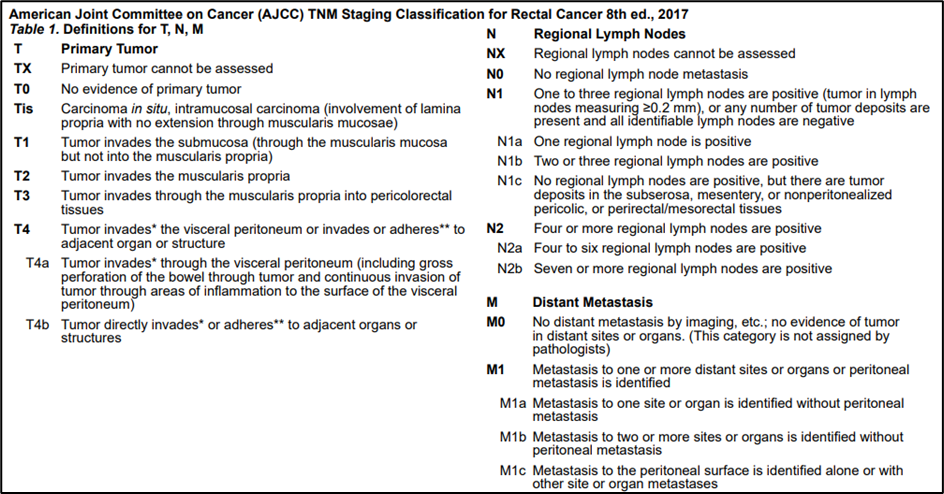
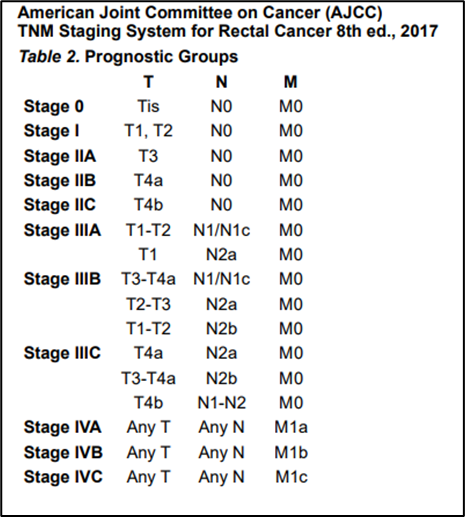
Annex 2.
DOSES OF ADJUVANT CHEMOTHERAPY REGIMENS
- mFOLFOX 6
Oxaliplatin 85 mg/m2 IV, day 1a Leucovorin 400 mg/m2 IV, day 1b
5-FU 400 mg/m2 IV bolus on day 1, followed by 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) continuous infusion.
Repeat every 2 weeks.1,2,3
- Capecitabine
Capecitabine 1000–1250c mg/m2 PO twice daily for 14 days every 3 weeks
- CAPEOX
Oxaliplatin 130 mg/m2 IVa day 1
Capecitabine 1000c mg/m2 PO twice daily for 14 days every 3 weeks
- 5-FU/leucovorin
- Leucovorin 500 mg/m2 given as a 2-hour infusion and repeated weekly x 6. 5-FU 500 mg/m2 given bolus 1 hour after the start of leucovorin and repeated 6 x weekly. Every 8 weeks for 4 cycles.
- Simplified biweekly infusional 5-FU/leucovorin (sLV5FU2)
Leucovorin 400b mg/m2 IV day 1, followed by 5-FU bolus 400 mg/m2, followed by 1200 mg/m2/day x 2 days (total 2400 mg/m2 over 46–48 hours) continuous infusion. Repeat every 2 weeks.
Annex 3.
l When determining if adjuvant therapy should be administered for stage II, the following should be taken into consideration:
- Number of lymph nodes analyzed after surgery (<12)
- Poor prognostic features (eg, poorly differentiated histology [exclusive of those that are MSI-H]; lymphatic/vascular invasion; bowel obstruction; PNI; localized perforation; close, indeterminate, or positive margins; or high tumor budding)

Annex 4. Recommendations from the IDEA study for adjuvant treatment of colon cancer (32).
➡️Diagnostic work-up for localized colon cancer
|
Other additional clinicopathological features such as the histological subtype and grading, lymphatic or venous or perineural invasion, lymphoid inflammatory response, involvement of resection margins and < 12 lymph nodes examined should be taken into consideration for refining the risk assessment on stage II tumours. |
